Surveillance and Control of Aedes aegypti and Aedes albopictus in the United States
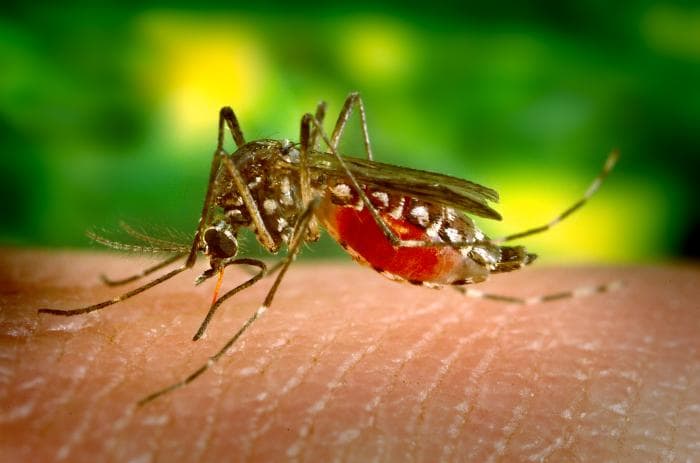
In the continental United States, vector control professionals use integrated vector management strategies to control Aedes aegypti and Aedes albopictus mosquitoes.
- Before a locally acquired case(s) of chikungunya occurs, professionals conduct surveillance to understand local populations of mosquitoes and determine what control efforts are needed.
- If a travel-related case pdf icon[PDF – 1 page] is reported or locally acquired case pdf icon[PDF – 1 page] is suspected, vector control professionals increase mosquito control activities to reduce the number of larvae and adult mosquitoes. This can help keep mosquitoes from biting infected people, which can break the transmission cycle.
- Vector surveillance and control efforts should target mosquito species that can transmit viruses. Control activities are generally similar for Aedes aegypti and Aedes albopictus mosquitoes.
Page last reviewed: January 21, 2022