COVID-19 Science Update released: July 31, 2020 Edition 35

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Here you can find all previous COVID-19 Science Updates.
PEER-REVIEWED
Neonatal management and outcomes during the COVID-19 pandemic: An observation cohort studyexternal icon. Salvatore et al. Lancet Child & Adolescent Health (July 23, 2020).
Key findings:
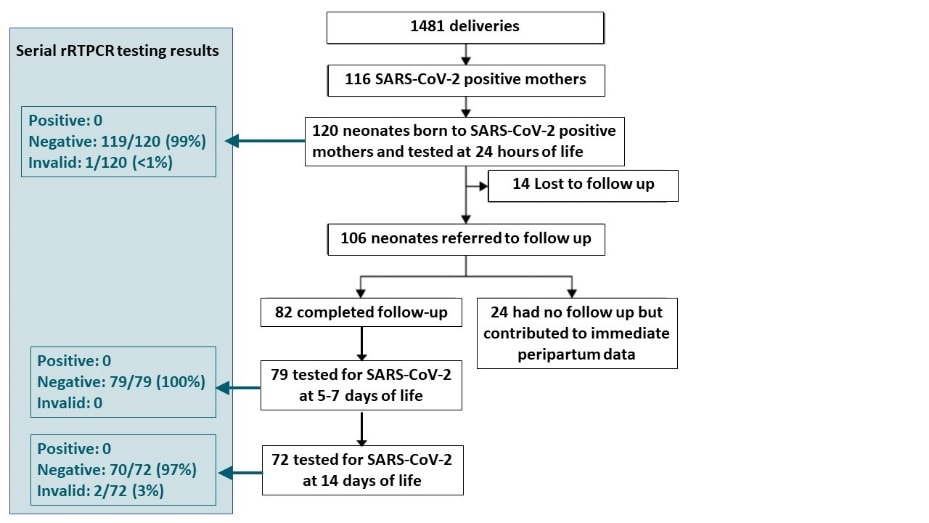
- 116 women positive for SARS-CoV-2 gave birth to 120 neonates.
- None of the neonates tested at 24 hours were positive for SARS-CoV-2 (Figure).
- 58 (74%) of 78 mothers for whom data were available were symptomatic.
- Of 82 neonates followed throughout the observation period:
- 68 roomed with mothers, and median length of hospital stay was 2 days (range 1-21 days).
- At 5-7 days of life, 64 (78%) were receiving breastmilk.
- None tested positive for SARS-CoV-2 at 5-7 days or 14 days (Figure).
- None showed signs or symptoms at 1 month.
- Of 73 parents assessed at 14 days, 85% reported always using masks, and 96% reported appropriate hand hygiene.
Methods: Observational cohort study, March 22-May 17, 2020, at 3 hospitals in New York City. Neonates born to mothers positive for SARS-CoV-2 at delivery were followed. Neonates were tested for SARS-CoV-2 by real-time reverse-transcriptase-polymerase chain reaction (rRT-PCR) on NP taken at 24 hours, 5–7 days, and 14 days of life (as outpatient care); neonates were also clinically evaluated by telemedicine at 1 month of age. Mothers had to wear a surgical mask when near their neonate and practice proper hand hygiene before skin-to-skin contact, breastfeeding, and routine care and were advised to continue to wear masks and practice hygiene after going home with neonates. Limitations: Significant attrition at follow-up; self-reported mask use and hand hygiene.
Implications: In this cohort, no infections were transmitted from mother to child during and in the few weeks after birth. Keeping neonates with their mothers, including skin-to-skin contact activities such as holding and breastfeeding, appears safe if appropriate hygiene precautions are observed. Parental education and support facilitate infant protective strategies.
Figure:
Note: Adapted from Salvatore et al. Study participant flow and rRT-PCR test results. This article was published in Lancet Child & Adolescent Health, Vol 4, Salvatore et al., Neonatal management and outcomes during the COVID-19 pandemic: An observation cohort study, P721-727, Copyright Elsevier 2020. This article is currently available at the Elsevier COVID-19 resource center: https://www.elsevier.com/connect/coronavirus-information-centerexternal icon.
Transplacental transmission of SARS-CoV-2 infectionexternal icon. Vivanti et al. Nature Communications (July 14, 2020).
Key findings:
- A male neonate born to a mother with confirmed COVID-19 was found to have SARS-CoV-2 infection.
- Both E and S genes of SARS-CoV-2 were found in placenta, amniotic fluid, cord blood, and mother’s and baby’s blood, indicating the presence of maternal viremia, placental infection, and neonatal viremia.
- Viral load was much higher in the placental tissue than in amniotic fluid or maternal blood, consistent with findings of inflammation seen at histological examination.
- Neonate presented with neurological manifestations and magnetic resonance imaging showed gliosis, a sign of infection.
- About two months later, neonate showed neurological improvement and normal growth.
Methods: A 23-year-old woman was admitted to hospital at 35 weeks of gestation with confirmed SARS-CoV-2 infection. After 3 days, the fetus developed intrauterine tachycardia, and an emergency caesarean delivery was performed. Biological samples from mother and newborn (Nb) were tested with RT-PCR targeting the envelope (E) gene (specific for lineage B-betacoronavirus) and the spike (S) gene (specific for SARS-CoV-2). Limitations: Single case; viral culture not performed.
Implications: This the first well-documented case consistent with transplacental viral transmission. Although published experience to date indicates a very low risk of perinatal SARS-CoV-2 transmission, risk of transplacental transmission may need to be considered in clinical procedures and practice guidelines.
PEER-REVIEWED
Seroprevalence of antibodies to SARS-CoV-2 in 10 sites in the United States, March 23-May 12, 2020external icon. Havers et al. JAMA Internal Medicine (July 21, 2020).
Key findings:
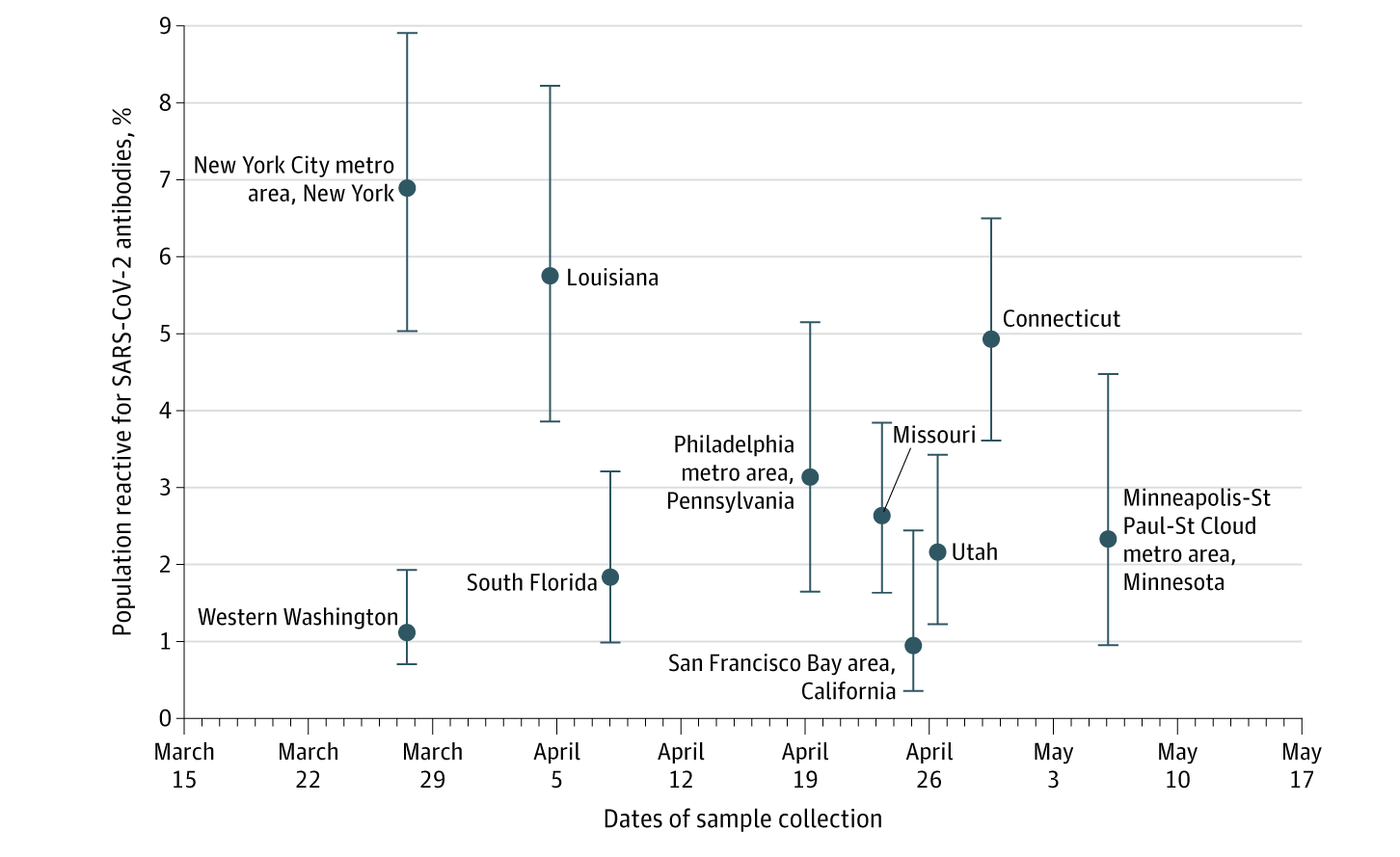
- Adjusted estimates of the proportion of persons with detectable SARS-CoV-2 antibodies ranged from 1.0% in the San Francisco Bay area (collected April 23-27, 2020) to 6.9% of persons in New York City (collected March 23-April 1, 2020) (Figure).
- 6-24 times more infections were estimated per site with seroprevalence testing than COVID-19 case reports indicate.
- In 7 sites (Connecticut, Florida, Louisiana, Missouri, New York City metro area, Utah, and western Washington State), there were more than 10 times more estimated SARS-CoV-2 infections than the number of reported cases.
Methods: A cross-sectional study of 16,025 residual clinical specimens from 2 commercial diagnostic laboratories to estimate the proportion of persons with detectable antibodies to SARS-CoV-2 spike protein in 10 US sites, between March 23 and May 12, 2020. Estimates were standardized by age and sex and adjusted for test performance characteristics (96.0% sensitivity and 99.3% specificity). Estimates were extrapolated to site populations and compared with the number of reported COVID-19 cases. Limitations: Specimens were obtained from people seeking health care and might not be representative of the population.
Implications: Reported cases of COVID-19 substantially underestimate the prevalence of infection.
Figure:
Note: Adapted from Havers et al. Estimates of seroprevalence to SARS-CoV-2 antibodies. Teal dots represent population prevalence estimates for antibodies, adjusted for age and sex and test performance characteristics; whisker bars represent 95% confidence intervals. Reproduced with permission from JAMA Intern Med. doi:10.1001/jamainternmed.2020.4130. Copyright©2020 American Medical Association. All rights reserved.
The contribution of the age distribution of cases to COVID-19 case fatality across countries: A 9-country demographic studyexternal icon. Sudharsanan et al. Annals of Internal Medicine. (July 22, 2020).
Key findings:
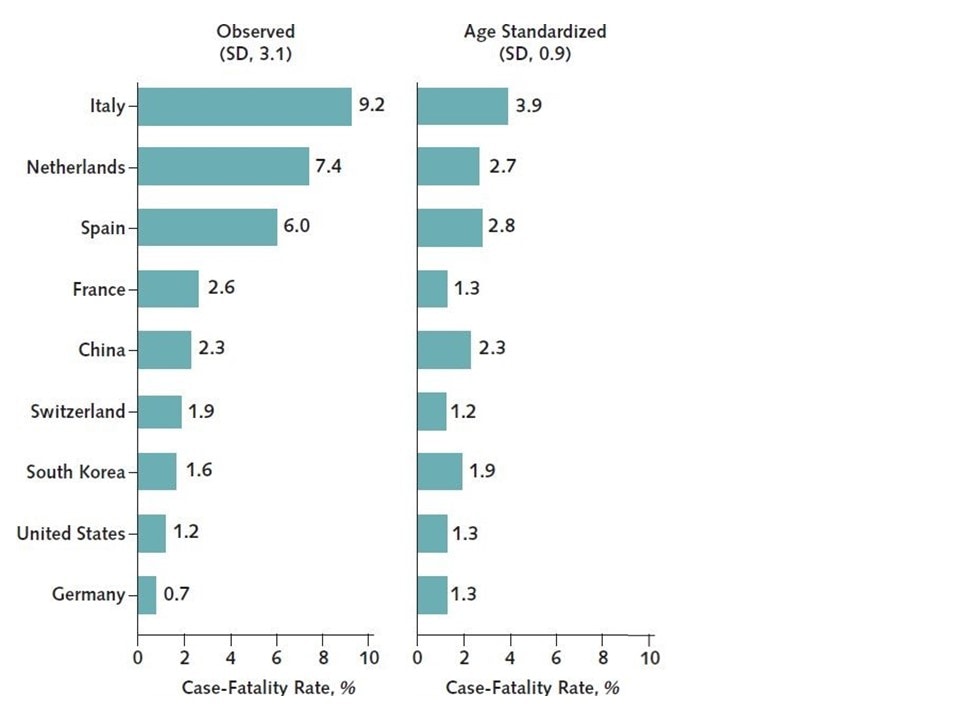
- Observed (unadjusted) COVID-19 case-fatality rates (CFRs) varied widely across 9 countries, with a range of 0.7% in Germany to 9.3% in Italy (Figure).
- Age-standardized median CFR was 1.9%, with a range of 1.2% to 3.9% (Figure).
- Adjustment for the age distribution of cases in each country significantly affected most national estimates and explained 66% of the CFR variation of across countries.
Methods: A cross-sectional demographic study of age-specific COVID-19 CFRs using aggregate population-based data on confirmed COVID-19 cases (N = 1,223,261) from China, France, Germany, Italy, the Netherlands, South Korea, Spain, Switzerland, and the United States. Age-standardized rates were calculated using the mean age distribution of diagnosed SARS-CoV-2 infections across the 9 countries. Limitations: Assumption that COVID-19 mortality among diagnosed cases is the same as that among undiagnosed cases and that individuals of all ages are equally susceptible to SARS-CoV-2 infection.
Implications: Age distributions of cases had a substantial effect on COVID-19 CFR estimates, although meaningful differences remain between countries after correcting for age. Strategies for reducing COVID-19 mortality still need to be attentive to underlying population health status and healthcare system characteristics.
Figure:
Note: Adapted from Sudharsanan et al. Observed and age-standardized COVID-19 case-fatality rates in 9 countries. Available via American College of Physicians Public Health Emergency Collection through PubMed Central.
Mental health before and during the COVID-19 pandemic: A longitudinal probability sample survey of the UK populationexternal icon. Pierce et al. Lancet Psychiatry (July 21, 2020).
Key findings:
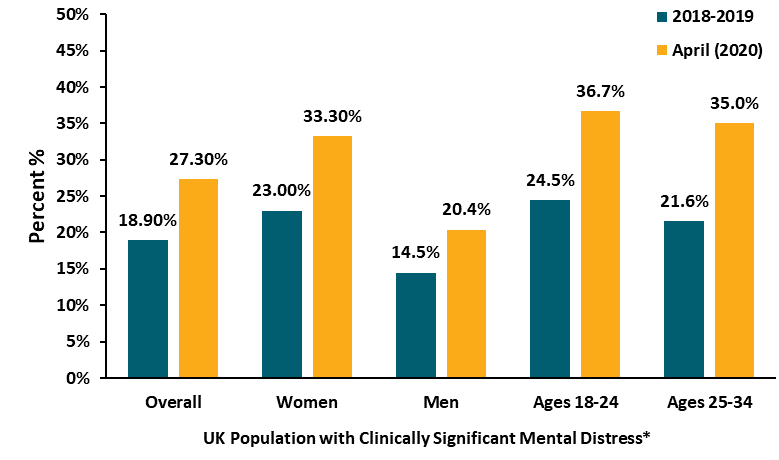
- In one year, clinically significant levels of mental distress rose from 18.9% (95% CI 17.8–20.0) to 27.3% (95% CI 26.3–28.2) (Figure).
- Controlling for other variables, the greatest changes were observed among women, younger persons (ages 18-24 and 25-34 years), and people who lived with children <5 years old.
Methods: Analysis of a web-based survey of 17,452 household members of UK Household Longitudinal Study (UKHLS) participants, between April 23 and 30, 2020, to track temporal changes in population mental health due to the COVID-19 pandemic. Mental distress was measured with the 12-item General Health Questionnaire (GHQ-12). Results were compared with scores from prior years. Limitations: Attrition and potential self-report biases due to online response format; GHQ-12 is not a diagnostic tool.
Implications: Mental distress increased in the UK population during the COVID-19 pandemic, with some subgroups disproportionately affected. Monitoring population-level mental health can inform development of public health responses to mitigate mental health aspects of the pandemic.
Figure:
Note: Adapted from Pierce et al. Proportion of participants with a clinically significant level of mental distress, by year. Data are from the 2018-2019 wave of UKHLS (n = 12,312) and April 2020 web-based survey of UKHLS household members (n = 17,452). *Mental distress is measured using GHQ-12. This article was published in Lancet Psychiatry, Vol 7, Pierce et al., Mental health before and during the COVID-19 pandemic: A longitudinal probability sample survey of the UK population, P883-892, Copyright Elsevier 2020. This article is currently available at the Elsevier COVID-19 resource center: https://www.elsevier.com/connect/coronavirus-information-centerexternal icon.
Potential influence of menstrual status and sex hormones on female SARS-CoV-2 infection: A cross-sectional study from multicentre in Wuhan, Chinaexternal icon. Ding et al. Clinical Infectious Diseases. (July 22, 2020)
Key findings:
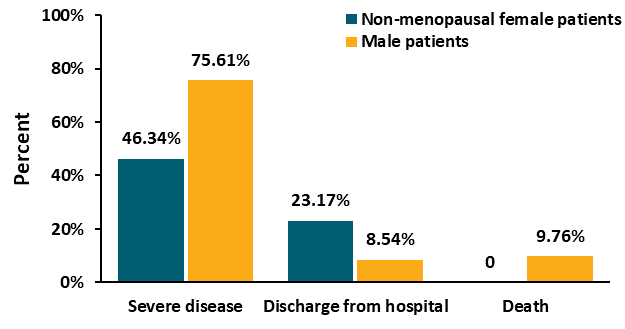
- Compared with age-matched male patients, pre-menopausal female patients were less likely to have severe COVID-19, had lower mortality, and were more likely to be discharged from hospital (Figure).
- Menopausal female patients did not differ from age-matched male patients.
- Among female patients <60 years of age, menopausal patients had longer hospitalization times than non-menopausal patients, hazard ratio (HR) 1.91 (95% CI 1.06-3.46), adjusted for age, comorbidities and severity of disease.
- In patients with measures of serum levels of sex hormones, anti-müllerian hormone (AMH) and estradiol were inversely associated with severity of infection.
- For AMH, adjusted HR 0.15 (95% CI 0.03-0.82).
- For estradiol, adjusted HR 0.30 (95% CI 0.09-1.00).
Methods: Cross-sectional clinical study among COVID-19-confirmed patients (N = 1,902) hospitalized in
Wuhan, China, between January 28 and March 8, 2020. Female patients, stratified by menopausal status (defined as amenorrhea for >1 year), were compared to age-matched male patients and evaluated for severity of disease and clinical outcomes, mortality, and discharge. 435 female patients <60 years were also stratified by menopausal status and compared for the same outcomes. 78 female patients were assessed for serum levels of sex hormones. Limitations: Non-randomized study; hospitalized sample; small numbers for some analyses.
Implications: Post-menopausal females might be at higher risk for complications from COVID-19 than women who have not experienced menopause. AMH and estradiol might be potential protective factors against COVID-19. Hormone supplement might be a potential therapy for female COVID-19 patients.
Figure:
Note: Adapted from Ding et al. Disease severity and clinical outcomes. Patients were age-matched for comparisons. Classification for severe disease followed guidelines of the American Thoracic Society pertaining to community-acquired pneumonia. For severe disease (versus non-severe disease), p <0.01; for the composite comparison of discharge (versus remaining in hospital) and death, p <0.01. Available via Oxford University Press Public Health Emergency Collection through PubMed Central.
COVID-19 and inequity: A comparative spatial analysis of New York City and Chicago hot spotsexternal icon. Maroko et al. Journal of Urban Health (July 20, 2020).
Key findings:
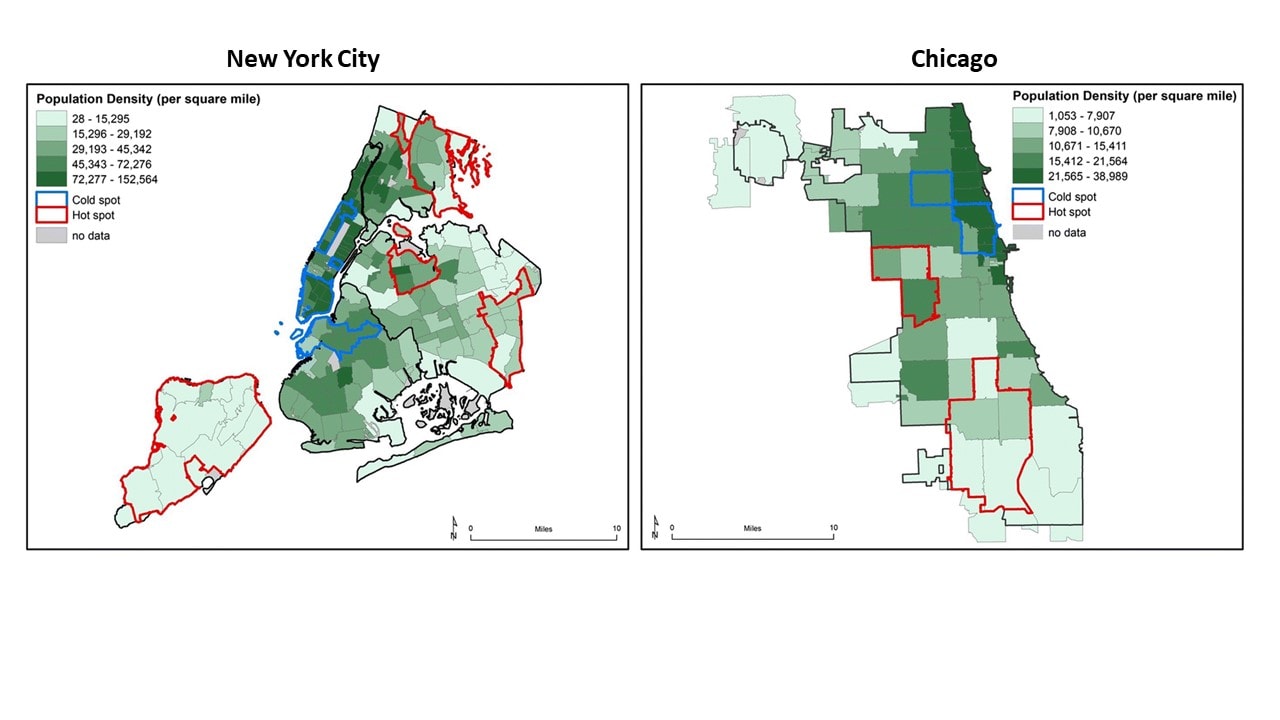
- In both New York City (NYC) and Chicago, greater mean household size, but lower neighborhood density, were associated with COVID-19 “hot spots” (Figure).
- NYC: 3.0 people per household in hot spots compared with 2.1 in cold spots; p = 0.01.
- Chicago: 2.8 people per household in hot spots compared with 2.0 in cold spots; p = 0.03.
- NYC: 22,900 people per square mile in hot spots and 68,900 in cold spots, p <0.01.
- Chicago: 10,000 people per square mile in hot spots and 23,400 in cold spots, p = 0.02.
- In both NYC and Chicago, hot spots, compared with cold spots, had significantly:
- Higher proportions of Black/African-American residents and older residents.
- Lower proportions of non-Hispanic White residents as well as workers in managerial occupations and college graduates.
- In Chicago, hotspots had higher poverty rates compared with cold spots (31.2% vs 9.9%, p <0.01).
Methods: Ecological cross-sectional study to examine spatial and demographic nature of reported COVID-19 diagnoses in New York City and Chicago as of April 13, 2020. COVID-19 high and low prevalence areas (“hot spots” and “cold spots”) were calculated using zip code tabulation areas (ZCTA). Limitations: Data may not represent the true distribution of COVID-19 cases based differential testing or access to testing. Results characterize neighborhoods and may not accurately represent the characteristics of individuals living in those neighborhoods.
Implications: Demographic and economic characteristics of hot spots may help inform public health prevention and mitigation strategies and resource allocation to reduce the vulnerability of critical populations to the impact of COVID-19.
Figure:
Note: From Maroko et al. ZCTA-level population density (people per square mile) for NYC and Chicago in quintiles. Cities are shown at different scales. Used by permission of Springer Nature.
PREPRINTS (NOT PEER-REVIEWED)
Clinical ordering practices of the SARS-CoV-2 antibody test at a large academic medical centerexternal icon. Wiencek et al. medRxiv (July 14, 2020). Publishedexternal icon in Open Forum Infectious Diseases (October 2020).
Key findings:
- Clinicians’ top reasons for ordering a SARS-CoV-2 antibody test:
- Public health epidemiology studies (n = 223).
- Patients with resolved COVID-19-compatible illness (n = 105) with no previous molecular testing.
- Asymptomatic patients believed to have had a past exposure (n = 60).
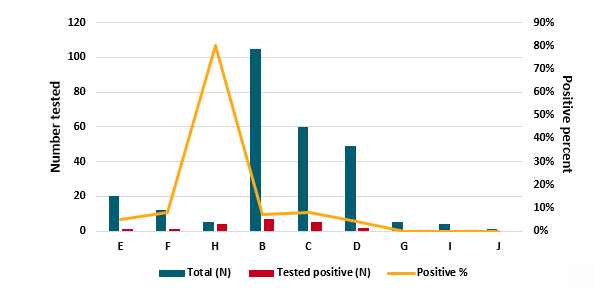
- Results were 17% positive when conducted according to current guidance, and 5% when not consistent with guidance (p <0.0001) (Figure).
- Testing consistent with guidance, excluding epidemiologic study data, had the lowest cost-per-positive test ($758.33-$1,283.33); but represented the lowest share of testing (8%).
Methods: Retrospective chart review of 447 patients who received SARS-CoV-2 antibody testing between May 14 and June 15, 2020, to describe clinician-described indications, cost implications, and comparison with current IDSA and CDC guidance. Limitations: True positives may be inaccurately counted due to timing of testing, symptom level of patients, and test characteristics.
Implications: Indications for testing that do not align with guidance lead to lower rates of positive results, which is unlikely to be of clinical value. SARS-CoV-2 antibody testing yield and utility is improved, and cost is reduced, when conducted according to guidance.
Figure:
Note: Adapted from Wiencek et al. Total (N) testing for SARS-CoV-2 antibodies according to expert guidance and Tested Positive (N) are shown on left axis, and Positive % is shown on right axis. Indication categories E-RT-PCR negative cases or when lower respiratory tract sampling is not possible; F-Patients presenting with late complications of COVID-19 illness, such as multisystem inflammatory syndrome in children; H-Convalescent plasma donors, (all consistent with guidance). B-Patients recovered from COVID-19-compatible illness but never had RT-PCR testing; C-Asymptomatic person believed to have had past close contact with a person with COVID-19 compatible illness; D-No indication provided; G-Other; I-Support a diagnosis of COVID-19 in a symptomatic patient in the absence of RT-PCR testing; J-confirm immune status for return to work, school, etc., (not consistent with guidance).Testing for public health epidemiology studies is not presented. Licensed under CC-BY-NC-ND 4.0.
PEER-REVIEWED
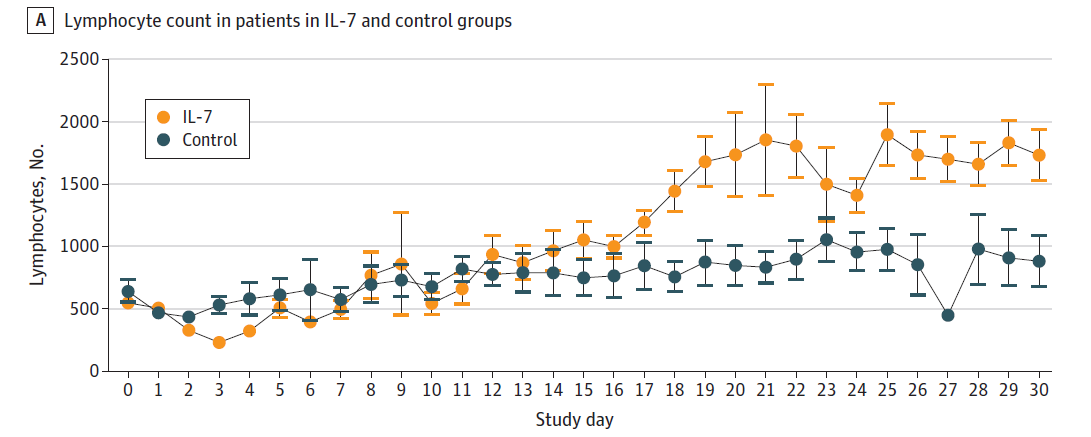
Association of interleukin 7 immunotherapy with lymphocyte counts among patients with severe coronavirus disease 2019 (COVID-19)external icon. Laterre et al. JAMA Network Open (July 22, 2020).
Key findings:
- IL-7 was associated with restored lymphocyte counts to reference levels at 30 days after administration.
- Patients who received IL-7 had than twice the number of lymphocytes 30 days after its administration than patients who did not: 1734/μL versus 885/μL, respectively, p = 0.02.
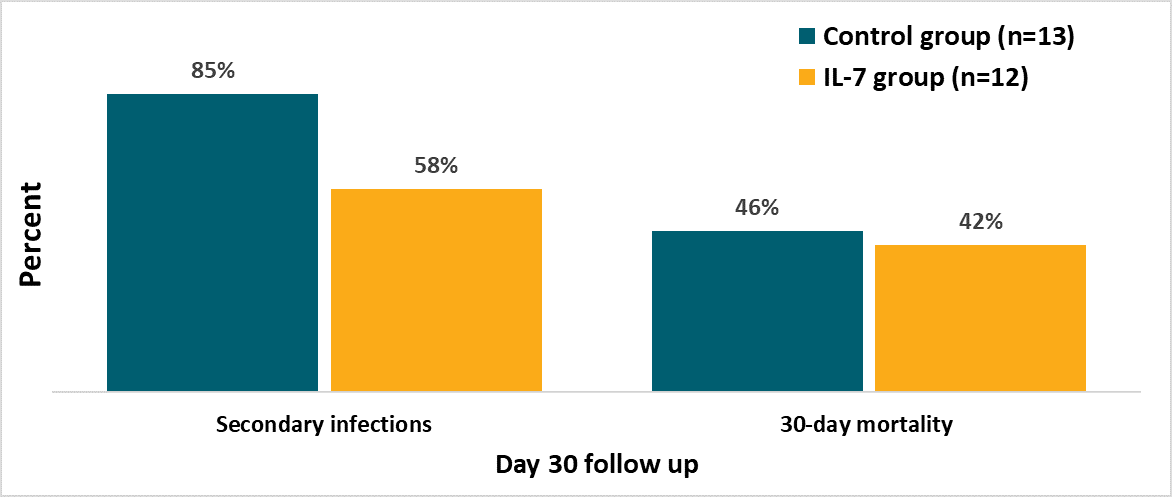
- At 30 days post administration, secondary infections had occurred in 7 patients (58%) in the IL-7 group compared with 11 (85%) in the control group; 30-day mortality was 42% (5) and 46% (6), respectively.
Methods: Series of 12 critically ill COVID-19 patients receiving interleukin 7 (IL-7, a cytokine essential for lymphocyte survival and expansion) treatment, compared with 13 COVID-19 patients who received standard-of-care treatment, matched for comorbidities and other factors. Proinflammatory cytokines were measured before and at 7 and 24 hours after IL-7 administration. Lymphocyte levels, secondary infections and mortality were measured out to 30 days after administration of IL-7. Limitations: Small samples; potential competing mechanisms to recovery in some patients; no phenotypic or functional studies of immune cells.
Implications: This small trial indicates that IL-7 can be administered to critically ill patients with COVID-19 without exacerbating inflammation or pulmonary injury and with possible clinical benefit.
Figure 1
Note: Adapted from Laterre et al. At day 30, secondary infections had occurred in 7 patients (58%) in the IL-7 group compared with 11 (85%) in the control group; 30-day mortality was 42% (5) and 46% (6), respectively. Licensed under CC-BY.
Note: Adapted from LaTerre et al. Lymphocyte count in patients in IL-7 and control groups. IL-7 group (n = 12) compared with control group (n = 13) with absolute lymphocyte count across 30 days after IL-7 administration. Licensed under CC-BY.
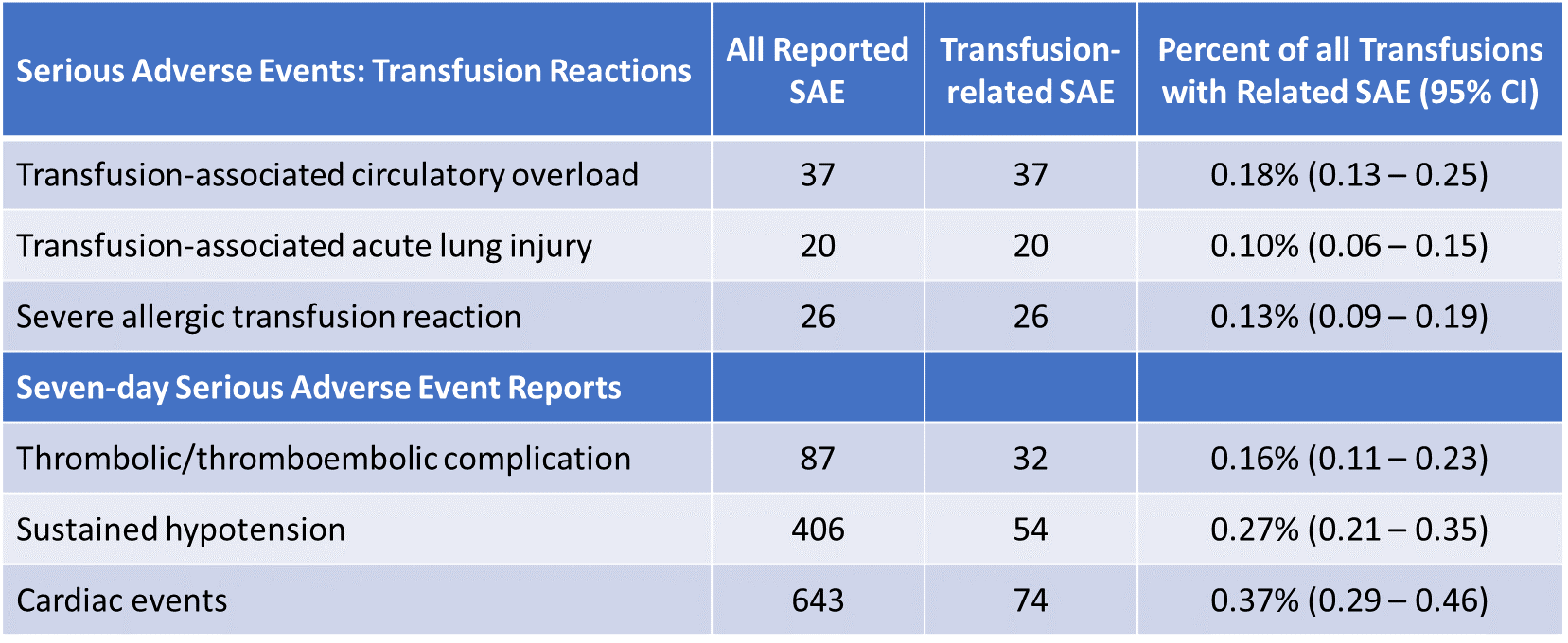
Safety update: COVID-19 convalescent plasma in 20,000 hospitalized patientsexternal icon. Joyner et al. Mayo Clinic Proceedings (July 19, 2020).
Key findings:
- 1,282 serious adverse events (SAE) post convalescent plasma transfusion occurred, but transfusion-related SAE occurred in <1% of patients (Table).
- 13 of 20,000 patients (0.06%, 95% CI 0.04%-0.11%) died within four hours of transfusions for transfusion-related reasons.
- Overall, 7-day post-transfusion mortality rate was 8.56% (95% CI 8.18%-8.95%).
Methods: Multi-site cohort of 20,000 hospitalized patients (60.8% male, 52.1% obese) between April 3 and June 2, 2020. Patients were >18 years, had laboratory-confirmed SARS-CoV-2 infection, and severe or life-threatening COVID-19 or were judged to be at high risk of progression to severe disease. Patients received 200-500 ml of ABO-compatible COVID-19 convalescent plasma donated by recently recovered COVID-19 survivors. Limitations: Short follow up window; not designed to evaluate efficacy of convalescent plasma.
Implications: Convalescent plasma appears generally safe. Data on efficacy of convalescent plasma for treatment of COVID-19 are needed.
Table:
Note: Adapted from Joyner et al. SAE- Serious Adverse Events. All reported SAE- all SAE occurring among transfused patients up to 7 days post-transfusion; Transfusion-related SAE- SAE occurring among transfused patients attributed to receipt of transfusion. Percentages and 95% CI calculated as fractions of 20,000 patients receiving convalescent plasma via transfusion. This article was published in Mayo Clinic Proceedings, Vol 95, Joyner et al., Safety update: COVID-19 convalescent plasma in 20,000 hospitalized patients, Page 1888-1897, Copyright Mayo Foundation for Medical Education and Research 2020. This article is currently available at the Elsevier COVID-19 resource center: https://www.elsevier.com/connect/coronavirus-information-centerexternal icon.
PEER-REVIEWED
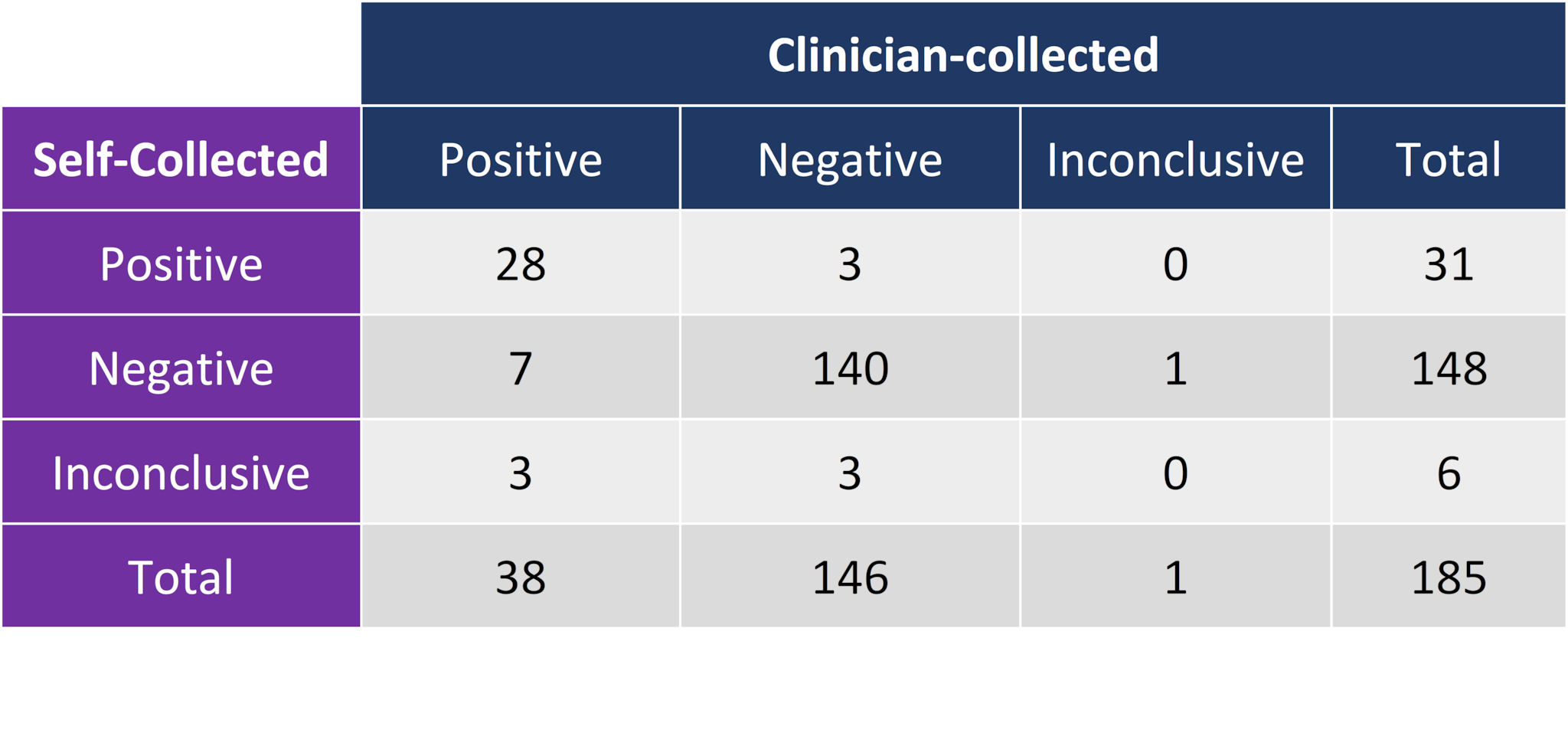
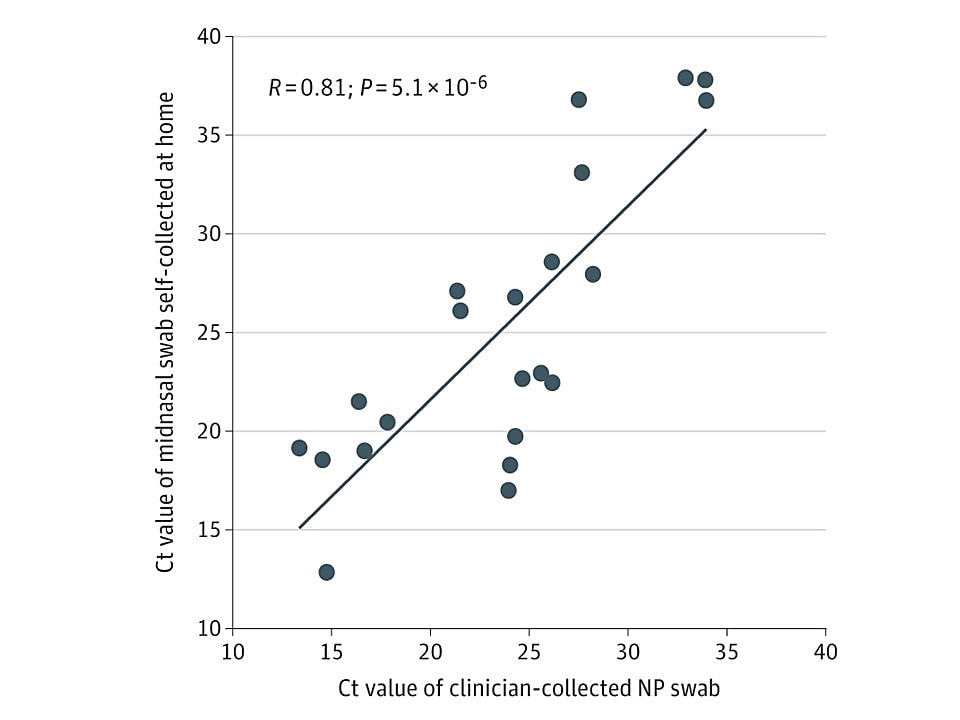
Comparison of unsupervised home self-collected midnasal swabs with clinician-collected nasopharyngeal swabs for detection of SARS-CoV-2 infectionexternal icon. McCulloch et al. JAMA Network Open (July 22, 2020).
Key findings:
- 41 (22.2%) participants had SARS-CoV-2 positive test results via clinician-collected NP swab, self-collected mid-nasal swab, or both (Table).
- Sensitivity self-collected swabs was 80.0% (95% CI 63%-91%)
- Specificity of self-collected swabs was 97.9% (95% CI 94%-99.5%).
- Ct values (i.e., viral burden) of self-collected swabs were highly correlated with those of clinician-collected swabs (R = 0.81; p <0.001) (Figure).
Methods: Cross-sectional study of symptomatic patients who tested positive for SARS-CoV-2 (n = 27) and symptomatic health care workers (n = 158), comparing performance of home self-collected swab with clinician-collected NP swabs for RT-PCR testing for SARS-CoV-2, between March 31 and April 13, 2020. Limitations: Relatively small sample; health care workers might have been unusually proficient at self-collected swabs, 1-day average delay in processing self-collected specimens might lead to false-negative test results.
Implications: Detection of SARS-CoV-2 using self-collected mid-nasal swabs was comparable to clinician-collected NP swabs. This approach may be safe and scalable, enabling increased access to testing.
Table:
Note. Adapted from McCulloch et al. Clinician-collected NP swabs are the gold standard for performance assessment. Licensed under CC-BY.
Figure:
Note: Adapted from McCulloch et al. Correlation of Ct values (teal dots) for home self-collected mid-nasal swabs (n = 28) and clinician-collected nasopharyngeal swabs (n = 30). Licensed under CC-BY.
SARS-CoV-2 Detection
- Brown et al. Serosurveillance and the COVID-19 epidemic in the US: Undetected, uncertain, and out of control.external icon Emphasizes the need to uncover the cumulative incidence of infection in a comment on Havers et al. (this update).
- Edson et al. Identification of SARS-CoV-2 in a proficiency testing program.external icon American Journal of Clinical Pathology. Assesses accuracy of SARS-CoV-2 testing at 346 laboratories in the U.S.
- Tromberg et al. Rapid scaling up of COVID-19 diagnostic testing in the United States — The NIH RADx Initiative.external icon Describes the four components of the Rapid Acceleration of Diagnostics (RADx) program, including a focus on underserved populations.
- Greene et al. Detecting emerging COVID-19 community outbreaks at high spatiotemporal resolution – New York City, June 2020.external icon Spatiotemporal analysis of COVID-19 data facilitated swift cluster detection and community mitigation activities.
Clinical Management and Treatment
- Spence et al. Mechanisms of stroke in COVID-19.external icon Cerebrovascular Diseases. Suggests three main mechanisms as responsible for the occurrence of ischemic strokes in COVID-19 patients specifically related to COVID-19: hypercoagulable state, vasculitis, and cardiomyopathy.
- Capobianco et al. COVID-19 in pregnant women: A systematic review and meta-analysis.external icon European Journal of Obstetrics & Gynecology and Reproductive Biology. Meta-analysis concluding high rates of maternal and neonatal complications in infected individuals, but low risk of neonatal infection.
- Keswani et al. Atopy is predictive of a decreased need for hospitalization for COVID-19.external icon Annals of Allergy, Asthma and Immunology. A co-existing atopic background may mitigate the severe inflammatory response syndrome of COVID-19 in allergic asthmatics, resulting in less severe disease.
Vaccine Development
- Eyal et al. It’s ethical to test promising coronavirus vaccines against less-promising ones. pdf iconexternal iconProceedings of the National Academy of Sciences. Presents an ethical case for testing vaccine efficacy even in the face of challenges to equipoise based on early data.
- Bar-Zeev et al. Encouraging results from phase 1/2 COVID-19 vaccine trials.external icon Discusses promising safety and immunologic profiles from trials and proposes conditions for efficacy trials and equitable distribution of vaccine.
Other Topics
- Bahl et al. Face coverings and mask to minimise droplet dispersion and aerosolisation: a video case study.external icon Provides visual evidence of the efficacy of various face coverings to reduce droplets and aerosols expelled during speaking, coughing and sneezing.
- Egede et al. Structural racism, social risk factors, and COVID-19 — A dangerous convergence for Black Americans.external icon NEJM. Identifies six recommendations to mitigate structural racism and its effects on racial health disparities in the context of COVID-19.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.