COVID-19 Science Update released: July 2, 2020 Edition 27

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Here you can find all previous COVID-19 Science Updates.
PEER-REVIEWED
COVID-19 in patients with rheumatic diseases in northern Italy: A single-centre observational study and case-control studyexternal icon. Fredi et al. Lancet Rheumatology (June 18, 2020).
Key findings:
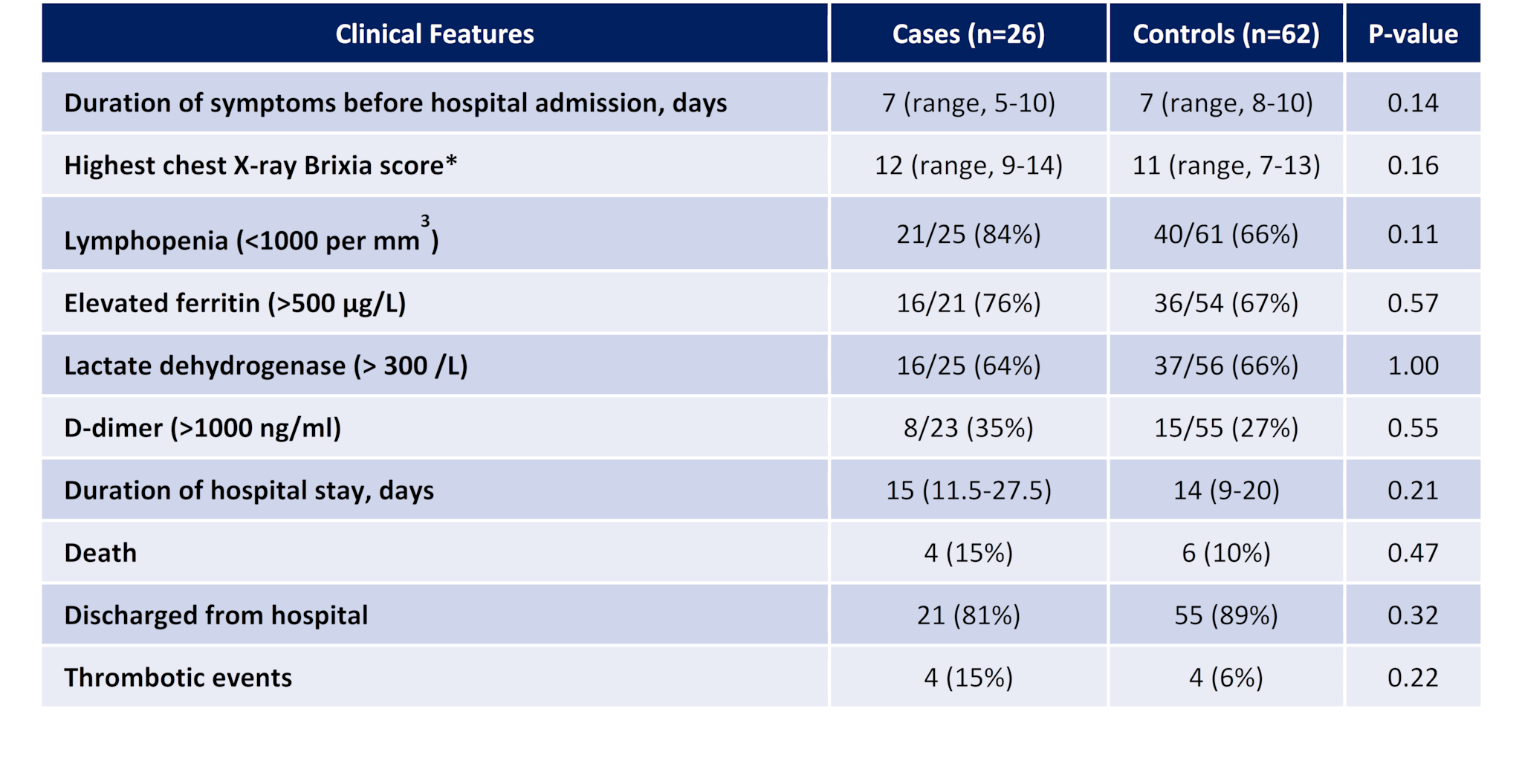
- Clinical features of COVID-19 patients with musculoskeletal or rheumatic diseases were similar to patients without musculoskeletal or rheumatic diseases (controls) (Table).
Methods: Age-, sex-, and month of admission- matched case-control study of 26 COVID-19 pneumonia patients with musculoskeletal and rheumatic diseases compared to 62 COVID-19 pneumonia patients without these diseases. Limitations: Observational study, one site.
Implications: COVID-19 patients with pre-existing musculoskeletal or rheumatic diseases did not have a different clinical course compared to other COVID-19 patients.
Table:
Note: Adapted from Fredi et al. Select clinical characteristics of cases (COVID-19 pneumonia patients with musculoskeletal or rheumatic disease) and controls (age-, sex- and month of admission-matched COVID-19 pneumonia patients without musculoskeletal or rheumatic diseases). *Brixia score is a radiologic interstitial pneumonia scoring with highest score being more severe pneumonia. This article was published in Lancet Rheumatology, Vol 2, Fredi et al., COVID-19 in patients with rheumatic diseases in northern Italy: A single-centre observational study and case-control study, Page e549-e556, Copyright Elsevier 2020. This article is currently available at the Elsevier COVID-19 resource center: https://www.elsevier.com/connect/coronavirus-information-centerexternal icon.
Disparities in Coronavirus 2019 reported incidence, knowledge, and behavior among US adultsexternal icon. Alsan et al. JAMA Network Open. (June 18, 2020).
Key findings:
- African American persons (compared with White persons) and those aged 18-29 and 30-54 (compared with persons age 55-64) years were more likely to self-report having or having had COVID-19 or know someone who has or has had COVID-19.
- African American persons (compared with White persons), men (compared with women) and persons 18-29 and 30-54 years (compared with adults age 55-64 years) were less likely to know about COVID-19 symptoms and SARS-CoV-2 transmission through fomites (Figure 1).
- Men (compared with women) and persons 18-29 years (compared with adults age 55-64 years) were less likely to wash their hands frequently (Figure 2).
- African American persons (compared with White persons) and men (compared with women) were more likely to leave their house frequently, but no difference by age was observed (Figure 2).
Methods: Internet survey between March 29 and April 13, 2020 of 5,198 US residents ≥18 years that examined self-reported COVID-19 diagnosis, COVID-19-related knowledge and behaviors to reduce spread of infection. Point estimates were adjusted for demographics, income, political orientation, comorbidities, health insurance and state of residence. Limitations: Cross-sectional survey; response-bias; limited to persons with internet access; did not adjust for type of employment and ability to telecommute.
Implications: Racial, sex and age-based disparities in COVID-19-related knowledge and behaviors exist. Public health messages on COVID-19 and interventions to reduce transmission need to more effectively reach African Americans, men and younger persons.
Figure 1
Note: Adapted from Alsan et al. Point estimates and 95% CIs for knowledge about COVID-19 spread via fomites (teal) and knowledge about COVID-19 symptoms (red). Teal and red dashed vertical lines indicate the point estimate for the reference value for the respective preventive measures. Superscripts a, b, and c indicate the reference values for race/ethnicity (white), sex (female), and age group (55 – 64 years), respectively. Licensed under CC-BY.
Note: Adapted from Alsan et al. Point estimates and 95% CIs for frequency of leaving the house (teal) and washing hands (red). Teal and red dashed vertical lines indicate the point estimate for the reference value for the respective preventive measures. Superscripts a, b, and c indicate the reference values for race/ethnicity (white), sex (female), and age group (55 – 64 years), respectively. Licensed under CC-BY.
County-level association of social vulnerability with COVID-19 cases and deaths in the USAexternal icon. Khazanchi et al. Journal of General Internal Medicine (June 23, 2020).
Key findings:
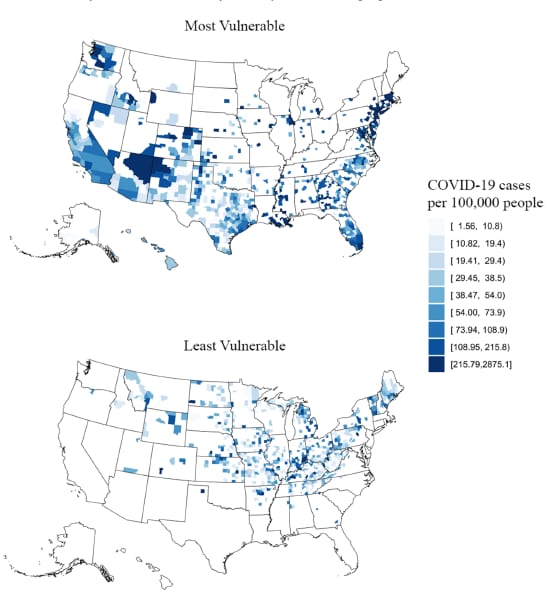
- People residing in counties with greater social vulnerability were more likely to be diagnosed with COVID-19 or die due to COVID-19, compared with people living in both urban and rural counties with lesser social vulnerability.
- The associations were most pronounced within the social vulnerability domain of combined minority status and language, which includes race, ethnicity, and English language proficiency (Figure).
Methods: Cross-sectional study examining county-specific social vulnerability indices and US COVID-19 case and deaths rates. Cases and deaths in the first and fourth quartiles of CDC’s Social Vulnerability Index overall and within each social vulnerability domain (minority status and language, socioeconomic status, housing and transportation, and household composition and disability) were compared. Limitations: Ecological study; individual-level effects not examined; causal relationships cannot be inferred; heterogeneity in testing rates and case reporting not accounted for.
Implications: Counties with higher levels of social vulnerability, such as those with higher proportion of racial/ethnic minorities and non-English speakers may benefit from targeted COVID-19 prevention and control strategies.
Figure:
Note: Adapted from Khazanchi et al. The most vulnerable quartile of counties (n = 706, top) and the least vulnerable quartile of counties (n = 625, bottom), as indicated by the minority status and language domain of CDC’s Social Vulnerability Index. The domain of combined minority status and language includes race, ethnicity, and English language proficiency. Available via Nature Public Health Emergency Collection through PubMed Central.
PREPRINTS (NOT PEER-REVIEWED)
Randomized re-opening of training facilities during the COVID-19 pandemicexternal icon. Bretthauer et al. medRxiv (June 25, 2020).
Key findings:
- In a study to assess transmission during two weeks in a “post-COVID” more distanced and hygienic gym environment, one case of SARS-CoV-2 infection was detected among 1,896 attendees.
- The case had not visited a training facility prior to SARS-CoV-2 testing.
- Investigations revealed that the case was likely infected in a separate workplace.
- None of 83 training facility employees tested positive for SARS-CoV-2 infection.
Methods: Members of five gyms in Oslo, Norway, 18-64 years with no history of cardiovascular disease or diabetes were randomized into training at the gym (N = 1,896) or no-training (N = 1,868). Facilities implemented social distancing and increased hygiene protocol. Self-administered NP and sputum samples were collected after two weeks for SARS-CoV-2 RT-PCR testing and electronic patient records were reviewed after three weeks. Limitations: Short observation period; self-collected specimen; low background prevalence of SARS-CoV-2.
Implications: Opening training facilities with good hygiene and social distance measures in similar settings is unlikely to lead to increased transmission of SARS-CoV-2.
PEER-REVIEWED
Effect of colchicine vs standard care on cardiac and inflammatory biomarkers and clinical outcomes in patients hospitalized with coronavirus disease 2019 the GRECCO-19 randomized clinical trialexternal icon. Deftereos et al. JAMA Network Open (June 24, 2020).
Key findings:
- The probability of survival was higher for patients treated with colchicine than standard therapy alone (97% vs 83% respectively; p = 0.03) (Figure).
- COVID-19 patients treated with colchicine had no significant changes in cardiovascular injury and inflammation markers (cardiac troponin and C-reactive protein).
- Adverse events were reported with similar frequency except for more frequent diarrhea in patients treated with colchicine (45%) compared with standard therapy alone (18%).
Methods: Prospective, open-label Phase 2 randomized clinical trial of 105 hospitalized symptomatic COVID-19 patients ≥18 years that compared colchicine and standard therapy to standard therapy alone, 16 Greek hospitals, April 3 -27, 2020. Limitations: Small sample size; not blinded.
Implications: Colchicine may be an effective treatment for hospitalized symptomatic COVID-19 patients. Larger randomized clinical trials in different populations are warranted.
Figure:
Note: Adapted from Deftereos et al. Proportion alive by days comparing colchicine to standard therapy. Licensed under CC-BY.
PREPRINTS (NOT PEER-REVIEWED)
Type and frequency of ocular and other known symptoms experienced by people who self–diagnosed as suffering from COVID-19 in the UKexternal icon. Pardhan et al. medRxiv. (June 22, 2020).
Key findings:
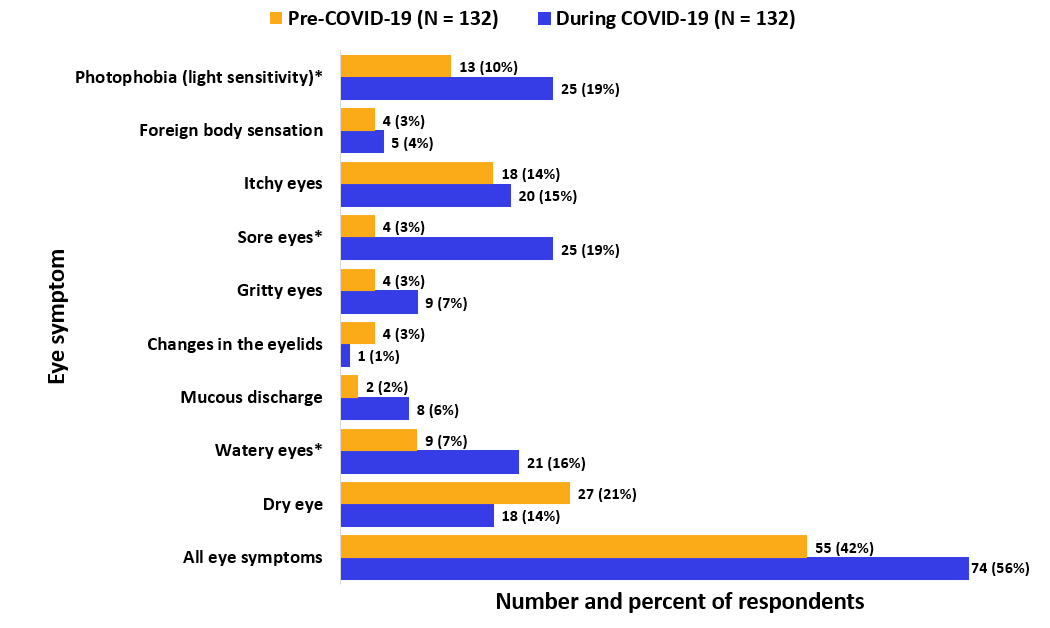
- 46% of survey respondents suspected to have COVID-19 reported an eye symptom indicative of viral conjunctivitis whilst also experiencing other COVID-19 symptoms.
- Watery eyes, sore eyes and photophobia (light sensitivity) were more frequently reported around the time of onset of other COVID-19 symptoms than before symptom onset (Figure).
- There was no difference in the duration of eye symptoms compared with duration of other symptoms.
Methods: Online survey of 132 adults with ≥2 COVID-19 symptoms that examined the type and frequency of eye symptoms, compared with other known COVID-19 symptoms. Limitations: Cross-sectional survey; self-reported COVID-19 and symptoms; participants were not tested for SARS-CoV-2.
Implications: Eye symptoms associated with SARS-CoV-2 infection may be overlooked in the presence of other well-known symptoms associated with severe disease. Although SARS-CoV-2 infection can cause eye symptoms like conjunctivitis, it remains to be definitively established whether the virus can infect the eyes or be transmitted by ocular duct and conjunctival fluids.
Figure:
Note: Adapted from Pardhan et al. Frequency of different eye symptoms before and during the COVID-19 symptoms in yellow and blue, respectively. Asterisk (*) indicates significant differences (p ≤0.02). Used by permission from author.
PEER-REVIEWED
Diagnostic accuracy of the FebriDx host response point-of-care test in patients hospitalised with suspected COVID-19external icon. Clark et al. Journal of Infection (June 21, 2020).
Key findings:
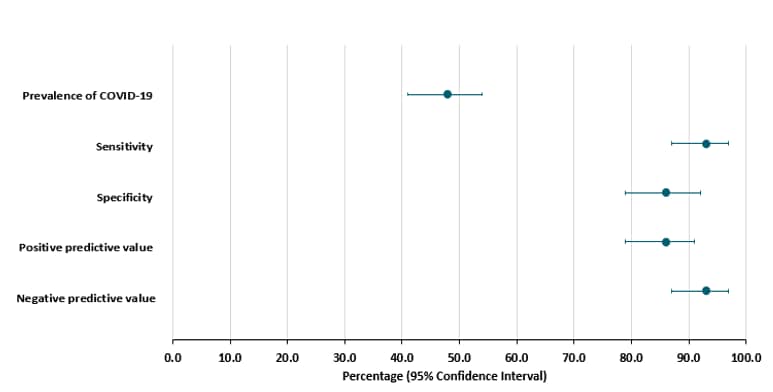
- FebriDx is a point-of-care test that detects host antiviral proteins (Myxovirus resistance protein A [MxA] and C-reactive protein [CRP]) in finger prick blood within 10 minutes.
- The sensitivity and specificity of FebriDx were high (93% and 86%, respectively) (Figure).
Methods: 251 hospitalized COVID-19 patients diagnosed by RT-PCR were tested with FebriDx. The RT-PCR result was used as the reference standard for sensitivity and specificity. Limitations: MxA and CRP are markers of antiviral host response and not specific for SARS-CoV-2; study performed in a high prevalence COVID-19 setting; study participants were almost exclusively immunocompetent adults.
Implications: FebriDx could be used to rapidly detect probable COVID-19 cases. However, detection of MxA and elevated CRP as markers of potential antiviral host response and is not specific for SARS-CoV-2; the high specificity of FebriDx MxA for COVID-19 may change with the seasonal circulation of other respiratory viruses such as influenza.
Figure:
Note: Adapted from Clark et al. Performance of FebriDx and 95% confidence intervals. Positive predictive value and negative predictive value calculated using the prevalence of COVID-19 in the study population (48%). This article was published in Journal of Infection, Vol 81, Clark et al., Diagnostic accuracy of the FebriDx host response point-of-care test in patients hospitalised with suspected COVID-19, Page 607-613, Copyright the British Infection Association 2020. This article is currently available at the Elsevier COVID-19 resource center: https://www.elsevier.com/connect/coronavirus-information-centerexternal icon.
- Edwards et al. Anticipating SARS-CoV-2 vaccine testing, licensure, and recommendations for use. Journal of Pediatrics.external icon The Journal of Pediatrics. Discusses the various steps of vaccine development and licensure to ensure the production and use of safe and effective vaccines.
- Mullangi et al. Shoring up the US safety net in the era of coronavirus disease 2019external icon. JAMA Health Forum. Discusses how the structure of safety-net hospitals puts them in a unique position to endure the COVID-19 pandemic and that with enough resources they can adequately serve underinsured and uninsured persons while also providing financial protection to neighboring hospitals.
- Wadman M. Vaccines that use human fetal cells draw fireexternal icon. Science. Religious and anti-abortion groups from the Unites States and Canada urge their respective governments against development and use of COVID-19 vaccines that rely on human fetal cells from elective abortions.
- Galiatsatos et al. Health equity and distributive justice considerations in critical care resource allocationexternal icon. Lancet. Objective scoring algorithms used to provide limited care resources need to be adapted to incorporate health equity principles before these types of algorithms are used to triage COVID-19 resources.
- Faris et al. Ramadan intermittent fasting and immunity: An important topic in the era of COVID-19.external icon Annals of Thoracic Medicine. Summarizes published studies on how intermittent fasting affects the immune system and potential implications during the COVID-19 pandemic.
- Varatharaj et al. Neurological and neuropsychiatric complications of COVID-19 in 153 patients: A UK-wide surveillance studyexternal icon. Lancet Psychiatry. Surveillance study that found stroke and altered mental status are the two most common neurological and psychiatric manifestations in COVID-19 patients.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.