COVID-19 Science Update released: June 23, 2020 Edition 24

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Here you can find all previous COVID-19 Science Updates.
PEER-REVIEWED
The epidemiology and clinical characteristics of co-infection of SARS-CoV-2 and influenza viruses in patients during COVID-19 outbreak.external icon Yue et al. Journal of Medical Virology (June 12, 2020).
Key findings:
- Of 307 patients with SARS-CoV-2, 153 (49.8%) had influenza A co-infection, 23 (7.5%) had influenza B co-infection.
- Age and gender were similar across groups.
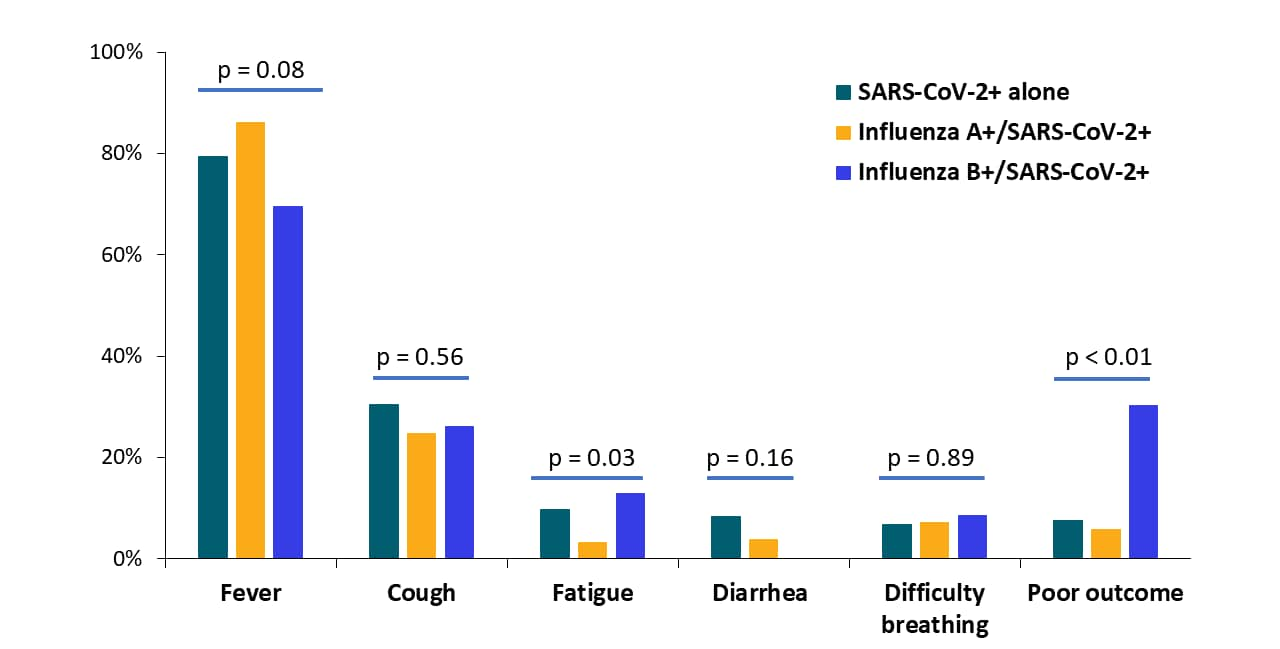
- COVID-19 patients co-infected with influenza A or B had similar clinical symptoms as patients with only SARS-CoV-2; fatigue was slightly more common those with influenza B co-infection (13%) (Figure).
- COVID-19 patients with influenza B more often had poor outcomes (30.4%) than those with only SARS-CoV-2 (7.6%) or SARS-CoV-2 and influenza A (5.9%) (Figure).
Methods: Cross-sectional study of 307 hospitalized COVID-19 patients, between January 12 and February 21, 2020, Wuhan, China. Limitations: Single center; small sample size, may not be representative; end point of poor outcome not clearly defined and likely includes death; number of deaths not reported.
Implications: In this small study, influenza B co-infection worsened COVID-19 prognosis.
Figure:
Notes: Adapted from Yue et al. Summary of symptoms and outcomes among patients with SARS-CoV-2 and co-infection with influenza viruses A or B. Statistical comparison among three groups was assessed using Pearson’s χ² test or Fisher’s exact test. Available via Wiley Public Health Emergency Collection through PubMed Central.
Effects of sterilization with hydrogen peroxide and chlorine dioxide on the filtration efficiency of N95, KN95, and surgical facemasks. external iconCai et al. JAMA open network (June 15, 2020).
Key findings:
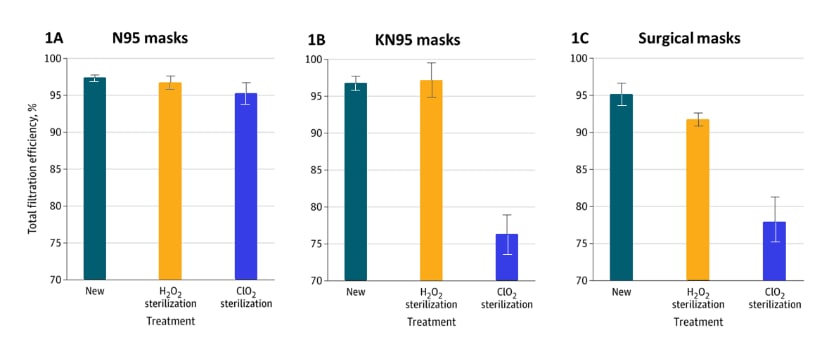
- After hydrogen peroxide (H2O2) facemask sterilization, N95s and KN95s (Chinese N95s) retained 95% filtration efficiency (ability to filter out tiny aerosol particles) (Figures 1A&B).
- H2O2 caused small declines in filtration efficiency of N95, KN95, and surgical masks (Figure 1A–C).
- After chlorine dioxide (ClO2) sterilization, overall filtration efficiency remained >95% with N95s, but declined sharply in KN95s (76%) and surgical masks (78%) (Figure 1A–C).
- N95 efficiency dropped to 86% for 0.3 micron particles.
Methods: Comparison of H2O2 and ClO2 sterilization on facemask filtration efficiencies. Limitations: Small variety of manufacturers; small sample sizes for masks; limited number of conditions and sterilization techniques; only 1 sterilization cycle.
Implications: Sterilization with H2O2 impacted facemask filtration efficiency less than with ClO2. Post-sterilization facemask function may depend on aerosol size.
Figure:
Note: Adapted from Cai et al. Figures 1A–C show the filtration efficiency (ability to filter out aerosol) and 95% CIs of new masks (left bar), masks after H2O2 sterilization (middle bar), and after ClO2 (right bar), for N95 (A), KN95 (B), and surgical masks (C).
Prevalence of COVID-19 infection and subsequent cohorting in a residential substance use treatment program in Boston, MAexternal icon. Barocas et al. Journal of Addiction Medicine (June 9, 2020).
Key findings:
- At a residential substance use treatment facility with shared rooms, 6/22 (27%) of residents and 4/9 (44%) of staff tested positive for SARS-CoV-2.
- After testing, disinfection, and separating residents by infection status (cohorting), no residents in the COVID-19 negative unit developed symptoms during the next 2 weeks.
- Residential addiction treatment continued.
Methods: NP RT-PCR testing of all 22 residents and 9/10 staff at a residential substance use treatment facility, April 2020. Limitations: No antibody testing to ascertain past infection; no follow-up RT-PCR testing to detect new asymptomatic infections.
Implications: Rapid universal testing and cohorting may allow residential facilities to continue operating during the COVID-19 pandemic.
PREPRINTS (NOT PEER-REVIEWED)
Interregional SARS-CoV-2 spread from a single introduction outbreak in a meat-packing plant in northeast Iowa.external icon Richmond et al. medRxiv (June 8, 2020).
Key findings:
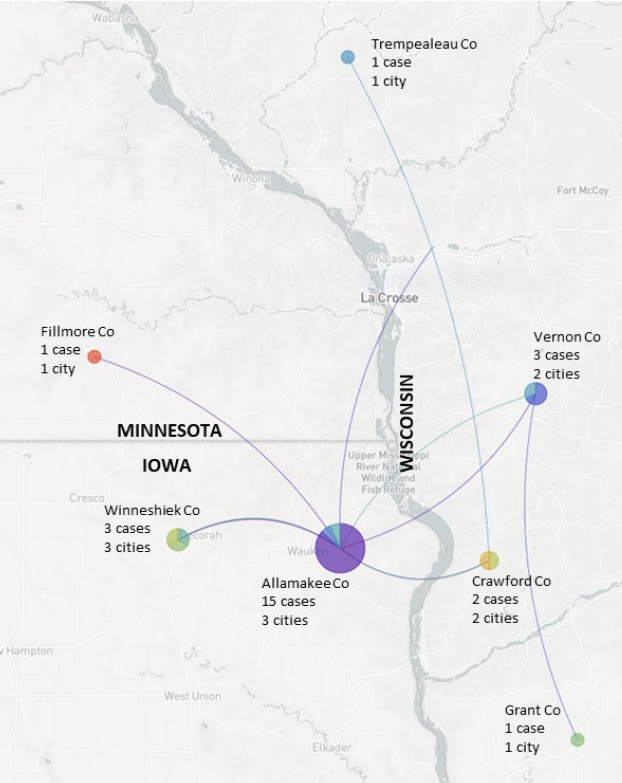
- Phylogenetic analysis suggests SARS-CoV-2 introduction from northeastern US into Postville, Iowa meatpacking plant in early March.
- Virus spread to plant workers and their households within 2 weeks.
- Outbreak spread to individuals in 13 cities in 7 counties in Iowa, Wisconsin and Minnesota (spanning 185 square miles) by mid-May.
Methods: Retrospective next-generation sequencing and phylogenetic analysis of complete viral genomes from NP swabs from 67 COVID-19 patients in Iowa (including from meatpacking plant), Wisconsin, and Minnesota, between March and May 2020. Limitations: Small sample size, spread may be under-estimated.
Implications: Phylogenetic analysis can identify transmission chains and spur public health response if available in real-time. Without rapid action and regional coordination, SARS-CoV-2 outbreaks in meatpacking plants may spread widely.
Figure:
Note: Adapted from Richmond et al. Phylogenetic data suggest that SARS-CoV-2 spread from Pottsville, Allamakee County, Iowa (central purple pie chart) to counties in Minnesota and Wisconsin, April-May, 2020. Pie charts show the distribution of cases from each city within that county: most Allamakee County cases came from one of the 3 involved cities. Thin purple and blue lines show imputed transmission chains. Licensed under CC-BY-NC-ND 4.0.
A large SARS-CoV-2 outbreak aboard the Diamond Princess cruise ship in February 2020 was the largest outbreak outside of China at the time and the first of many outbreaks on cruise ships. After a disembarking passenger tested positive for SARS-CoV-2 in Hong Kong, the ship was quarantined off the Japanese coast. Owing to close confines and older passenger ages, >700 of 3,711 passengers and crew members became infected and were admitted to multiple hospitals in Japan. We feature 3 articles that examined development of symptoms, duration of RT-PCR positivity, and seroconversion among those with SARS-CoV-2 infection.
PEER-REVIEWED
A. Clinical characteristics of COVID-19 in 104 people with SARS-CoV-2 infection on the Diamond Princess cruise ship: a retrospective analysisexternal icon. Tabata et al. Lancet Infectious Disease (June 12, 2020).
Key findings:
- Among 104 persons with SARS-CoV-2, 33 (32%) remained asymptomatic, 43 (41%) developed mild COVID-19, 28 (27%) developed severe COVID-19.
- Of 43 initially asymptomatic persons, 10 (23%) developed symptoms (had been pre-symptomatic) and 3 had severe COVID-19.
- On admission, pre-symptomatic persons more often had evidence of tissue damage (lactate dehydrogenase [LDH] >230 IU/L) than asymptomatic persons (50% vs. 12%, p=0.02).
- Among 41 passengers with initial mild symptoms, 5 (12%) developed severe COVID-19.
Methods: 104 older adult (median age 68) passengers and crew members with SARS-CoV-2 (by RT-PCR) admitted to a Japanese hospital, February 2020, and followed for ~2 weeks. Limitations: Unclear discharge criteria; follow-up did not extend beyond discharge; mean/median LDH levels not presented.
B. SARS-CoV-2 shedding and seroconversion among passengers quarantined after disembarking a cruise ship: a case seriesexternal icon. Hung et al. Lancet Infectious Diseases (June 12, 2020).
Key findings:
- Of 9 participants found to have SARS-CoV-2 infection (by PCR or serology) following enrollment, 6 were asymptomatic.
- 3/6 asymptomatic individuals had lung inflammation by CT; 2/3 symptomatic participants had lung inflammation by CT.
- At baseline;
- 8/9 participants had serum IgG against spike receptor-binding protein (a protein on the surface of the virus); the 9th seroconverted by day 8 of quarantine.
- 4/9 had IgG against nucleoprotein (a structural protein inside the virus); this rose to 7/9 by day 8.
- IgG levels were higher among those with lung inflammation by CT (Figures 1&2).
Methods: Prospective study of 215 individuals who were PCR-negative while aboard the Diamond Princess cruise ship and were repatriated to Hong Kong, February 20–21, 2020. During 14 day-quarantine, specimens collected at baseline and every 4 days for RT-PCR, IgG, and IgM. Limitations: Small sample size.
Figure 1
Figure 2
Note: Adapted from Hung et al. SARS-CoV-2-specific anti-nucleoprotein IgG levels (Figure 1) and SARS-CoV-2-specific anti-spike receptor-binding domain (RBD) IgG levels (Figure 2) (optical density [450 nm]) were higher in participants with evidence of pneumonitis (lung inflammation) by computed tomography (CT) than those without pneumonitis. This article was published in Lancet Infectious Diseases, Vol 20, Hung et al., SARS-CoV-2 shedding and seroconversion among passengers quarantined after disembarking a cruise ship: a case series, P1051-1060, Copyright Elsevier 2020. This article is currently available at the Elsevier COVID-19 resource center: https://www.elsevier.com/connect/coronavirus-information-centerexternal icon.
C. Natural history of asymptomatic SARS-CoV-2 infectionexternal icon. Sakurai et al. NEJM (June 12, 2020).
Key findings:
- Of 90 asymptomatic persons with SARS-CoV-2, nearly half (n=43) tested negative (2 consecutive tests) within 8 days of the first positive PCR test (range 5–21 days).
- Older individuals had higher viral loads and took ~4.4 days longer to turn PCR-negative than younger individuals (95% CI, 2.3–5 days) (Figure).
Methods: 128 persons (RT-PCR-positive asymptomatic persons and RT-PCR-negative cabinmates) were transferred from the Diamond Princess cruise ship to a Japanese hospital for observation, between February 19 and 26, 2020. RT-PCR testing performed every 48 hours. Limitations: Exact time of infection not known; positive RT-PCR test may not indicate patient was infectious.
Figure:
Note: Adapted from Sakurai et al. Older adults (pink and purple lines) exhibited higher viral loads (crossing-point [Cp] y-axis) over a longer time period than younger individuals. From NEJM. Sakurai et al. 383:885-886. DOI: 10.1056/NEJMc2013020. Copyright ©2020 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
Implications for 3 studies (Tabata et al., Hung et al. & Sakurai et al.): Among older persons with SARS-CoV-2, over 1/4 developed severe COVID-19. Even in asymptomatic persons with SARS-CoV-2, lung inflammation may be present and spur more robust immune responses. SARS-CoV-2 infections may resolve more slowly in older asymptomatic adults than in younger asymptomatic adults.
PEER-REVIEWED
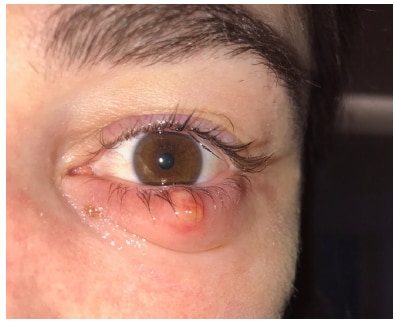
Cluster of chalazia in nurses using eye protection while caring for critically ill patients with COVID-19 in intensive care. external iconMégarbane et al. Journal of Occupational and Environmental Medicine (May 28, 2020).
Key findings:
- Three ICU nurses caring for COVID-19 patients each developed a chalazion, which is a painless inflammatory lump in blocked oil glands of eyelid (Figure).
- The cause was attributed to irritation by chemically-disinfected protective glasses, face masks, and reduced blinking and touching one’s face.
- All chalazia resolved over a month with antibiotic and anti-inflammatory eye ointments and supportive care.
- No further cases after recommendations to improve rinsing of glasses after chemical disinfection.
Methods: Case reports of ICU nurses, Paris, France. Limitations: No historical data on chalazion frequency.
Implications: Chalazia might occur among healthcare workers wearing PPE for extended periods, and may indicate a need for changes in how eyewear is disinfected, rinsed, and worn.
Figure:
Note: Adapted from Mégarbane et al. Chalazion of the lower eyelid in a nurse. The nurse was wearing protective glasses disinfected with 0.5% didecyldimethylammonium chloride and chlorhexidine digluconate. Available for use in accordance with BMJ website terms and conditions for the duration of the COVID-19 pandemic or until otherwise determined by BMJ.
PEER-REVIEWED
Airborne SARS-CoV-2 is rapidly inactivated by simulated sunlightexternal icon. Schuit et al. Journal of Infectious Disease (June 11, 2020).
Key findings:
- 90% of aerosolized SARS-CoV-2 was inactivated within 6 minutes of exposure to simulated high-intensity summer sunlight.
- 90% inactivation took 19 minutes in simulated mid-intensity sunlight (early fall/late winter conditions) and >2 hours in darkness.
- Relative humidity did not affect inactivation times.
Methods: Aerosol particles (~2 microns) of SARS-CoV-2 suspended in artificial saliva were exposed to simulated sunlight at different intensities mimicking summer or early fall/late winter in the middle of the US. Viral titers determined via infection assays using Vero-6 cells. Limitations: The role of aerosol-based SARS-CoV-2 transmission not fully established.
Implications: Natural sunlight may be an effective disinfectant for SARS-CoV-2 in aerosols and on surfaces (as previously demonstrated, Ratnesar-Schumate et alexternal icon., see May 29, 2020 Edition of the Science Update). Outdoor gatherings may be less risky than indoor gatherings. Further studies are needed to determine SARS-CoV-2 infectious dose and role of aerosols in spreading SARS-CoV-2.
- Rubino et al. New-onset diabetes in COVID-19external icon. NEJM. Explores the interactions of COVID-19 and diabetes mellitus and describes a global registry to track epidemiologic features and pathogenesis of new-onset diabetes with COVID-19.
- Scully et al. Considering how biological sex impacts immune responses and COVID-19 outcomesexternal icon. Nature Reviews Immunology. Thorough review of biological sex, immune responses, and differential COVID-19 outcomes by biological sex.
- Sneed et al. Social and psychological consequences of the COVID-19 pandemic in African-American communities: Lessons from Michiganexternal icon. Psychological Trauma: Theory, Research, Practice, and Policy. Authors explore experiences with COVID-19 by communities of color and explore community-based approaches to mitigate psychological consequences.
- Galea et al. COVID-19 pandemic, unemployment, and civil unrest. Underlying deep racial and socioeconomic divides.external icon JAMA. Discusses intersection of COVID-19, economic upheaval, and racial injustice.
- Evans et al. Diagnosing and treating systemic racismexternal icon. NEJM. Explores the need to address systemic racism through changes in the healthcare system.
- Mahase. COVID-19: Urgent cancer referrals fall by 60%, showing “brutal” impact of pandemicexternal icon. BMC. During the COVID-19 pandemic, urgent referrals for cancer care dropped dramatically in England. Delayed cancer diagnoses and treatment are anticipated.
- Oreshkova et al. SARS-CoV-2 infection in farmed minks, the Netherlands, April and May 2020external icon. Eurosurveillance. Investigation of SARS-CoV-2 among minks on farms in the Netherlands.
- Chaumont et al. Mixed central and peripheral nervous system disorders in severe SARS-CoV-2 infectionexternal icon. Journal of Neurology. Case reports of 4 patients with severe central and peripheral nervous system disorders as late complications of severe COVID-19 following extubation.
- Ogburn et al. Aggregating data from COVID-19 trialsexternal icon. Science. Discusses a platform for sharing RCT protocols, which may facilitate efficient collaborative research.
- Falkenhain-López et al. COVID-19-related acute genital ulcersexternal icon. Journal of European Academy of Dermatology and Venereology. Case report of acute genital ulceration attributed to immune response to SARS-CoV-2. Work-up insufficient to attribute to SARS-CoV-2 with confidence.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.

![Figure 1 SARS-CoV-2-specific anti-nucleoprotein IgG levels (optical density [450 nm]) were higher in participants with evidence of pneumonitis (lung inflammation) by computed tomography (CT) than those without pneumonitis.](/library/covid19/images/0623_Figure_4a.jpg?_=62359)
![Figure SARS-CoV-2-specific anti-spike receptor-binding domain (RBD) IgG levels (Figure 2) (optical density [450 nm]) were higher in participants with evidence of pneumonitis (lung inflammation) by computed tomography (CT) than those without pneumonitis.](/library/covid19/images/0623_Figure_4b.jpg?_=62360)
![Figure Older adults (pink and purple lines) exhibit higher viral loads (crossing-point [Cp] y-axis) over a longer time period than younger individuals.](/library/covid19/images/0623_Figure_5.jpg?_=62361)