COVID-19 Science Update released: October 15, 2021 Edition 109

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Section headings in the COVID-19 Science Update have been changed to align
with the CDC Science Agenda for COVID-19.
Here you can find all previous COVID-19 Science Updates.
We honor Hispanic Heritage Month by highlighting scientific manuscripts published during the last month. The manuscripts identify disparate burdens experienced by Hispanic/Latino populations, including essential workers and their families, during the COVID-19 pandemic. The manuscripts also highlight opportunities to improve vaccine uptake, prevention measures, and healthcare access by working together with the rich diversity of Hispanic and Latino people in the United States.
PEER-REVIEWED
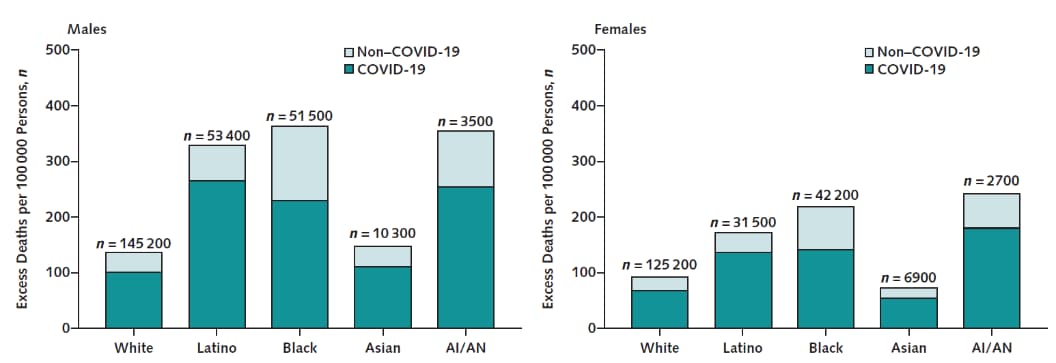
Racial and ethnic disparities in excess deaths during the COVID-19 pandemic, March to December 2020external icon. Shiels et al. Annals of Internal Medicine (October 5, 2021).
Key findings:
- 477,200 excess all-cause deaths occurred in the United States during March–December 2020.
- 74% (n = 351,400) of excess deaths were attributed to COVID-19.
- Excess deaths were ≥2 times higher among Black/African American, Hispanic/Latino, and American Indian/Alaska Native (AI/AN) persons than among White persons (Figure).
Methods: Provisional death certificate and U.S. Census Bureau data were used to calculate age-standardized expected and excess deaths per 100,000 persons, by race, ethnicity and other demographics. Estimates were compared to those in 2019. Limitations: Incomplete death certificate data; no precision estimates around results.
Implications: Excess deaths occurred disproportionately among Hispanic/Latino, Black/African American, and AI/AN persons, likely worsening existing disparities. COVID-19 vaccination and other prevention efforts are important for reversing disparities in mortality.
Figure:
Note: Adapted from Shiels et al. Excess deaths in 2020 from COVID-19 and non-COVID-19 related causes. Excess deaths among males shown on the left and females on the right. Permission request in process.
PEER-REVIEWED
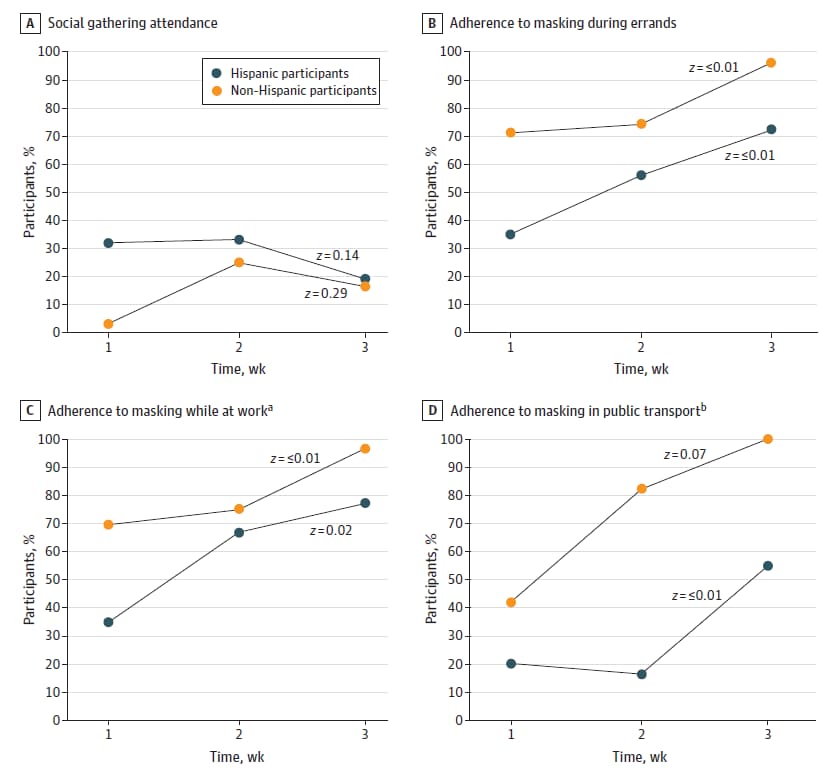
Disparities and temporal trends in COVID-19 exposures and mitigating behaviors among Black and Hispanic adults in an urban settingexternal icon. Badri et al. JAMA Network Open (September 28, 2021).
Key findings:
- Self-reported adherence to limiting social gatherings and compliance with a statewide mask mandate was consistently lower among Hispanic/Latino participants vs. non-Hispanic/Latino participants (Figure).
- Mask wearing increased after the start of a statewide mask mandate.
- Hispanic/Latino participants were less likely to have received a stimulus check from the CARES Act (adjusted OR [aOR] 0.03, 95% CI 0.02-0.07) or unemployment benefits (aOR 0.36, 95% CI 0.16-0.74) compared with non-Hispanic/Latino participants.
Methods: Telephone survey on COVID-19 exposure risks, modifiable behaviors, and economic support among 750 randomly selected persons who had SARS-CoV-2 testing within a safety-net healthcare system in Chicago, July─August 2020. Limitations: May not be generalizable to other cities; survey conducted relatively early in pandemic; potential recall bias (survey conducted 8 weeks after testing).
Implications: Early in the pandemic, Hispanic/Latino persons in this sample were less likely to wear masks or physically distance, suggesting public health communication tailored to Hispanic/Latino populations might improve adoption of prevention approaches. However, disparities in economic assistance might undermine the impact on SARS-CoV-2 prevention and advancement of health equity.
Figure:
Note: Adapted from Badri et al. Reported participant adherence to public health recommendations for Hispanic/Latino participants and non-Hispanic/Latino participants for A) social gathering attendance, B) masking during errands, C) masking while at work, and D) masking in public transport. Superscripts “a” and “b” indicate analysis was performed on subsets who reported working outside the house (n = 160) or using public transportation (n = 88), respectively. Statistical significance of trends shown by z scores. Licensed under CC BY.
Risk factors associated with SARS-CoV-2 infection among farmworkers in Monterey County, Californiaexternal icon. Mora et al. JAMA Network Open (September 15, 2021).
Key findings:
- ~40% of farmworkers had possible workplace COVID-19 exposure since the start of the pandemic.
- 13.5% reported possible workplace exposure during the 2 weeks before testing.
- Outdoor agricultural work and indigeneity were associated with current SARS-CoV-2 infection.
- Living with children ≤5 years or with unrelated roommates was associated with increased risks of past infection.
- Farmworkers whose employer provided them with workplace prevention information had lower risk of past infection (aRR 0.59, 95% CI 0.40-0.86).
Methods: Observational study of 1,107 current and former farmworkers (83% born in Mexico; 85% spoke Spanish at home) tested for current (n = 911) or past SARS-CoV-2 infection (n = 1,058) and interviewed about COVID-19 risks and working and living conditions, Monterey County, CA, July─November 2020. Limitations: Possible misclassification of past infection due to potential waning immunity.
Implications: Farmworkers may be at increased risk of exposure due to working and living conditions. Vaccination and workplace prevention strategies are important for protecting the health of this population.
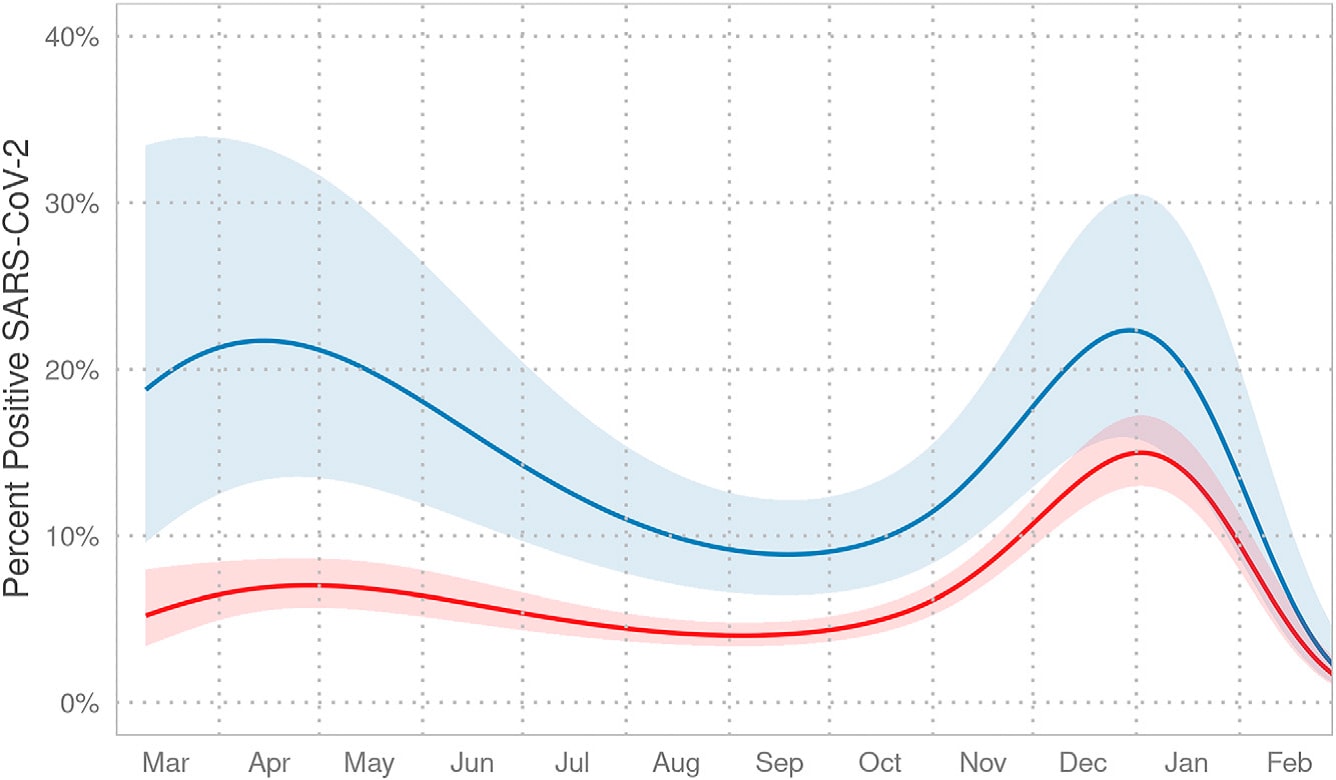
Association between preferred language and risk of Severe Acute Respiratory Syndrome Coronavirus 2 infection in children in the United States.external icon Otto et al. American Journal of Tropical Medicine and Hygiene (September 1, 2021).
Key findings:
- A higher proportion of children from Spanish-speaking families tested positive for SARS-CoV-2 infection than children from English-speaking families (22.5% vs. 12.3%).
- This difference was largest during the first wave and diminished over time (Figure).
Methods: Electronic health record data from 6 Children’s Hospital of Philadelphia Care Network primary care practices extracted for children aged <19 years (n = 10,138), March 1, 2020–February 28, 2021. Data included preferred language of caregiver, age, race/ethnicity, and SARS-CoV-2 PCR test result. Limitations: Data derived from single health system in single city; preferred language may be a surrogate for other unmeasured factors.
Implications: Children from non-English speaking families may be at greater risk for SARS-CoV-2 infections. Equitable access to healthcare and public health information may be improved with culturally and linguistically appropriate interventions.
Figure:
Note: Adapted from Otto et al. Probability of a SARS-CoV-2 positive test (y-axis) when adjusting for age and race/ethnicity over time (x-axis), March 2020–February 2021. The 2 lines with shaded 95% confidence intervals represent children in families preferring English or languages other than English. Licensed under CC BY 4.0.
Hispanic English language learner families and food insecurity during COVID-19: risk factors and systems of food supportexternal icon. Partika et al. Families, Systems, & Health (September 16, 2021).
Key findings:
- Food insecurity among English language learner (ELL) families increased from 41% before the COVID-19 pandemic to 66% during the pandemic.
- Food insecurity was relatively unchanged (31% to 35%) among non-ELL families.
- Among food insecure families during the pandemic, ELL families were less likely to use SNAP than non-ELL families (15% vs. 36%; p <0.001) but were more likely to use WIC (28% vs. 14%; p <0.05) and had similar use of school meals (59% vs. 62%).
Methods: Longitudinal study of 468 low-income Hispanic/Latino families whose children were in 1st grade in March 2020, Tulsa, Oklahoma. Food insecurity, based on a question from the USDA Household Food Security Scale, was examined at 3 timepoints (March 2019–June 2020) and compared by English language status of children. Use of WIC (Special Supplemental Nutrition Program for Women, Infants, and Children), SNAP (Supplemental Nutrition Assistance Program), and school meals were examined May–June 2020 only. Limitations: No information on immigration status; findings may not be generalizable.
Implications: Food insecurity increased during the pandemic among families not proficient in English. Sufficient access to food may require improved uptake of resources, such as SNAP, and stable access to school meals.
PREPRINTS (NOT PEER-REVIEWED)
COVID-19 pandemic response in a migrant farmworker community: Excess mortality, testing access and contact tracing in Immokalee, Floridaexternal icon. Limaye et al. medRxiv (October 1, 2021).
Key findings:
- 167 excess deaths (108% excess mortality) occurred during April–August 2020 compared to prior years.
- Among participants, a lower percentage of people who spoke Haitian Creole were tested for SARS-CoV-2 infection (42.4%) than those who spoke English (61.9%) or Spanish (51.9%).
- During contact tracing, only 39% of Spanish speakers were asked about their ability to safely quarantine compared with 70% of English speakers (p = 0.041).
- 28% of Spanish speakers reporting receiving information on local quarantine resources compared with 45% of English speakers.
Methods: Calculation of excess deaths (April─August 2020) and household survey (n = 318) conducted in Florida county with high SARS-CoV-2 infection rate and a high percentage of migrant and seasonal farmworkers who spoke Spanish or Haitian Creole, March–November 2020. Limitations: Survey response rate was <50%; may not be generalizable to other farmworker populations.
Implications: Inequities in provision of healthcare and contact tracing for migrant farmworkers were observed during the early months of the pandemic. Healthcare access and prevention efforts for this population should address structural factors and barriers posed by language to protect health and advance health equity.
Household density and influences from family and friends shape vaccine intent among Latino families in the San Francisco Bay area.external icon Wojcicki et al. medRxiv (September 27, 2021).
Key findings:
- Intention to receive or having already received COVID-19 vaccination was reported by 80% (TAB study cohort) and 84.3% (HEN/LEAD combined cohorts) of mothers from Hispanic/Latino study cohorts.
- Only 66.7% and 74.1%, respectively, reported intent to vaccinate their children.
- Participants who planned to receive COVID-19 vaccines more often cited family and friends as influencing their vaccine decisions than those who did not plan to receive vaccines.
Methods: Telephone survey to assess COVID-19 vaccination intent among 318 Hispanic/Latino families from 3 existing family-based cohorts in San Francisco, February–June 2021. Limitations: Study may not be generalizable to other Hispanic/Latino persons in the United States; may not have measured other factors that may impact vaccine availability and acceptance.
Implications: Social networks, including family and friends, may be important resources in efforts to increase vaccine uptake, especially among women and adolescents
PEER-REVIEWED
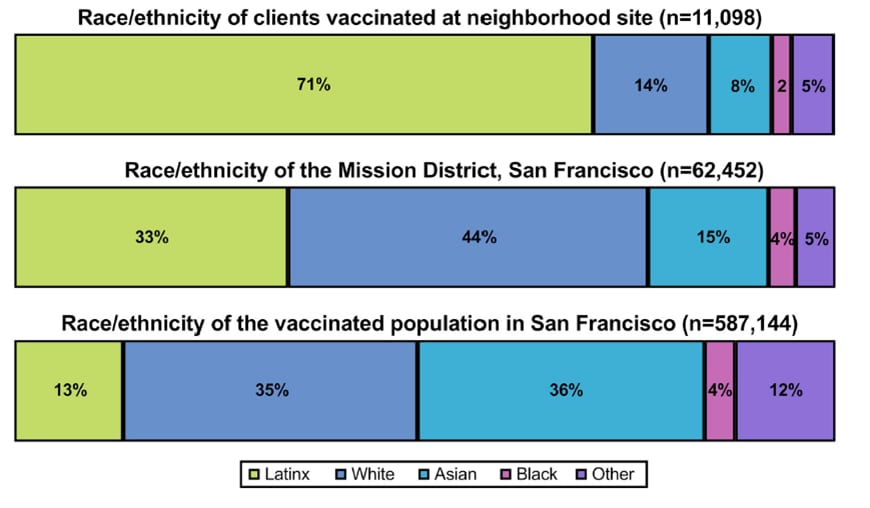
A multi-component, community-based strategy to facilitate COVID-19 vaccine uptake among Latinx populations: From theory to practiceexternal icon. Marquez et al. PLoS One (September 20, 2021).
Key findings:
- A culturally tailored program of “Motivate, Vaccinate, and Activate” successfully increased vaccine uptake among a Hispanic/Latino population in San Francisco’s Mission District.
- 70.5% of persons vaccinated at the program’s outdoor neighborhood site (n = 11,098) were Hispanic/Latino; by comparison, 33% of the Mission District and 13% of the vaccinated San Francisco populations were Hispanic/Latino (Figure).
- Among Hispanic/Latino vaccine recipients participating in the evaluation, 60.2% were first-generation immigrants, 76.1% had a household income <$50,000/year, 62.4% did not have a primary care provider, and 47.3% lacked health insurance.
- 91% of fully vaccinated persons reported personally reaching out to ≥1 unvaccinated persons to recommend vaccination at the site.
Methods: Community-academic partnership (Unidos en Salud) in San Francisco designed a multifaceted COVID-19 neighborhood vaccination strategy (February 1–May 19, 2021) that addressed barriers to vaccination in the Hispanic/Latino community. Program was evaluated using program data, citywide COVID-19 surveillance data, and a survey of vaccinated clients ≥16 years (n = 997). Limitations: Evaluation could not quantify number of people who were influenced by program but received vaccinations elsewhere.
Implications: Community-based programs can successfully address barriers to vaccination by using trusted messengers and social networks, adaptable mobilization strategies, and a convenient, culturally appropriate neighborhood site.
Figure:
Note: Adapted from Marquez et al. Race/ethnicity of clients vaccinated at the neighborhood site, of the Mission District, and of the vaccinated population in San Francisco. Licensed under CC BY 4.0.
Vaccines
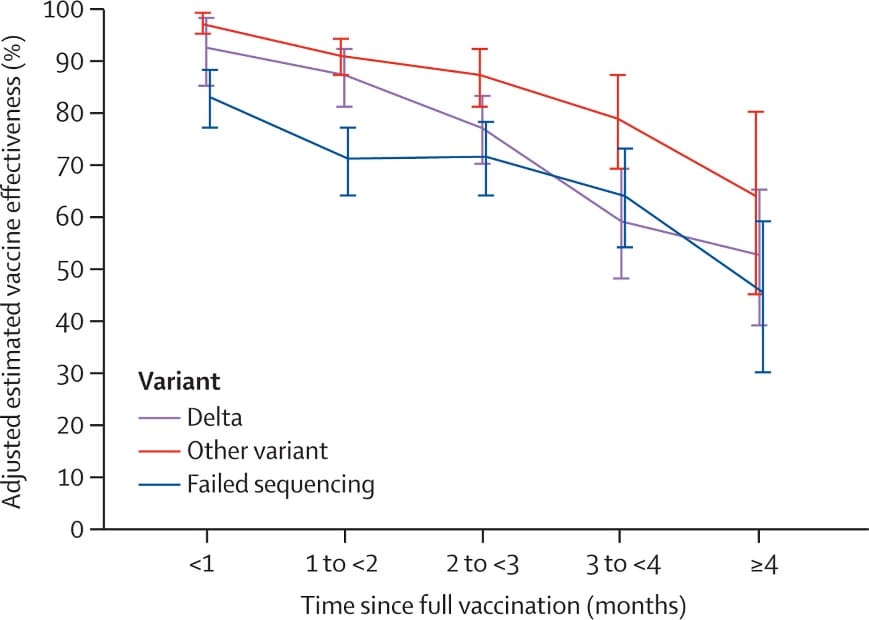
- Effectiveness of mRNA BNT162b2 COVID-19 vaccine up to 6 months in a large integrated health system in the USA: A retrospective cohort study.external icon Tartof et al. The Lancet (October 4, 2021). Using electronic health records from a large managed-care system in the United States (n = 3,436,957), December 2020–August 2021, vaccine effectiveness (VE) against SARS-CoV-2 infection among fully vaccinated persons (2 doses BNT162b2) aged ≥ 12 years was 75% (95% CI 71%-78%) for Delta compared with 91% (95% CI 88%-92%) for other variants. At ≥4 months post vaccination, VE declined to 53% (95% CI 39%-65%) for Delta compared with 67% (95% CI 45%-80%) for other variants.
Note: Adapted from Tartof et al. Adjusted estimated vaccine effectiveness against SARS-CoV-2 infection by viral variant. During the study, 28.4% of 5,008 SARS-CoV-2-positive specimens with genomic results were Delta variant. Data are shown for number of months since being fully vaccinated with BNT162b2 with 95% CIs. Reprinted from The Lancet, October 4, 2021, Tartof et al., Effectiveness of mRNA BNT162b2 COVID-19 vaccine up to 6 months in a large integrated health system in the USA: A retrospective cohort study, online ahead of print. Copyright 2021, with permission from Elsevier.
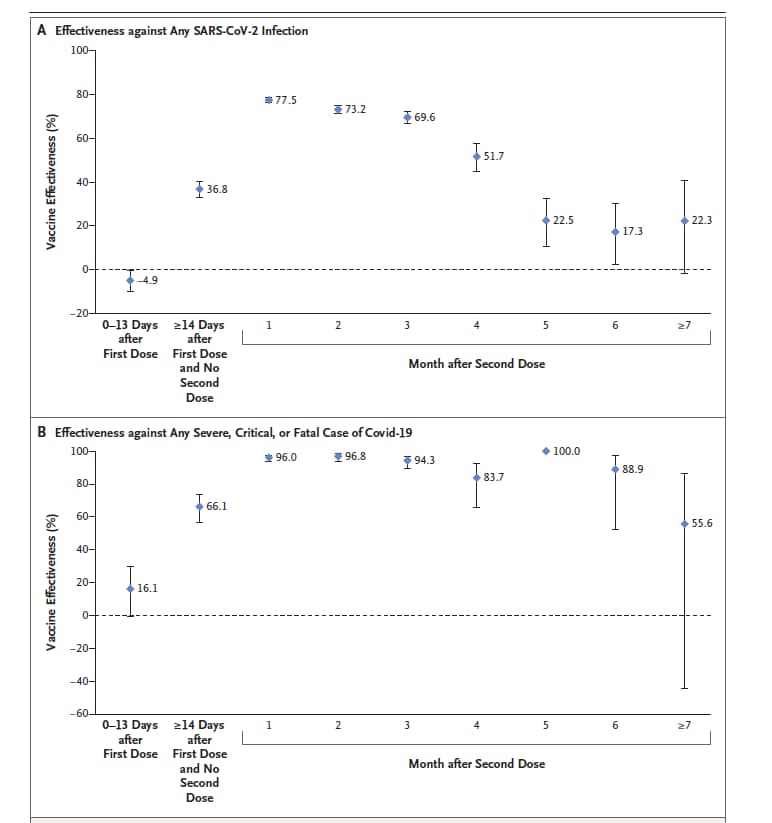
- Waning of BNT162b2 vaccine protection against SARS-CoV-2 infection in Qatarexternal icon. Chemaitelly et al. NEJM (October 6, 2021). Among fully vaccinated people (BNT162b2) in Qatar (n = 907,763), December 2020–September 2021, vaccine efficacy (VE) against SARS-CoV-2 infection declined to ~20% by 5–7 months after dose 2. However, VE against severe, critical, or fatal COVID-19 remained high for 6 months.
Note: Adapted from Chemaitelly et al. BNT162b2 vaccine effectiveness estimates against A) SARS-CoV-2 infection or B) severe COVID-19. Bars are 95% confidence intervals. From the New England Journal of Medicine, Chemaitelly et al., Waning of BNT162b2 vaccine protection against SARS-CoV-2 infection in Qatar. October 6, 2021, online ahead of print. Copyright © 2021 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
- Acute myocarditis following COVID-19 mRNA vaccination in adults aged 18 years or olderexternal icon. Simone et al. JAMA Internal Medicine (October 4, 2021). A study of acute myocarditis among adults (>18 years; n ~2.4 million) after mRNA vaccination (December 2020–July 2021) found 15 confirmed cases in the vaccinated group (n = 2 after dose 1; n = 13 after dose 2), for an observed incidence of 0.8 cases per 1 million and 5.8 cases per 1 million (doses 1 and 2, respectively). All cases were among men, with a median age of 25 years.
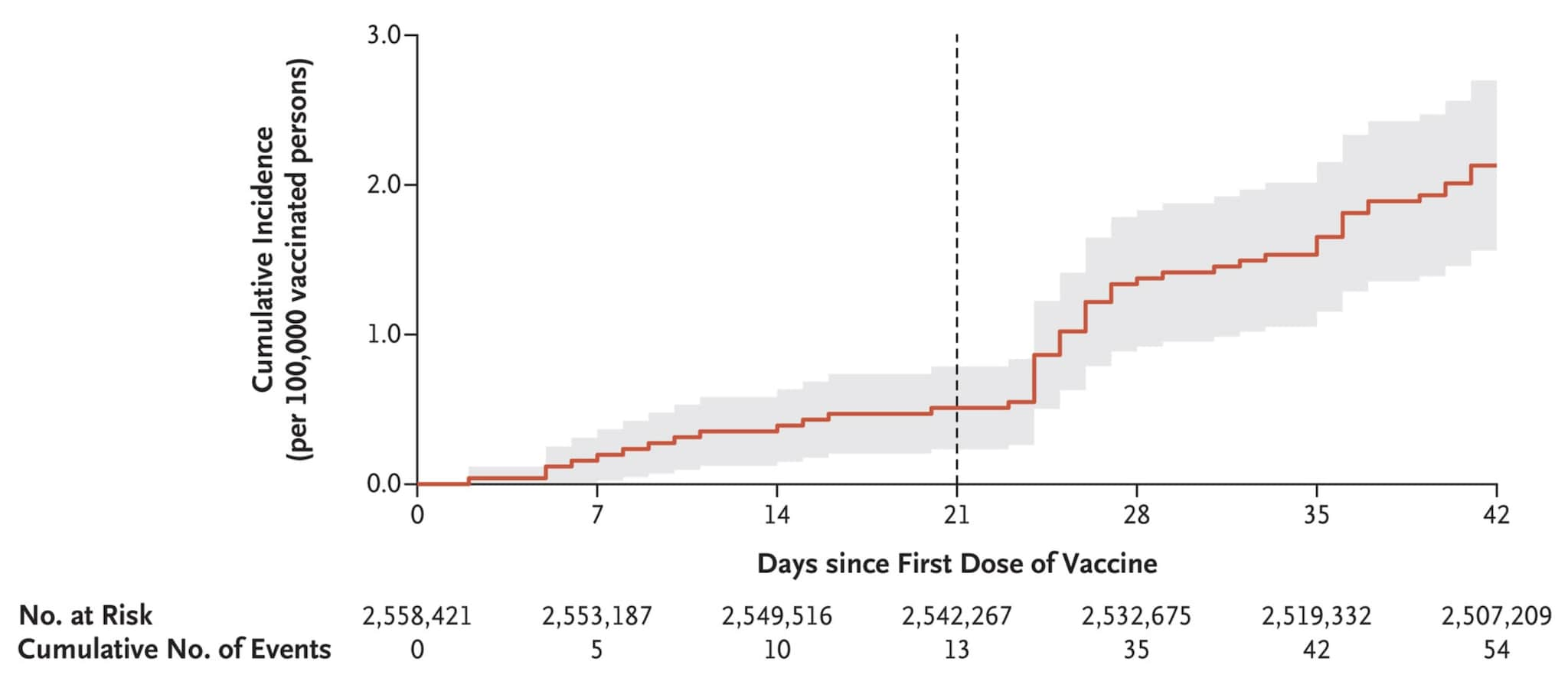
- Myocarditis after COVID-19 vaccination in a large health care organizationexternal icon. Witberg et al. NEJM (October 6, 2021). Among 2.5 million persons ≥16 years of age in the Israeli healthcare system who had received at least 1 dose of the BNT162b2 mRNA vaccine, the estimated myocarditis incidence was 2.13 cases per 100,000 persons; the highest incidence was among males aged 16–29 years. Most of the 54 cases were mild or moderate.
Note: Adapted from Witberg et al. Cumulative incidence of myocarditis during a 42-day period after receipt of 1st dose of BNT162b2. The vertical line at day 21 shows median day of 2nd vaccine dose. Shaded area is 95% CI. From the New England Journal of Medicine, Witberg et al., Myocarditis after COVID-19 vaccination in a large health care organization. October 6, 2021, online ahead of print. Copyright © 2021 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
- Association of receipt of the Ad26.COV2.S COVID-19 vaccine with presumptive Guillain-Barré Syndrome, February-July 2021external icon. Woo et al. JAMA (October 7, 2021). As of July 24, 2021, the Vaccine Adverse Event Reporting System identified 130 reports of presumptive Guillain-Barré syndrome (GBS) after receipt of Ad26.COV2.S vaccine (median age = 56 years; 59.7% men), including 122 hospitalized patients (93.8%) and 1 death. Median time to GBS onset was 13 days after vaccination; 95.3% had onset within 42 days. Among those with onset ≤42 days, the estimated observed-to-expected rate ratio was 4.18 (95% CI 3.47-4.98), corresponding to an absolute rate increase of 6.36 cases per 100,000 person-years, assuming a background rate of 2 cases per 100,000 person-years.
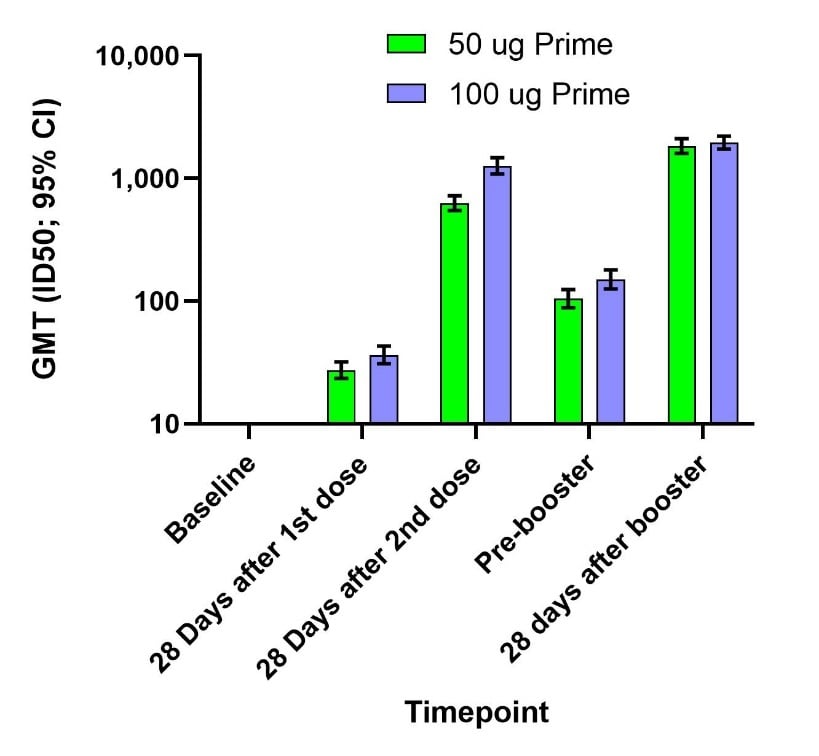
- Immune memory response after a booster injection of mRNA-1273 for Severe Acute Respiratory Syndromeexternal icon Coronavirus-2 (SARS-CoV-2). Chu et al. medRxiv (Preprint; October 1, 2021). Published in Nature Medicine as Immune response to SARS-CoV-2 after a booster of mRNA-1273: an open-label phase 2 trialexternal icon (March 3, 2022). Among 344 trial participants fully immunized (mRNA-1273 vaccine) who received a 50 µg mRNA-1273 booster, booster-related side effects were similar to those after dose 2, and included arm pain, fatigue, headache, muscle/joint pain, and chills. Compared to dose 2 of the primary series, the booster raised neutralizing antibody levels, with a 2.1-fold increase against the Delta variant 4 weeks after the booster.
Note: Adapted from Chu et al. Geometric mean titers (GMTs) and 95% CIs against wild-type virus (not the Delta variant) for serum collected at baseline, after the primary series (50 µg or 100 µg), just prior to the booster dose, and 28 days after the booster dose among 344 participants who received a 50-µg booster 6–8 months after the primary series. Used by permission of authors.
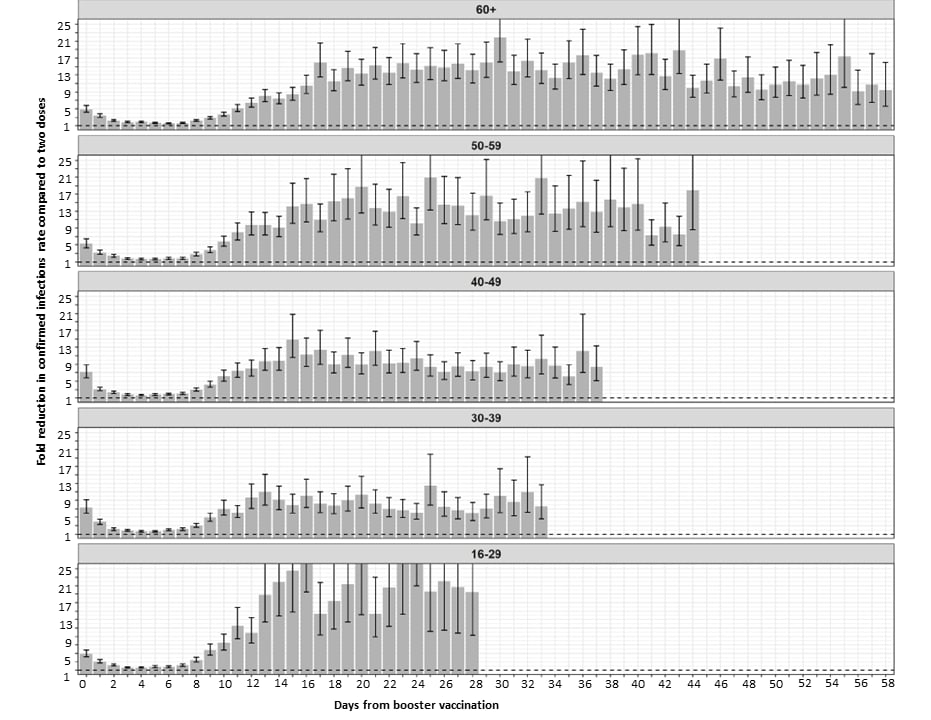
- Protection across age groups of BNT162b2 vaccine booster against COVID-19external icon. Bar-On et al. medRxiv (Preprint; October 7, 2021). Published in NEJM as Protection against COVID-19 by BNT162b2 booster across age groupsexternal icon (December 23, 2022). As of October 4, 2021, across 4.6 million fully vaccinated individuals in Israel (≥16 years), a 3rd BNT162b2 dose ≥12 days earlier reduced SARS-CoV-2 infection rates 10-fold (range: 8.8–17.6 across age groups), compared with no booster. Severe illness rates decreased 18.7-fold (95% CI 15.7-22.4) among persons ≥60 years and 22.0-fold (95% CI 10.3-47.0) for persons aged 40–60 years. For persons ≥60 years, COVID-19 associated mortality was 14.7-fold (95% CI 9.4-23.1) lower for the booster group.
Note: Adapted from Bar-On et al. The fold reduction in the rate of confirmed infections in the booster group compared to the non-booster group as a function of the number of days following the booster dose (day 0 = day receiving the booster dose), for the different age groups. The dashed horizontal line represents no added protection by the booster dose. Error bars are 95% confidence intervals not corrected for multiplicity. Permission request in process.
Natural History, Reinfection, and Health Impact
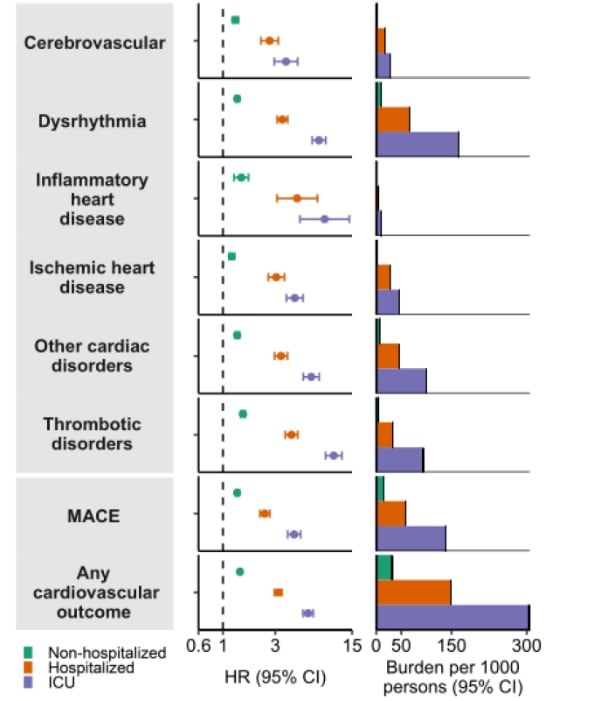
- One-year risks and burdens of incident cardiovascular disease in COVID-19: Cardiovascular manifestations of Long COVIDexternal icon. Al-Aly et al. Research Square (Preprint; October 5, 2021). United States veterans with SARS-CoV-2 infection (n = 151,195) had an increased risk of incident cardiovascular outcomes ≥30 days post-infection, compared with 3,670,087 contemporary controls, including cerebrovascular disorders (hazard ratio [HR] 1.48, 95% CI 1.40-1.57), dysrhythmias (HR 1.66, CI 1.60-1.72), and major adverse cardiovascular events (HR 1.55, CI 1.50-1.60). Risk increased for veterans hospitalized or admitted to the ICU for COVID-19.
Note: Adapted from Al-Aly et al. Adjusted hazard ratios (HR) and 1-year burdens of post-acute COVID-19 cardiovascular outcomes compared to contemporary controls, by acute infection care setting (non-hospitalized, hospitalized, intensive care unit [ICU]). Outcomes ascertained 30–286 days after initial COVID-19 diagnosis. MACE = major adverse cardiovascular event: a composite of myocardial infarction, stroke, and all-cause mortality. Licensed under CC BY 4.0.
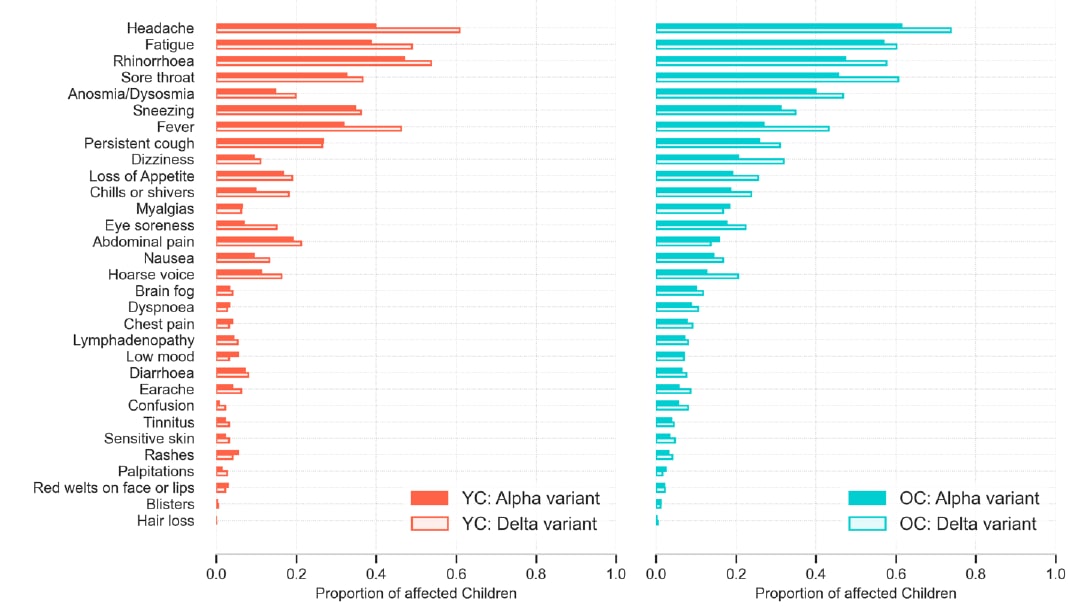
- Illness characteristics of COVID-19 in children infected with the SARS-CoV-2 Delta variantexternal icon. Molteni et al. medRxiv (Preprint; October 7, 2021). Published in Childrenexternal icon (May 3, 2022). In a United Kingdom COVID-19 symptom study that compared 694 symptomatic children (aged 5─17 years) enrolled December 2020─May 2021 (Alpha [B.1.1.7] variant period) with 706 children enrolled May─July 2021 (Delta [B.1.617.2] variant period), the median number of symptoms was slightly higher during the Delta than the Alpha period (in younger children: 4 vs. 3; in older children: 6 vs. 5). The odds of headache, rhinorrhea, sore throat, dysosmia, fever, dizziness, chills or shivers, eye soreness, and hoarse voice were higher during the Delta period. The top 7 symptoms for each age group were common to both periods, and the median illness duration (5 days) was similar.
Note: Adapted from Molteni et al. Symptoms reported over the course of illness (up to 28 days) in younger (aged 5–11 years) and older (aged 12–17 years) children with symptomatic COVID-19 during Alpha (solid bar) or Delta (hollow bar) variant periods. YC: younger children; OC: older children. Permission request in progress.
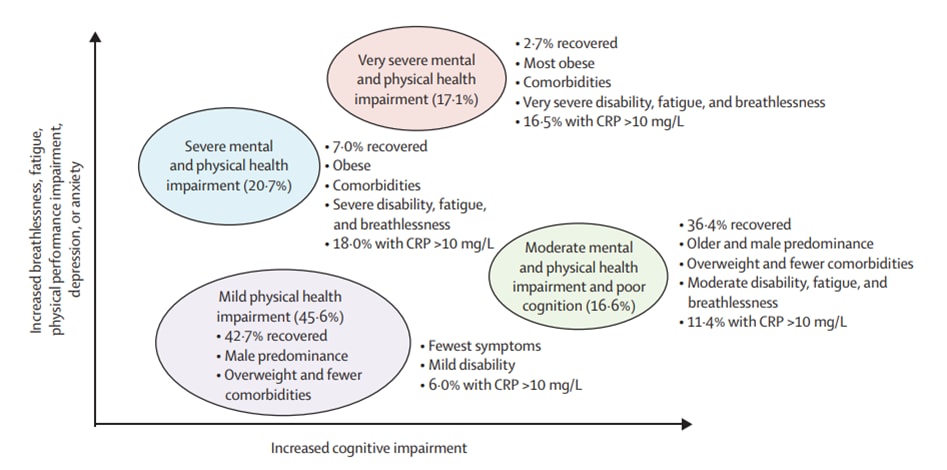
- Physical, cognitive, and mental health impacts of COVID-19 after hospitalisation (PHOSP-COVID): A UK multicentre, prospective cohort study.external icon Evans et al. The Lancet Respiratory Medicine (October 7, 2021). Among 1,077 COVID-19 patients discharged from United Kingdom hospitals (March–November 2020), 29% felt fully recovered on follow-up assessments 2–7 months after discharge. Among 767 patients with available data on physical, mental, and cognitive outcomes, 4 recovery phenotype clusters were identified: very severe mental and physical impairment (MPI), severe MPI, moderate MPI with poor cognition, and mild physical impairment.
Note: Adapted from Evans et al. Four recovery phenotype clusters: very severe MPI (131; 17.1%); severe MPI (159; 20.7%); moderate MPI with poor cognition (127; 16.6%); mild physical health impairment (350; 45.6%) with associated demographics, symptoms, and physical function. CRP = C-reactive protein. Licensed under CC BY.
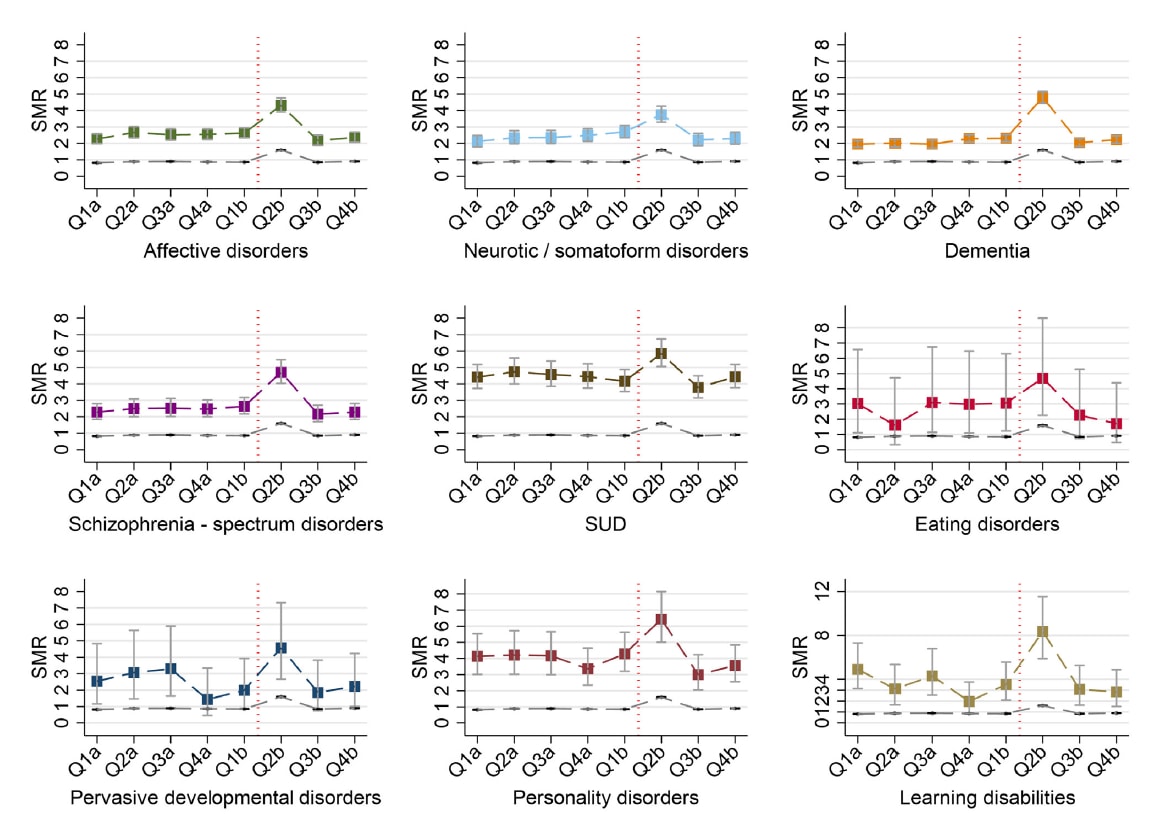
- All-cause and cause-specific mortality in people with mental disorders and intellectual disabilities, before and during the COVID-19 pandemic: Cohort study.external icon Das-Munshi et al. The Lancet Regional Health – Europe. (October 7, 2021). An analysis of prospective data (n = 167,122) from a large mental healthcare provider in the United Kingdom showed that by the 2nd quarter of 2020 (after the pandemic began), age- and gender-standardized mortality ratios (SMR) among persons with 9 psychiatric diagnoses were 2–9 times higher than the general population. SMRs among persons with learning disabilities (9.24, 95% CI 5.98-13.64) were particularly high.
Note: From Das-Munshi et al. Age- and gender-SMRs by psychiatric diagnosis (affective disorders, neurotic/somatoform disorders, dementia, schizophrenia-spectrum disorders, substance use disorders (SUD), eating disorders, pervasive developmental disorders, personality disorders, learning disabilities) with deaths from 2019 to end of 2020. Q: Quarter; a: 2019; b: 2020. Vertical red line indicates January 30, 2020, when the WHO declared COVID-19 a public health emergency of international concern. Square markers indicate SMRs for psychiatric diagnoses and dashed gray line indicates age and gender-SMRs for general population (London). Licensed under CC BY.
From the Morbidity and Mortality Weekly Report (October 15, 2021).
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.