COVID-19 Science Update released: August 28, 2020 Edition 43

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Here you can find all previous COVID-19 Science Updates.
Anti-SARS-CoV-2 neutralizing antibodies (NAbs) can be found in persons who have recovered from COVID-19. Characterizing NAb activity might provide relevant data for understanding NAbs levels needed for natural protection against reinfection. It could also help determine the optimal design and dosing of vaccines.
PEER-REVIEWED
Evaluating the association of clinical characteristics with neutralizing antibody levels in patients who have recovered from mild COVID-19 in Shanghai, Chinaexternal icon. Wu et al. JAMA Internal Medicine (August 18, 2020).
Key findings:
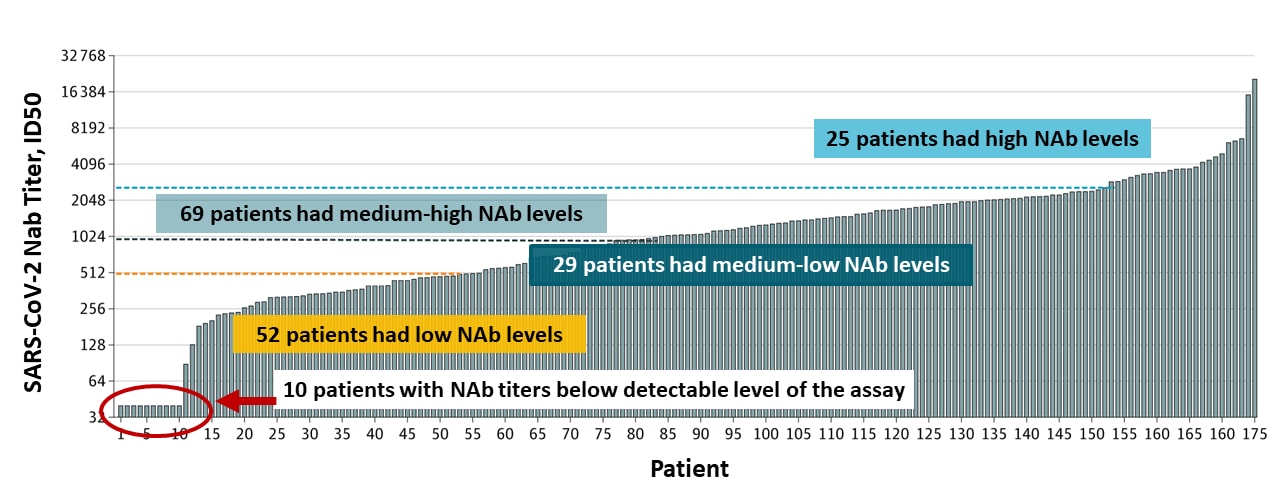
- SARS-CoV-2-specific neutralizing antibody (Nab) titers varied substantially, including less than the detectable level of the assay (Figure 1).
- NAbs were detected from day 4 to day 6 after symptom onset and peaked at day 10 to day 15.
- NAb titers were significantly correlated with levels of spike-binding antibody and plasma C-reactive protein.
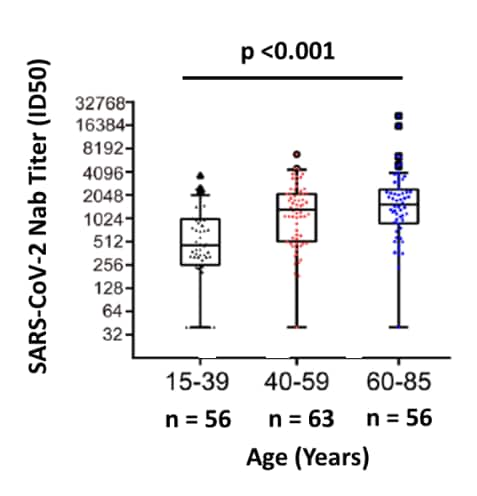
- NAb titers were significantly higher in men compared with women (p = 0.01) and in middle-aged (40-59 years) and older adults (60-85 years) compared with persons 15-39 years, p <0.001 (Figure 2).
Methods: Cohort study of 175 patients with laboratory-confirmed mild COVID-19 hospitalized from January 24 to February 26, 2020 at a single hospital in Shanghai, China. Plasma was tested for SARS-CoV-2–specific NAbs titers and virus spike-binding antibodies every 2 to 4 days from admission until discharge and then two weeks after discharge. Limitations: Single setting; results might not be generalizable.
Figure 1
Note: Adapted from Wu et al. Levels of SARS-CoV-2-specific NAbs in patients. SARS-CoV-2–specific NAbs titer (50% inhibitory dose [ID50]) for each patient at the time of discharge is shown as an individual histogram. Dashed lines show the upper bounds of NAb levels: low (ID50 <500); medium-low (ID50 500-999), and medium-high (ID50 1000-2500). Licensed under CC-BY.
Figure 2
Note: Adapted from Wu et al. NAbs titers in patients 15-39 years, 40-59 years, and 60-85 years. Licensed under CC-BY.
Neutralizing antibodies correlate with protection from SARS-1 CoV-2 in humans during a fishery vessel outbreak with high attack rateexternal icon. Addetia et al. medRxiv (August 13, 2020). Published external iconin Journal of Clinical Microbiology (August 21, 2020).
Key findings:
- Prior to a voyage on a fishing vessel, 120/122 crew members had negative RT-PCR tests for SARS-CoV-2.
- Six of 120 were seropositive with antibodies against the viral nucleoprotein (Figure).
- Illness in a crew member prompted vessel return and testing where 104/122 (85.2%) crew members tested positive by RT-PCR or serology.
- Three crew members who were seropositive with neutralizing and spike-reactive antibodies prior to the voyage did not become infected (p = 0.002).
Methods: Study of 122 crew members involved in a SARS-CoV-2 outbreak aboard a fishing vessel. Pre- and post-departure serological and RT-PCR testing performed in 120 of the 122 crew members over a median follow-up of 32.5 days and specimens positive on serologic testing were assessed for SARS-CoV-2 neutralizing and spike-binding activity. Limitations: Lack of clinical and contact information, small number of crew members with NAbs in order to better assess protection from infection.
Figure:
Note: Adapted from Addetia et al. Pre-departure RT-PCR and serology and subsequent infection in crew members on the vessel. Crew members who were seropositive pre-departure were further characterized by neutralizing activity in pre-departure specimen. There was one additional infected crew member who did not have testing prior to the voyage so the total number of infected crew members was 104. Licensed under CC-BY.
Implications of both studies (Wu et al. & Addetia et al.): Both studies showed that NAbs naturally develop from SARS-CoV-2 infection with variable levels and might confer protection from later infection. Implications of variable levels of SARS-CoV-2–specific NAbs on protection against SARS-CoV-2 re-infections deserve further exploration.
PEER-REVIEWED
Characteristics and outcomes of COVID-19 in patients with HIV: A multi-center research network study. external iconHadi et al. AIDS (August 10, 2020).
Key findings:
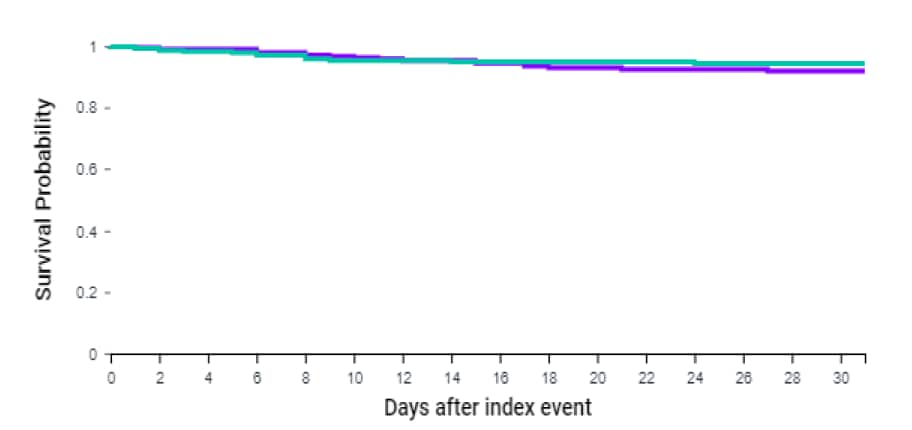
- In unmatched analysis, persons with HIV infection had higher mortality at 30 days (Risk Ratio [RR] 1.55, 95% CI 1.01 – 2.39) compared to those without HIV; however, after propensity score matching on race, sex, smoking, and chronic diseases, no difference in mortality was seen (RR 1.33, 95% CI 0.69 – 2.57) (Figure).
Methods: Retrospective cohort of 50,167 patients with COVID (49,763 without and 404 with HIV infection). The cohort with HIV was compared with a 1:1 propensity-matched cohort of patients without HIV to account for confounding. Limitations: Retrospective design; biases associated with analyses of data from electronic records.
Implications: After controlling for comorbidities that increase risk for severe COVID-19 in persons with HIV infection, survival between those with and without HIV appears similar.
Figure:
Note: From Hadi et al. Survival probability in persons with SARS-CoV-2 without HIV and with HIV in matched analysis. Used with permission from Wolters Kluwer Health, Inc.: Hadi et al. Characteristics and outcomes of COVID-19 in patients with HIV: A multi-center research network study. AIDS. doi: 10.1097/QAD.0000000000002666.
Association of race with mortality among patients hospitalized with coronavirus disease 2019 (COVID-19) at 92 US hospitals. external iconYehia et al. JAMA Network Open (August 18, 2020).
Key findings:
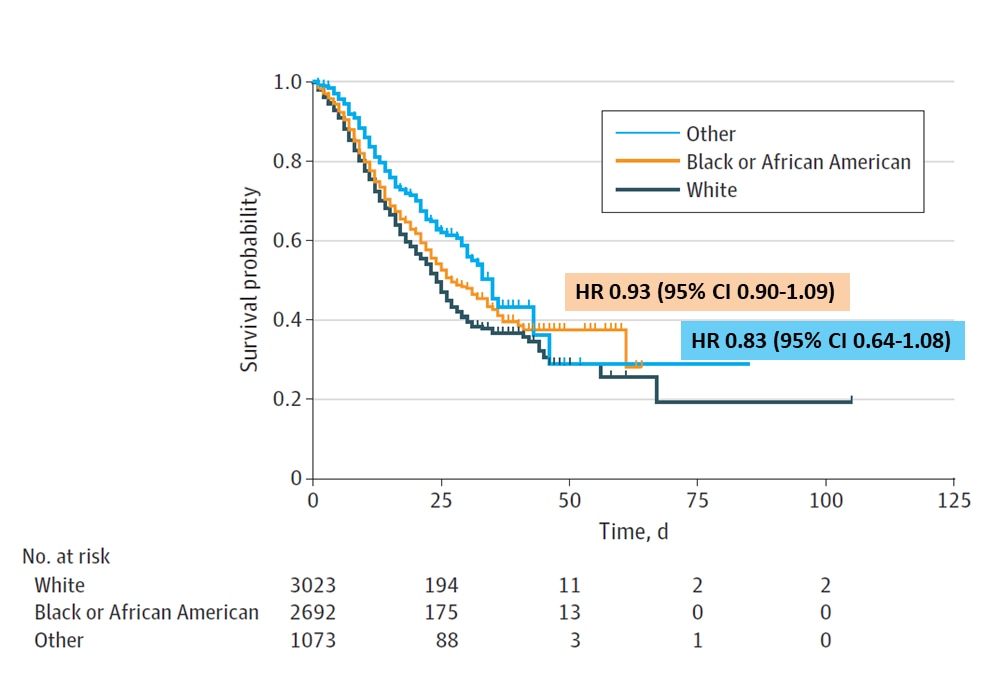
- After adjusting for sociodemographic and clinical factors, there was no association between mortality risk and Black race (hazard ratio = 0.93, 95% CI 0.90 – 1.09).
- In-hospital mortality among White and Black hospital patients was similar (23.1% vs 19.2%, respectively).
- Among persons requiring ICU care, 36.4% of White patients and 35.2% of Black patients died.
- In the final model, older age, male sex, persons with Medicare or unknown insurance, and persons with chronic kidney or heart disease had increased risk.
Methods: Cohort of 11,210 adults with confirmed SARS-CoV-2 from 92 hospitals in 12 states from February 19 to May 31, 2020. Cox proportional hazards regression was used to evaluate associations between patient characteristics and time to all-cause, in-hospital mortality. Limitations: Results apply to persons able to access hospital care; mortality might vary when accounting for death before and after hospitalization; neighborhood deprivation measured at zip code level.
Implications: Studies examining race and risk of death before and after hospitalization for SARS-CoV-2 are warranted. As suggested in the accompanying commentary by Boulwareexternal icon, greater neighborhood deprivation and comorbidities in Black Americans should be addressed.
Figure:
Note: From Yehia et al. Survival by race among hospitalized adults with COVID-19. Hazard ratios calculated relative to the referent White race. Licensed under CC-BY.
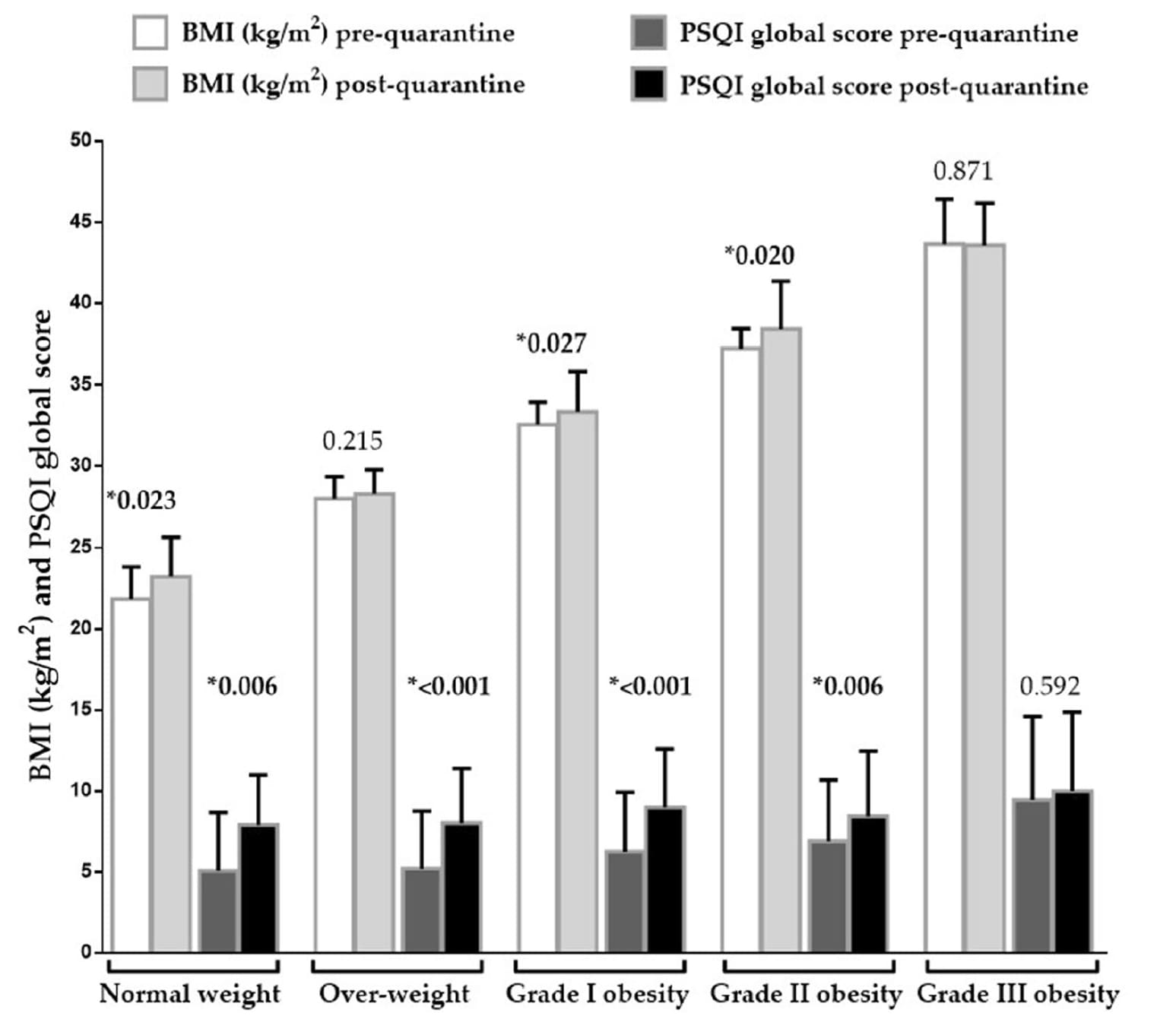
Does SARS-CoV-2 threaten our dreams? Effect of quarantine on sleep quality and body mass index.external icon Barrea et al. Journal of Translational Medicine (August 18, 2020).
Key findings:
- Relative to pre-quarantine, post-40-day post-quarantine, body mass index (BMI) was significantly increased for normal weight persons (BMI 18.5-24.9 kg/m2, p = 0.023) and those in Grade I (30.0-34.9 kg/m2, p = 0.027) and II (35.0-39.9 kg/m2, p = 0.02) obese groups and both genders (p <0.001) (Figure).
- Daily physical activity decreased during quarantine (51.2% vs 32.2%, p = 0.004)
- Percentage of poor sleepers increased during quarantine (relative to pre-quarantine) in normal weight (72.7% vs 36.4%), overweight (80.6% vs 29%), and grade I obesity (85.4% vs 57.3%) groups (p <0.001). No differences were found in grade II and grade III obesity.
Methods: Retrospective study January to April 30, 2020 in 121 adults, Naples, Italy, measuring height and weight, sleep quality, and physical activity. Initial assessment was at an outpatient clinic; follow-up conducted by phone interview after 40 days of quarantine. Limitations: Small sample size; self-reported weight; dietary intake, which is related to weight gain and sleep disturbance, was not evaluated.
Implications: Quarantine was associated with decreased sleep and physical activity and increased BMI. Lifestyle strategies to counteract these effects should be considered.
Figure:
Note: From Barrea et al. BMI and sleep quality score pre- and post-quarantine in a population study of BMI categories. Poor sleep quality = Pittsburgh Sleep Quality Index (PSQI) score ≥5; good sleep quality = PSQI score <5. Licensed under CC-BY 4.0.
PEER-REVIEWED
Association between number of in-person health care visits and SARS-CoV-2 infection in obstetrical patients.external icon Reale et al. JAMA (August 14, 2020).
Key findings:
- Among 2,968 deliveries, 111 patients (3.7%, 95% CI 3.1% -4.5%) tested positive for SARS-CoV-2.
- No association was found between number of in-person health care visits (mean 2.5 vs 2.6 visits) and SARS-CoV-2 infection (aOR for infection was 0.93 [95% CI 0.80-1.08] per additional health care visit).
Methods: Surveillance for SARS-CoV-2 was conducted for 2,968 deliveries from 4 hospitals, Boston, Massachusetts, April 19 to June 27, 2020. Nested case-control study of obstetric patients who tested positive for SARS-CoV-2 and matched control patients without SARS Co-V-2 infection (93 and 372, respectively). Conditional logistic regression was used to assess association of the number of in-person visits with SARS-CoV-2 infection. Limitations: One US community; results might not be externally generalizable.
Implications: This study suggests necessary in-person health care visits can be safely conducted without increased risk of SARS-CoV-2 infection, offering some reassurance to patients, providers, and the community.
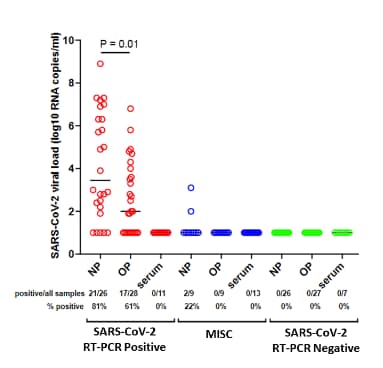
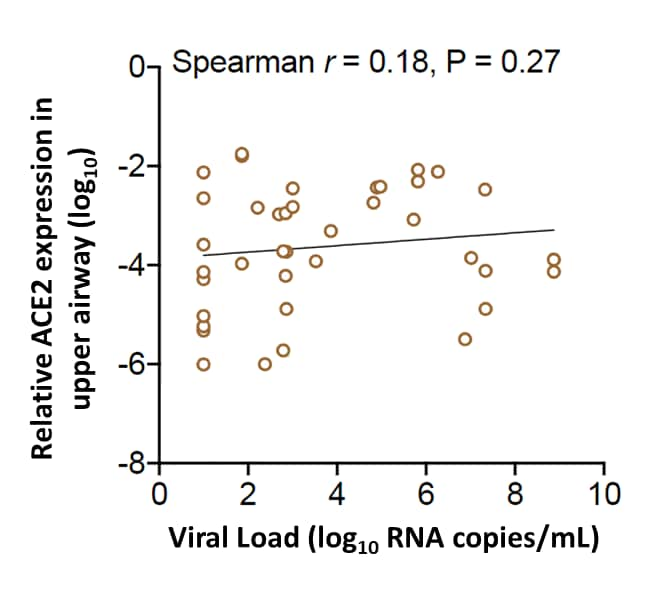
Pediatric SARS-CoV-2: Clinical presentation, infectivity, and immune responsesexternal icon. Yonker et al. Journal of Pediatrics (August 20, 2020).
Key findings:
- 25.5% tested positive for SARS-CoV-2; 9.4% were diagnosed with multisystem inflammatory syndrome in children (MIS-C).
- Viral load (VL) was higher in NP specimens than in OP specimens in children who tested positive for SARS-CoV-2, p = 0.01 (Figure 1) and did not correlate with age.
- Angiotensin Converting Enzyme 2 (ACE2) expression (the receptor for SARS-CoV-2 infection) was related to testing positive for SARS-CoV-2 or diagnosis with MIS-C, p = 0.004, but was not related to VL (Figure 2).
Methods: Sample of 192 children aged 0-22 years with suspected or confirmed SARS-CoV-2 or MIS-C, Boston, MA. Participants provided NP (n = 83), OP (n = 105), and/or blood specimens (n = 100). SARS-CoV-2 viral load, ACE2 gene expression, and SARS-CoV-2 serology were measured. Limitations: An enrolled cohort that may not be representative of overall pediatric SARS-CoV-2 infections. Low proportion of children provided each specimen type; low numbers of children <5 years in sample.
Implications: Children might have high levels of virus in upper airways despite showing few symptoms and might be effective transmitters regardless of age. Findings could help inform guidance for operating schools and daycare institutions.
Figure 1
Figure 2
Note: Adapted from Yonker et al. Figure 1. Viral load of NP swabs, OP swabs, and serum for children testing positive for SARS-CoV-2, with MIS-C, or testing negative for SARS-CoV-2. Figure 2. Correlation between SARS-CoV-2 viral load and ACE2 expression in upper airway. This article was published in Journal of Pediatrics, Yonker et al., Pediatric SARS-CoV-2: Clinical presentation, infectivity, and immune responses, Copyright Elsevier 2020. This article is currently available at the Elsevier COVID-19 resource center: https://www.elsevier.com/connect/coronavirus-information-centerexternal icon.
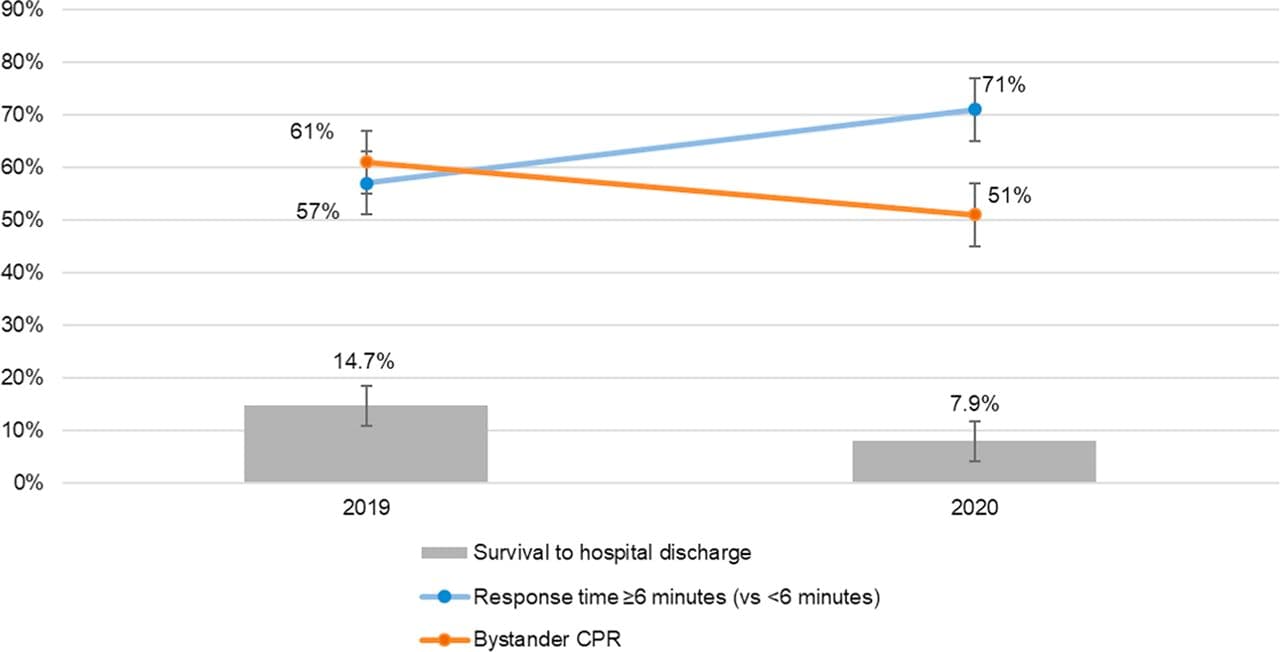
Out-of-hospital cardiac arrest response and outcomes during the COVID-19 pandemicexternal icon. Uy-Evanad et al. Journal of American College of Cardiology: Clinical Electrophysiology. (August 10, 2020)
Key Findings:
- In 2020 (Figure):
- Emergency Medical Services (EMS) response time increased, (6.6 vs 7.6 minutes, p <0.001).
- Bystander cardiopulmonary resuscitation decreased (61% vs 51%, p = 0.02).
- Use of automated external defibrillators decreased (5% to 1%, p = 0.02).
- Survival to hospital discharge for OHCA decreased (14.7% to 7.9%, p = 0.02).
Methods: Consecutive patients with out-of-hospital cardia arrests (OHCA) were enrolled to assess EMS and bystander response and outcomes from March 1 to May 31, 2019 compared with the same period in 2020 in two U.S. communities with relatively low SARS-CoV-2 infection rates. Limitations: Not a systematic sample; potential reasons for findings not given.
Implications: The COVID-19 pandemic might adversely affect survival to hospital discharge from OHCA, even in areas with relatively low COVID-19 incidence. Community and EMS response during the ongoing pandemic and future outbreaks should be optimized to improve survival after OHCA.
Figure:
Note: Adapted from Uy-Evanado et al. Comparison between 2019 and 2020 in EMS response time, the proportions of bystander CPR use and of survival to hospital discharge. This article was published in Journal of American College of Cardiology: Clinical Electrophysiology, Uy-Evanado et al., Out-of-hospital cardiac arrest response and outcomes during the COVID-19 pandemic, Copyright Elsevier on behalf of American College of Cardiology Foundation 2020. This article is currently available at the Elsevier COVID-19 resource center: https://www.elsevier.com/connect/coronavirus-information-centerexternal icon.
PEER-REVIEWED
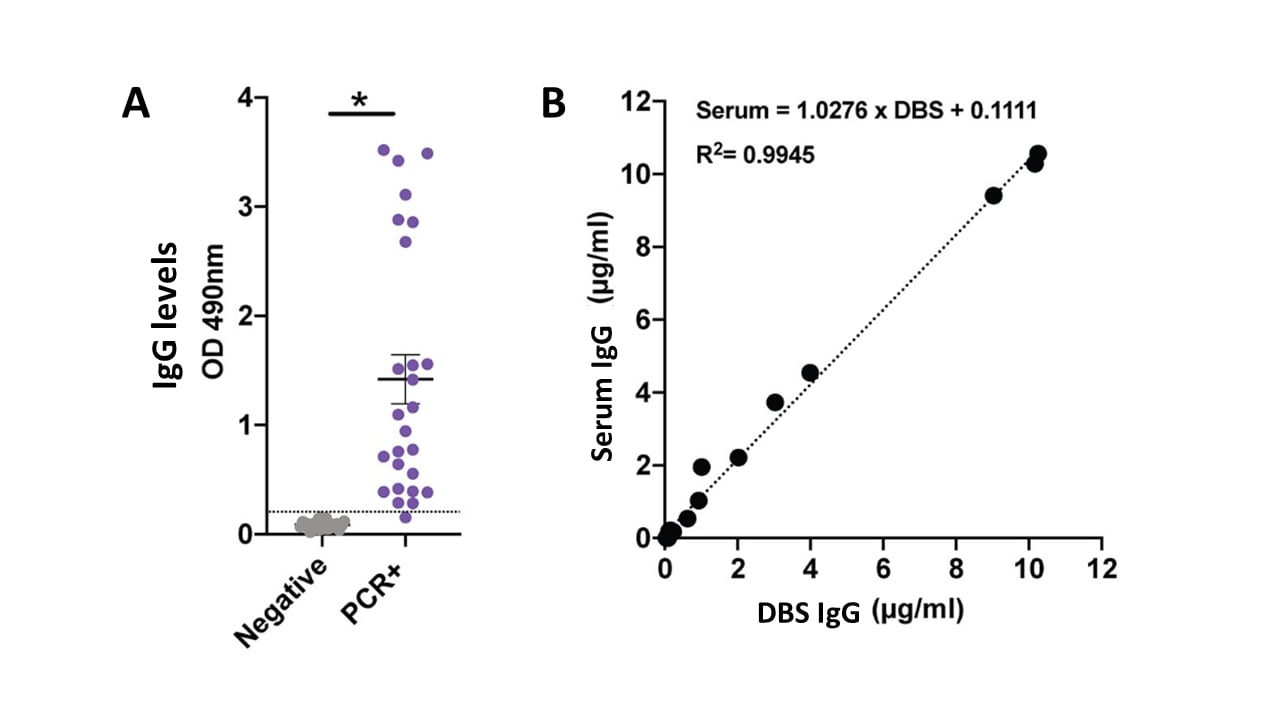
High seroprevalence for SARS-CoV-2 among household members of essential workers detected using a dried blood spot assayexternal icon. McDade et al. PLOS One (August 14, 2020).
Key findings:
- A self-collected Dried Blood Spot (DBS) IgG assay had high agreement (R2 = 0.99) with serum levels in RT-PCR-positive cases (p <0.001) (Figure).
Methods: Community sample of 232 adults from April 18 to May 20, 2020 who provided self-collected DBS finger stick samples and information on symptoms and SARS-CoV-2 diagnosis. FDA-approved immunoassay was adapted to measure IgG antibodies in DBS samples. Limitations: Unclear recruitment strategy; small numbers; potential selection bias; multiple sources of exposure of household members not assessed; assumptions of transmission and causality, however, most of these do not impact the analytic comparison between assays.
Implications: Self-collected DBS could be a useful tool to more easily assess community seroprevalence, increase COVID-19 serologic testing, and assess viral spread within populations.
Figure:
Note: Adapted from McDade et al. A: IgG levels in DBS measured by optical density (OD) among RT-PCR-positive cases and RT-PCR-negative cases. B: Agreement between DBS IgG and serum IgG levels for PCR-positive cases.
PREPRINTS (NOT PEER-REVIEWED)
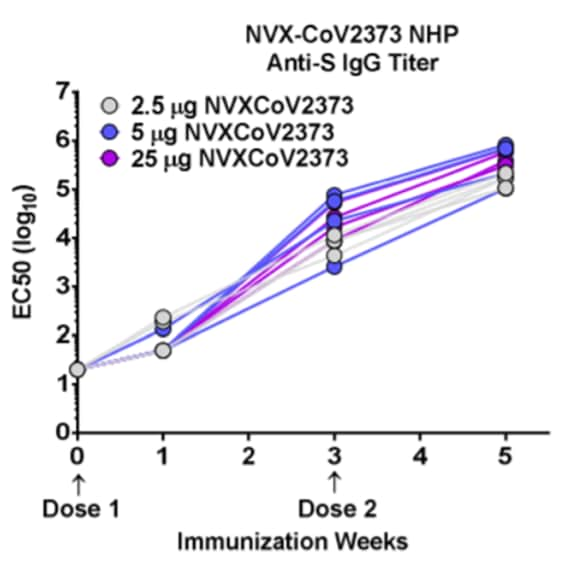
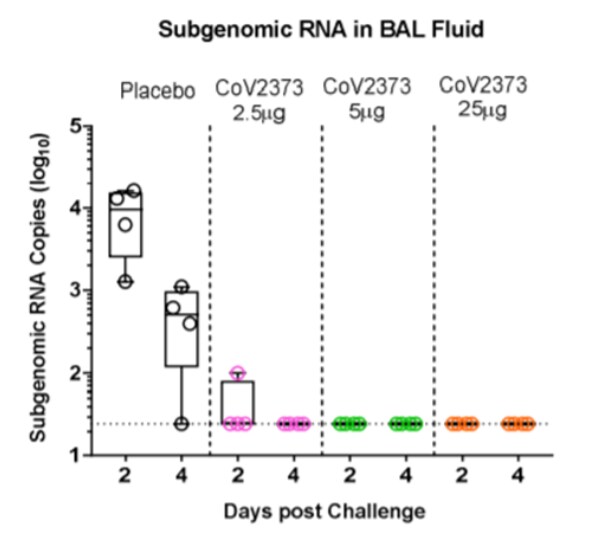
NVX-CoV2373 vaccine protects cynomolgus macaque upper and lower airways against SARS-CoV-2 challenge.external icon Guerbre-Xabier et al. bioRxiv (August 19, 2020). Publishedexternal icon in Vaccine (November 25, 2020)
Key findings:
- Macaques receiving vaccine showed elevated levels of anti-spike IgG titers (Figure 1).
- Immunized macaques were protected against upper and lower respiratory tract infection with SARS-CoV-2 (Figure 2).
Methods: Randomized, controlled trial of NVX-CoV2327 (a subunit of the SARS-CoV-2 spike glycoprotein) vaccine efficacy. 16 macaques (4 per group) received two injections 21 days apart of 2.5 μg vaccine with adjuvant, 5 or 25 μg vaccine with adjuvant, or placebo. Serum was collected on Days 0 and 21 (pre-immunization) and at Day 33. All animals received intranasal and intratracheal challenges with SARS-CoV-2 circa Day 35 and were tested 2 and 4 days later.
Implications: Protection from infection in this animal trial supports Phase 3 efficacy trials for humans.
Figure 1
Note: Adapted from Guebre-Xabier et al. Anti-spike IgG titers (EC50) at Days 0 (immediately pre-vaccination dose 1), 21 (immediately pre-injection dose 2) and 33. Each dot represents one macaque. Used by permission from the author.
Figure 2A
Figure 2B
Note: Adapted from Guebre-Xabier et al. 2A: Subgenomic RNA copies of replicating SARS-CoV-2 in BAL fluid after intratracheal challenge and 2B: Subgenomic RNA copies in nasal swab after intranasal challenge in macaques by immunization status. Each dot represents one macaque. BAL-bronchoalveolar lavage. Used by permission from the author.
Clinical Manifestations
- Hessami et al. COVID-19 and maternal, fetal and neonatal mortality: A systematic reviewexternal icon. Journal of Maternal-Fetal and Neonatal Medicine. Fetal and neonatal deaths were associated with severity of maternal infection and prematurity rather than vertical transmission of infection.
- Jiang et al. COVID-19 and multisystem inflammatory syndrome in children and adolescentsexternal icon. Lancet Infectious Diseases. Review lists various definitions of multisystem inflammatory syndrome in children (MIS-C), compares MIS-C with Kawasaki disease, and describes clinical management of MIS-C.
- Fox et al. Cardiac endotheliithis and multisystem inflammatory syndrome after COVID-19external icon. Annals of Internal Medicine. A case report of a young adult who died with findings of lymphadenopathy, pulmonary thrombi, and cardiac vasculitis and endotheliithis of small cardiac vessels but not coronary arteries on autopsy.
- Alwan N. A negative COVID-19 test does not mean recovery.external icon Nature. Many persons with COVID have had long-term health effects and that surveillance of disease should include monitoring for persons with “long” COVID.
- Fifi et al. COVID-19 related stroke in young individuals.external icon Lancet Neurology. Brief review and commentary on reports of stroke associated with SARS-CoV-2 infection in relatively young patients (<50 years old) are increasing, even in patients with mild symptoms.
Health Policy
- Teixeira da Silva et al. Policy determinants of COVID-19 pandemic-induced fatality across nations.external icon Public Health. Delays in policy implementation (testing, international travel restrictions, public health information campaigns) led to a higher case fatality rate internationally than countries with more rapid policy implementation rates (p = 0.0013).
- Rahi et al. Mass vaccination against COVID-19 may require replays of the polio vaccination drives.external icon eClinicalMedicine. Authors suggest lessons learned from polio vaccination campaigns be implemented for future COVID-19 vaccines to avoid roadblocks to effective vaccine rollout.
- Boulware L. Race disparities in the COVID-19 pandemic—Solutions lie in policy, not biologyexternal icon. JAMA Internal Medicine. Commentary on Yehia paper that argues for policies that improve social and environmental conditions leading to poor health and ensure access to health care for Black U.S. residents.
- Sommers et al. Prescribing paid sick leave—An important tool for primary care during the pandemic.external icon JAMA. Families First Coronavirus Response Act allows employees to obtain paid leave for up to 80 hours if they have or are suspected to have COVID-19 and can’t work from home.
Other articles of interest
- Bhaskar et al. SARS-CoV-2 infection among community health workers in India before and after use of face shields.external icon JAMA. After face shields were added to PPE (mask, gloves, hand sanitizer, and shoe covers), community health worker infection rate decreased from 19% to 0%.
- Bunders et al. Implications of sex differences in immunity for SARS-CoV-2 pathogenesis and design of therapeutic interventions.external icon Immunity. Differences in immunity between men and women that might lead to more severe SARS-CoV-2 infections in men.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.