COVID-19 Science Update released: June 4, 2021 Edition 92

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Section headings in the COVID-19 Science Update align with the CDC Science Agenda for COVID-19.
Section headings in the COVID-19 Science Update have been changed to align
with the CDC Science Agenda for COVID-19.
Here you can find all previous COVID-19 Science Updates.
PEER-REVIEWED
Safety, immunogenicity, and efficacy of the BNT162b2 COVID-19 vaccine in adolescents.external icon Frenck et al. NEJM (May 27, 2021).
Key findings:
- Vaccine efficacy was 100% (95% CI 75.3%-100%) in 12- to 15-year-olds.
- There were no cases in the vaccinated group compared with 16 cases among the placebo group, 7 or more days after dose 2.
- Compared with baseline, geometric mean neutralizing antibody titers were 118.3-fold higher 1 month after dose 2.
- Vaccine reactions were mainly transient, mild to moderate, and similar to a comparator group of 16–25-year-olds.
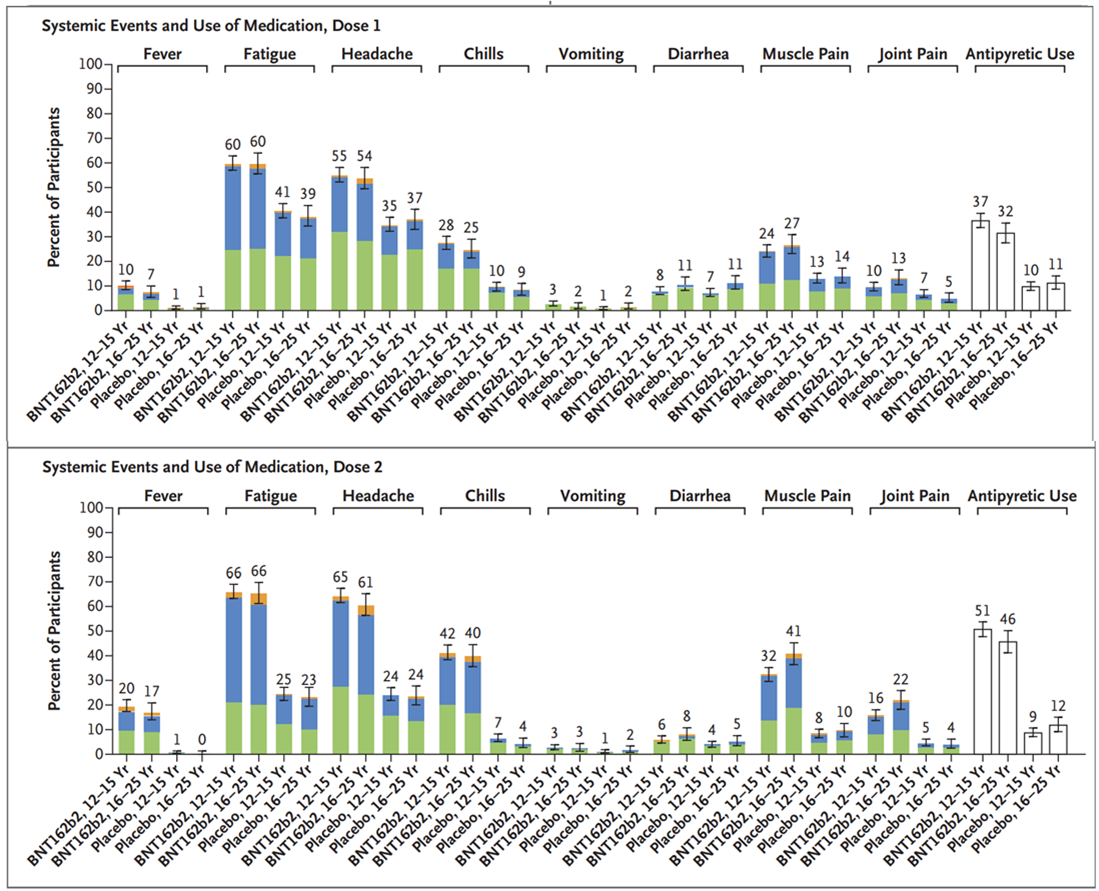
- Injection-site pain was reported by 79% to 86%, fatigue was reported by 60% to 66%, and headache was reported by 55% to 65% of participants (Figure).
Methods: A randomized, placebo-controlled, observer-blinded trial of Pfizer/BioNTech BNT162b2 in 2,260 adolescents 12–15 years old (1,129 received placebo). Efficacy of the vaccine was assessed based on confirmed SARS-CoV-2 infection with onset 7 or more days after dose 2. Reactogenicity events (assessed for 7 days after each dose) and unsolicited adverse events compared with 16–25 age group (n = 3,610). SARS-CoV-2 serum neutralization assays were performed. Limitations: Racial and ethnic diversity of participants 12-15 years does not reflect the general US population; short (1 month) post-vaccination safety evaluation.
Implications: Vaccination of adolescents with BNT162b2 was safe and effective. Vaccinating adolescents will broaden community protection, and it will likely facilitate reintegration into society and resumption of in-person learning.
Figure:
Note: Adapted from Frenck et al. Systemic events reported within 7 days after receiving dose 1 (top) or dose 2 (bottom) of vaccine or placebo. 1 participant in the 12-to-15-year-old group had a fever with a temperature >40°C after dose 1. From the New England Journal of Medicine, Frenck et al., Safety, immunogenicity, and efficacy of the BNT162b2 COVID-19 vaccine in adolescents. May 27, 2021, online ahead of print. Copyright © 2021 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
Occurrence of severe COVID-19 in vaccinated transplant patientsexternal icon. Caillard et al. Kidney International. (May 21, 2021).
Key findings:
- 55 solid organ transplant recipients developed COVID-19 after receiving 2 doses of mRNA vaccine.
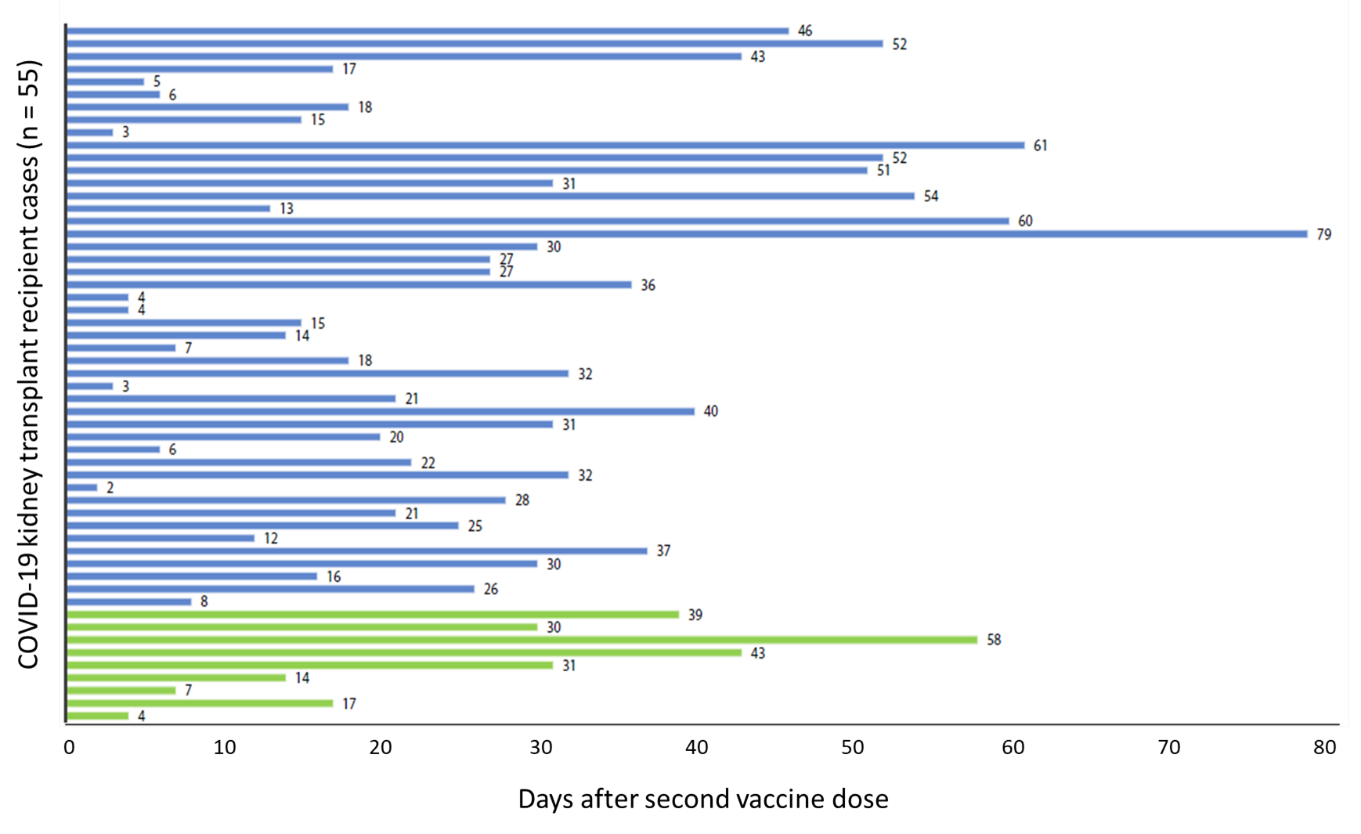
- Symptoms began a median of 22 days after the second vaccine dose (Figure).
- 15 cases required hospitalization; of these, 6 were admitted to an intensive care unit, and 3 died.
- Of 25 patients with post-vaccination serology, 24 were antibody negative; 1 was antibody positive but had low titers.
Methods: Case series of 55 solid organ transplant recipients (52 kidney and 3 kidney-pancreas) who developed COVID-19 after receiving 2 doses of SARS-CoV-2 mRNA vaccines. Each case received the second dose of vaccine between February 8 and April 22, 2021. Limitations: Total number of solid organ transplant recipients (i.e., denominator) is unclear.
Implications: Vaccinated transplant patients should consider continuing non-pharmaceutical preventive measures such as wearing a face mask and maintaining social distance. The potential value of post-vaccination serology to assess immune responses in vulnerable groups should be further evaluated.
Figure:
Note: Adapted from Caillard et al. Time in days from the second vaccine dose of Pfizer BNT162b2 and Moderna mRNA-1273 vaccine to the onset of COVID-19 symptoms in each transplant recipient. Reprinted from Kidney International, online May 21, 2021, Caillard et al., Occurrence of severe COVID-19 in vaccinated transplant patients. Copyright 2021, with permission from Elsevier.
Prevalence of clinical and subclinical myocarditis in competitive athletes with recent SARS-CoV-2 infection: Results from the Big Ten COVID-19 cardiac registryexternal icon. Daniels et al. JAMA Cardiology (May 27, 2021).
Key findings:
- Among 1,597 college athletes who had cardiac magnetic resonance (CMR) imaging after SARS-CoV-2 infection, 37 (2.3%, 95% CI 1.6%-3.2%) were diagnosed with clinical (9) or subclinical (28) myocarditis.
- Based on cardiac symptoms alone, only 5 athletes would have would have been screened and diagnosed with myocarditis.
- The prevalence of myocarditis per university ranged from 0% to 7.6% with 3 universities reporting 0 cases.
Methods: Observational study of SARS-CoV-2 RT-PCR positive athletes from 13 of 14 Big Ten universities between March 1 and December 15, 2020. Cardiac symptoms and details about cardiac testing were reported for athletes with myocarditis. Myocarditis was categorized as either clinical or subclinical based on symptoms and CMR findings. Limitations: Prior to September 2020, not all athletes who tested positive for SARS-CoV-2 infection underwent CMR imaging, potentially affecting prevalence estimates; myocarditis prevalence may not be representative of general population.
Implications: Comprehensive testing with CMR imaging identified many more athletes with myocarditis following SARS-CoV-2 infection than would have been identified by symptoms alone or by less intensive screening. An accompanying editorial by Udelson et al. outlines challenges in widespread use of CMR screening and suggests “the more practical and more widely available approach of testing with ECG, echocardiography, and serum troponin likely improves specificity and decreases burden of potentially unwarranted athletic restriction.”
Same-day SARS-CoV-2 antigen test screening in an indoor mass-gathering live music event: a randomized controlled trial.external icon Revollo et al. Lancet Infectious Diseases (May 27, 2021).
Key findings:
- Of 980 adult volunteers randomly assigned to experimental and control groups with negative rapid antigen tests (Ag-RDT) prior to a 5-hour indoor concert:
- None of the volunteers in the experimental group who wore N-95 masks but were otherwise unrestricted and attended the concert, tested positive for SARS-CoV-2 by RT-PCR on day 8 (estimated incidence 0.14% [95% CI 0%-0.61%]) .
- 2 of the volunteers in the control group, 0.31%, (95% CI 0.04%-0.73%), who were screened at the same time and did not attend the concert tested positive for SARS-CoV-2 on day 8.
Methods: Randomized controlled trial (ages 18–58 years) to assess the effectiveness of a comprehensive preventive intervention (same-day screening with Ag-RDTs, required N-95 face masks, and adequate air ventilation) at an indoor concert held December 12, 2020, in Barcelona, Spain. The primary outcome was 7-day cumulative incidence of RT-PCR-confirmed SARS-CoV-2 infection in the control (n = 465) and intervention (n = 465) groups. Limitations: Participants knew they were participating in a clinical trial.
Implications: Although less accurate than NAAT, the Ag-RDT, combined with masking and ventilation, reduced risk at a large indoor gathering. Indoor mass gatherings might be safe with appropriate efforts to pre-screen participants, compliance with masking, and adherence to air ventilation protocols.
PREPRINTS (NOT PEER-REVIEWED)
Variants of concern are overrepresented among post-vaccination breakthrough infections of SARS-CoV-2 in Washington Stateexternal icon. McEwen et al. medRxiv (May 25, 2021). Published in Clinical Infectious Diseasesexternal icon (June 24, 2021).
Key findings:
- Of the 20 SARS-CoV-2 breakthrough infections following full vaccination, all were caused by variants.
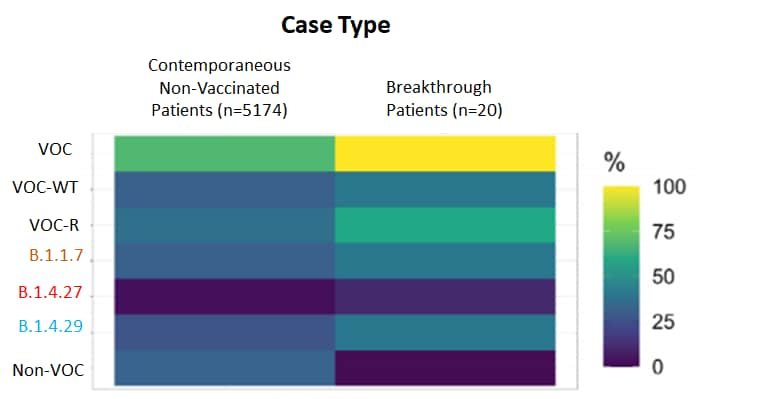
- The frequency of variants in breakthrough cases was 1.47-fold higher than controls [95% CI 1.45-1.50, p = 0.001], for which only 68% of cases were caused by these variants (Figure).
- The 20 variant breakthrough cases were classified as: 8 (40%) B.1.1.7, 1 (5%) B.1.351, 2 (10%) B.1.427, 8 (40%) B.1.429, and 1 (5%) P.1.
Methods: 20 SARS-CoV-2 breakthrough infections detected at the University of Washington between February 23 and April 27, 2021 were sequenced and compared to 5,174 contemporaneous control non-vaccinated patients to identify the prevalence of variants among breakthrough cases. Breakthrough cases were defined as patients who were fully vaccinated against SARS-CoV-2 (>2 weeks post-2nd dose of mRNA vaccine) who subsequently tested positive by RT-PCR. Limitations: Small number of subjects in the vaccine breakthrough group; might have missed breakthrough infections in the community.
Implications: Continued surveillance of post-vaccine breakthrough cases might help refine development of vaccine products and strategies.
Figure:
Note: Adapted from McEwen et al. Heat map showing relative frequencies of variants (B.1.427, B.1.429, and B.1.1.7) in breakthrough vs. control cases. VOC = variant of concern; WT = wild type; VOC-R = variants of concern with reduced neutralization. Licensed under CC BY 4.0.
Effectiveness of COVID-19 vaccines against the B.1.617.2 variantexternal icon. Bernal et al. medRxiv (May 24, 2021). Published in NEJM as Effectiveness of COVID-19 vaccines against the B.1.617.2 (Delta) variantexternal icon (July 21, 2021).
Key findings:
- Following 2 doses of Pfizer/BioNTech BNT162b2, effectiveness against B.1.1.7 was 93.4% (95% CI 90.4%-95.5%) compared to 87.9% (95% CI 78.2%-93.2%) against B.1.617.2.
- Following 2 doses of Oxford/AstraZeneca ChAdOx1, effectiveness against B.1.1.7 was 66.1% (95% CI 54.0%-75.0%) compared to 59.8% (95% CI 28.9%-77.3%) against B.1.617.2.
- After 1 dose, both vaccines were significantly less effective against B.1.617.2 (33.5%, 95% CI 20.0%-44.3%) compared with B.1.1.7 (51.1%, 95% CI 47.3%-54.7%).
Methods: Linked data (n = 12,675) on symptomatic sequenced cases of COVID-19 and England’s National Immunization Management System for vaccinations up to May 16, 2021 were used to estimate the proportion of cases with B.1.617.2, based on S gene, compared to the predominant strain (B.1.1.7) by vaccination status. Limitations: Does not account for differences in populations that may have received each vaccine; timing of rollout for different vaccines varied.
Implications: Vaccine effectiveness against B.1.617.2 after 1 dose was only 33.5%, suggesting a need to maximize uptake of 2nd doses of these vaccines.
PEER-REVIEWED
Face masks effectively limit the probability of SARS-CoV-2 transmissionexternal icon. Cheng et al. Science (May 20, 2021).
Key findings:
- The mean number of SARS-CoV-2 particles exhaled over 30 minutes ranged from 1 to 600.
- The number of viral particles was low compared to the overall number of respiratory particles and varied across settings.
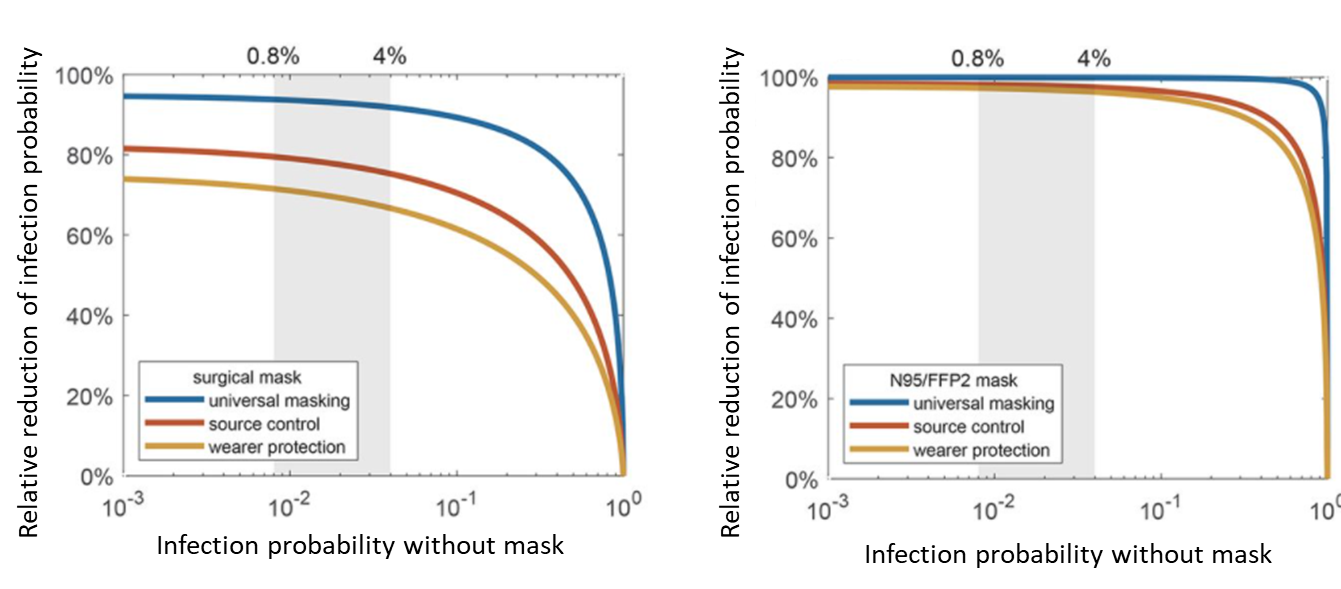
- Universal masking was more effective at reducing airborne transmission compared with masking by infectious persons (source control) or susceptible persons (wearer protection) only.
- N95 and equivalent masks were more effective than surgical masks (Figure 1).
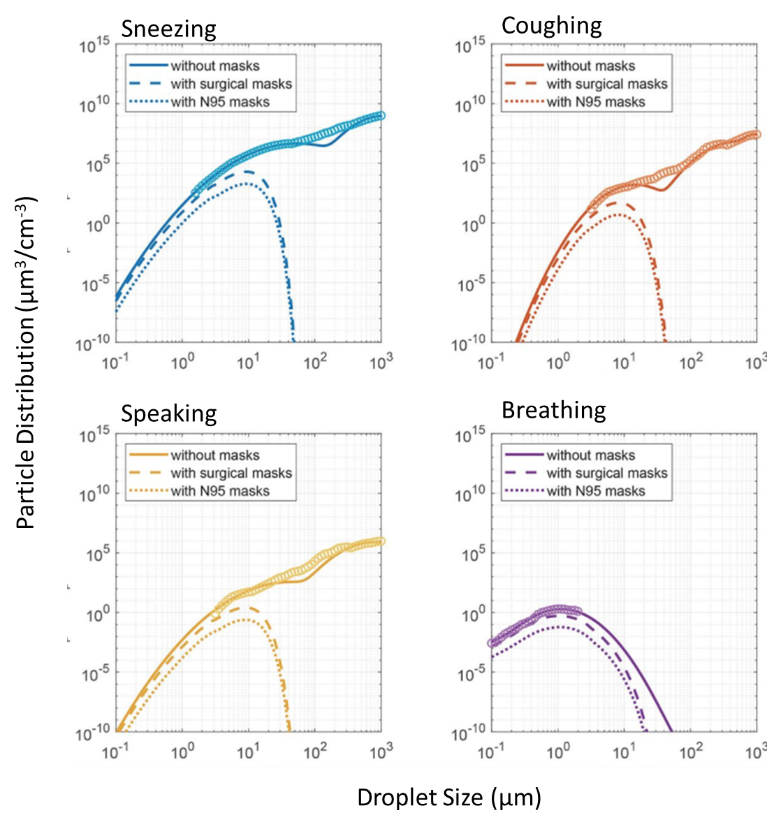
- Masks were more effective at removing larger particles that are more likely to be generated and dispersed during coughing or sneezing than smaller particles from speaking or breathing (Figure 2).
Methods: Published data from medical centers in China, Singapore, and the US were used to model the mean number of SARS-CoV-2 particles exhaled. The probability of infection as a function of the number of inhaled virus particles was used to evaluate the effectiveness of masks against respiratory particles and droplets with diameters <100 μm. Limitations: Did not model cloth or polyester mask effectiveness.
Implications: Because few respiratory particles contain viruses and most environments do not have abundant virus, wearing masks with limited filtration efficiency (30%–70%) can limit the number of inhaled virus particles and keep the probably of infection low in most settings. In settings with higher respiratory particle concentrations (e.g., some healthcare settings), higher filtration masks such as N95 might be needed.
Figure 1:
Note: Adapted from Cheng et al. Relative reduction in proportion infected if wearing a surgical mask (left) or N95/FFP2 mask (right) with universal masking, source (infectious person) masking, or wearer (susceptible person) masking. Shaded areas indicate the level of basic population-average infection probability, corresponding to the basic reproduction number for SARS-CoV-2. Licensed under CC BY 4.0.
Figure 2:
Note: Adapted from Cheng et al. Volume distribution (y-axis) of respiratory particles of different sizes (x-axis) emitted during different respiratory activities without masks (solid line) and if wearing a surgical mask (dashed line) or N95 mask (dotted line) from sneezing, coughing, speaking, or breathing. The open circles (chain-link line) are measurement data obtained without masks, and the solid lines are bi- or trimodal fits to the measurement data.
Immune response to SARS-CoV-2 variants of concern in vaccinated individualsexternal icon. Becker et al. Nature Communications (May 25, 2021).
Key findings:
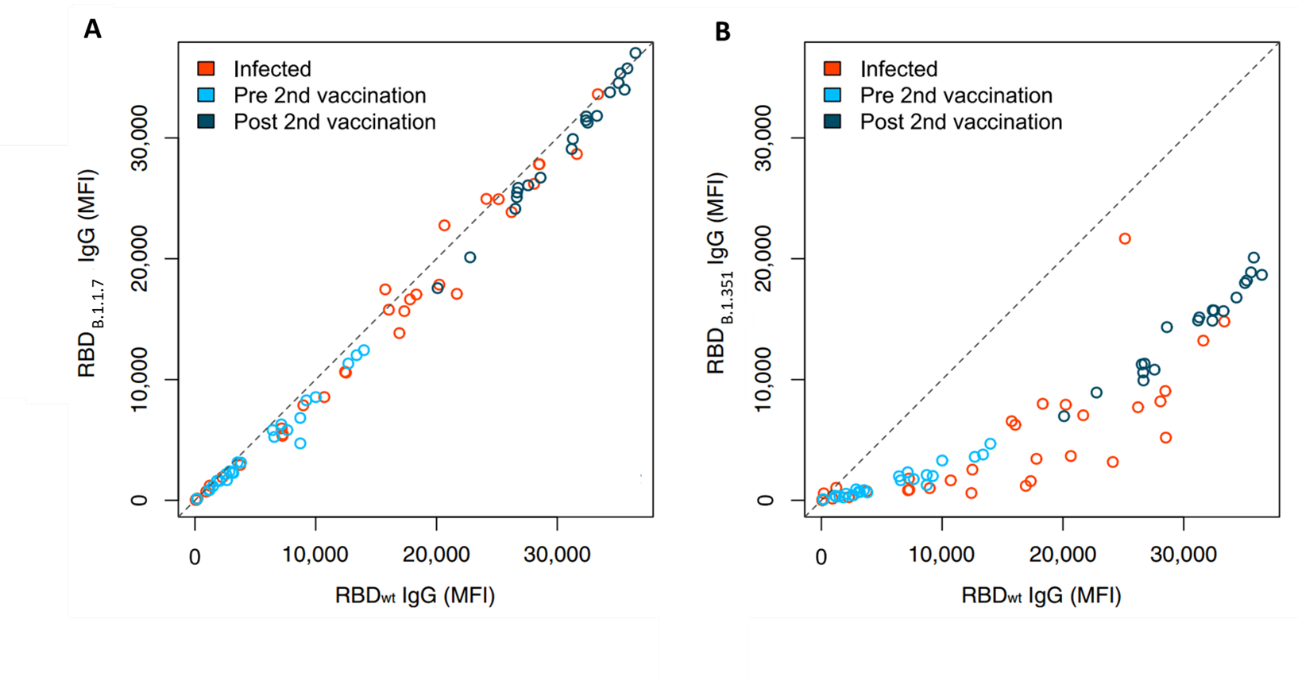
- Sera from partially and fully vaccinated persons had lower antibody responses to the receptor binding domain (RBD) from the B.1.351 variant compared with RBD from the B.1.1.7 variant (Figure).
- Sera from fully vaccinated individuals had normal antibody responses to RBD from the Cluster 5 and B.1.429 variants.
- Fully vaccinated individuals had high IgG titers in saliva.
Methods: Sera and saliva antibody responses to 4 variants (B.1.1.7, B.1.351, Cluster 5, and B.1.429) were examined in infected (n = 35) and uninfected (n = 20) individuals and those vaccinated with Pfizer/BioNTech BNT162b2 (n = 25). Levels of IgG antibody to RBD and neutralization of RBD were determined an average of up to 17 days after vaccination. Limitations: Small sample size and limited follow-up time; saliva not tested against variants.
Implications: Although Pfizer/BioNTech BNT162b2 induces a robust antibody response to 3 circulating SARS-CoV-2 variants, the response to B.1.351 is reduced. High antibody titers in saliva suggest that vaccinated individuals might have reduced transmission potential.
Figure:
Note: Adapted from Becker et al. Receptor binding domain (RBD)-binding antibody responses against B.1.1.7 (A) and B.1.351 (B) from infected, vaccinated pre-second dose and post second dose individuals, compared to the wildtype (wt) RBD. The dashed gray line indicates an identical response between wild-type and variant of concern. MFI = median fluorescence intensity, a measure of the amount of antibody in a sample. Licensed under CC-BY 4.0
PREPRINTS (NOT PEER-REVIEWED)
Antibody responses after first and second COVID-19 vaccination in patients with Chronic Lymphocytic Leukaemiaexternal icon. Parry et al. SSRN (May 25, 2021). . Published in Blood Cancer Journalexternal icon (July 30, 2021).
Key findings:
- Among B-cell chronic lymphocytic leukemia (CLL) patients vaccinated for COVID-19:
- 34% were seropositive after 1 vaccine dose compared with 94% of controls.
- 75% were seropositive after 2 vaccine doses compared with 100% of controls.
- Antibody titers were approximately 104-fold lower compared with controls.
- CLL patients receiving Bruton tyrosine kinase inhibitor (BTKi) treatment and those with IgA deficiency were least likely to develop antibody responses following vaccination.
Methods: SARS-CoV-2 spike-specific antibody responses were examined in CLL patients following vaccination with either Pfizer/BioNTech BNT162b2 (n = 154) or Oxford/AstraZeneca ChAdOx1 (n = 145) and compared with healthy donors (n = 93). Patients were vaccinated on either a 3-week or extended 10–12-week interval. Antibody response was determined 2–5 weeks after vaccination. Limitations: Most samples obtained before the second vaccine dose and inability to test for previous infection in many samples.
Implications: Vaccinated patients with CLL, particularly those on BTKi therapy or with IgA deficiency, produce reduced spike-specific antibody titers, which might represent reduced vaccine-induced immunity to SARS-CoV-2.
PEER-REVIEWED
Factors associated with racial/ethnic group–based medical mistrust and perspectives on COVID-19 vaccine trial participation and vaccine uptake in the USexternal icon. Thompson et al. JAMA Network Open (May 27, 2021).
Key findings:
- 75% of participants reported low willingness to participate in vaccine trials, and 52% reported low willingness to be vaccinated (Figure).
- Black adults were less willing to participate in COVID-19 vaccine trials and to be vaccinated compared with other racial/ethnic groups (Figure).
- Black adults reported greater medical mistrust (mean [SD], 2.35 [0.96]) compared with other racial/ethnic groups (mean [SD] for the total sample, 1.83 [0.91]).
- Experiences of racism were cited as a leading reason for medical mistrust.
Methods: A cross-sectional survey of 1,835 adults in Michigan conducted between June and December 2020 assessed whether respondents, if asked, would participate in a research study to test a COVID-19 vaccine or receive a COVID-19 vaccine. Participants also completed the suspicion subscale of the Group-Based Medical Mistrust Scale using a Likert-type scale ranging from 1 (strongly disagree) to 5 (strongly agree), with higher scores representing greater mistrust. Limitation: Data not representative of the Michigan population.
Implications: Socially and culturally responsive strategies that acknowledge and address racial/ethnic health care disparities and historical and contemporary experiences of racism are needed to promote COVID-19 vaccination, particularly among individuals identifying as Black.
Figure:
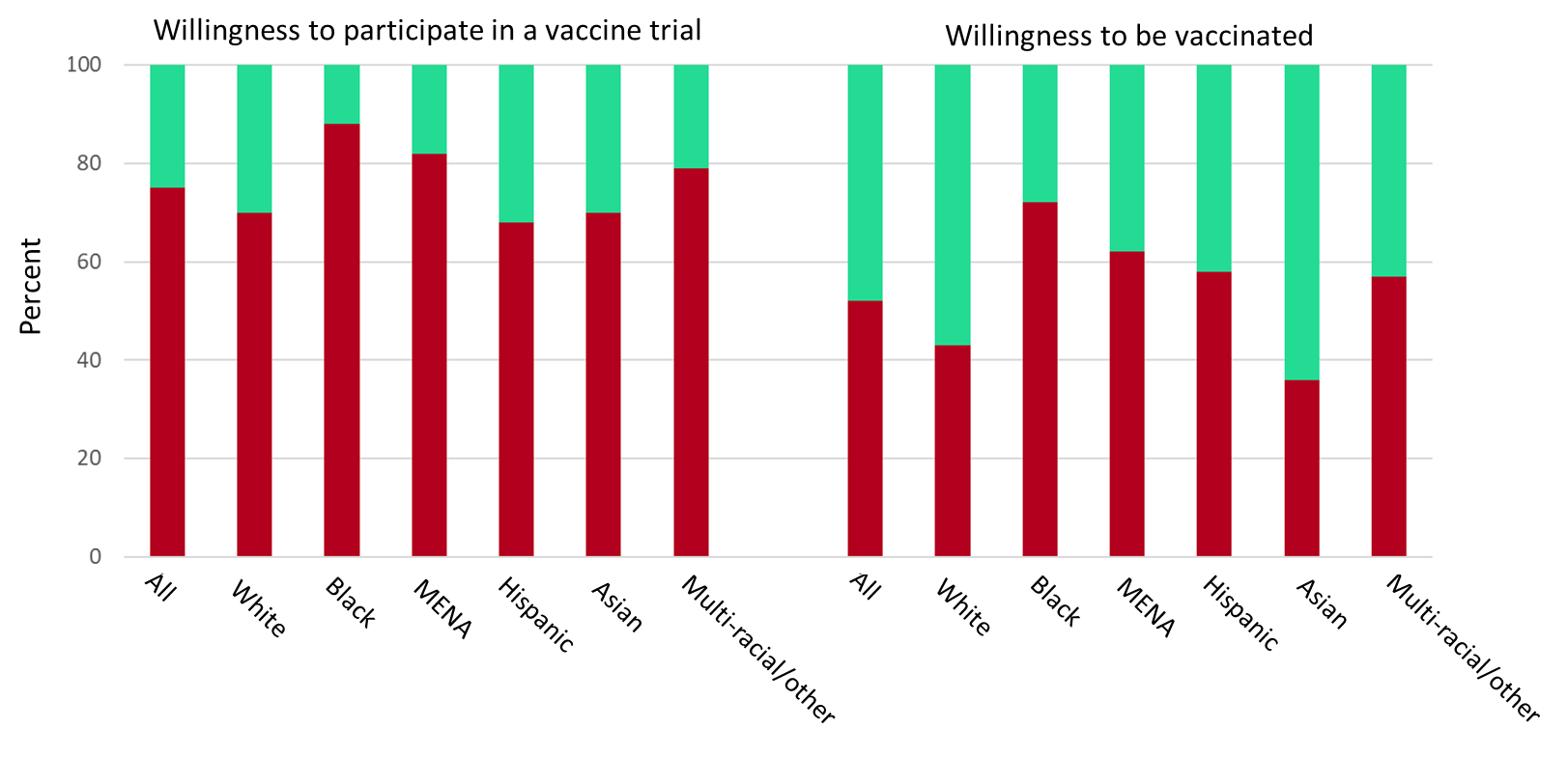
Note: Adapted from Thompson et al, Table 3. Percentage of respondents answering that they would definitely not, probably not or be unsure, or definitely will or probably will participate in COVID-19 vaccine trials (left) or be vaccinated (right) by race/ethnicity. MENA: Arab, Chaldean, Middle Eastern, or North African. Licensed under CC BY.
Detection, Burden, and Impact
- Ngiam et al. Acute coronavirus disease (COVID-19) in vaccinated frontline healthcare workers in India: An observational cohort studyexternal icon. SSRN (Preprint; May 26, 2021). 14.6% of 2,160 vaccinated healthcare workers had a positive RT-PCR test for SARS-CoV-2 2 weeks after first or both vaccine doses; 84.2% of infections were in persons receiving 2 doses (98.4% received Covishield vaccine, a version of the Oxford/AstraZeneca ChAdOx1 vaccine manufactured by the Serum Institute of India). Infections in persons with 2 doses were more likely to be asymptomatic compared with 1 dose, but outcomes were similar between groups.
- Magalis et al. SARS-CoV-2 infection of BNT162b2(mRNA)-vaccinated individuals is not restricted to variants of concern or high-risk exposure environments.external icon medRxiv (Preprint; May 30, 2021). 10 SARS-CoV-2 infections were identified in fully vaccinated persons (Pfizer/BioNTech BNT162b2) between February and March 2021 in Alachua Co, FL where 59,000 persons were at least partially vaccinated by March 17th. 8 of these were <35 years old and worked in the healthcare field, but 7 had non-workplace contacts that were the likely source.
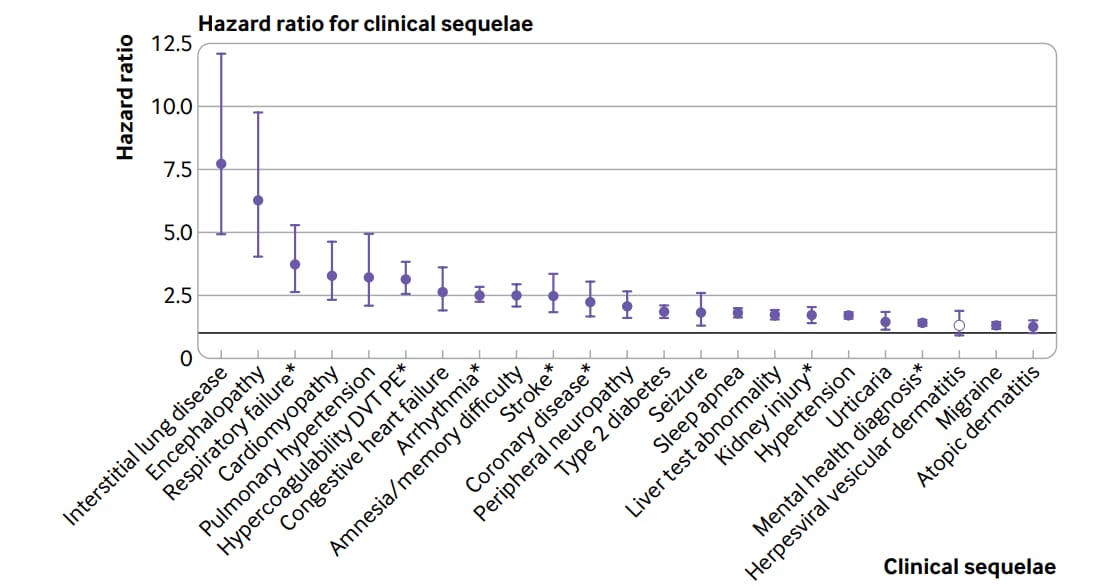
- Daugherty et al. Risk of clinical sequelae after the acute phase of SARS-CoV-2 infection: retrospective cohort studyexternal icon. BMJ (May 19, 2021). Administrative claims, outpatient laboratory results and hospital admissions data through October 31, 2020 showed that 14% (27,074/193,113) of individuals ≤65 years with SARS-CoV-2 infection developed a new type of clinical sequelae that required medical care 4 months after the acute phase of COVID-19, 4.95% higher than a 2020 pre-pandemic comparator group.
Note: Adapted from Daugherty et al. Hazard ratios and 95% CIs for the most common clinical sequelae after SARS-CoV-2 infection versus the 2020 comparator group. DVT = deep vein thrombosis, PE = pulmonary embolism. Licensed under CC-BY-NC 4.0.
Natural History of SARS-CoV-2 Infection
- Singh et al. SARS-CoV-2 variants of concern are emerging in India.external icon Nature Medicine (May 27, 2021). Since March 2021, COVID-19 cases in India have surged from 53 to >200 per million population; this surge has been associated with a succession of variant strains, including B.1.1.7, B.1.351, and B.1.1.28.1. There is now a steep rise in cases caused by the variant B.1.617.2, that has 3 key S-protein mutations (L452R, E484Q, P681R).
Prevention, Mitigation, and Intervention Strategies
- Sahin et al. BNT162b2 vaccine induces neutralizing antibodies and poly-specific T cells in humans.external icon Nature (May 27, 2021). Sera from fully vaccinated participants in a Phase 1/2 trial of Pfizer/BioNTech BNT162b2 neutralized 22 SARS-CoV-2 pseudoviruses with single mutations in the spike protein representing variants B.1.351, B.1.1.7, P.1, and B.1.1.298. Spike-specific T cell responses were detected in 37/37 (for CD4+ T cells) and 34/37 (for CD8+ T cells) fully vaccinated participants.
- Turner et al. SARS-CoV-2 infection induces long-lived bone marrow plasma cells in humans.external icon Nature (May 24, 2021). Spike protein-specific bone marrow plasma cells, the source of long-lived antibodies, were detected from bone marrow aspirates of 15 of 19 persons evaluated 7 and 11 months after mild SARS-CoV-2 infection but not from 11 healthy controls with no history of SARS-CoV-2 infection.
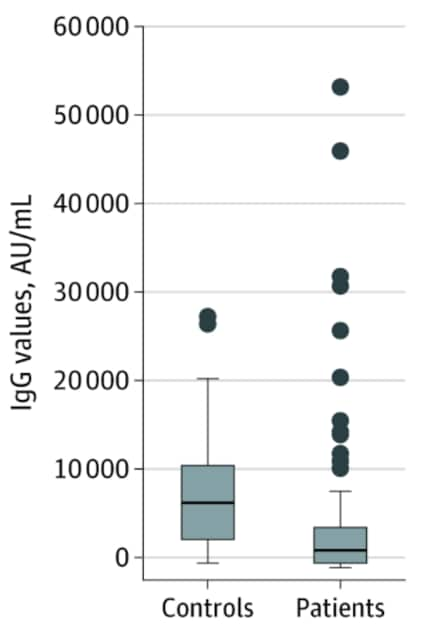
- Massarweh et al. Evaluation of seropositivity following BNT162b2 messenger RNA vaccination for SARS-CoV-2 in patients undergoing treatment for cancer.external icon JAMA Oncology (May 28, 2021). 13 to 54 days following dose 2 of Pfizer/BioNTech BNT162b2, 90% of 102 adult solid tumor patients in Israel and 100% of 78 controls developed IgG antibodies against spike protein. IgG levels were lower in the patient group compared with the control group.
Note: Adapted from Massarweh et al. IgG antibody levels against spike protein following Pfizer/BioNTech vaccination. AU = absorbance units. Licensed under CC BY.
- Harrington et al. Single dose of BNT162b2 mRNA vaccine against SARS-CoV-2 induces high frequency of neutralizing antibody and polyfunctional T-cell responses in patients with myeloproliferative neoplasms.external icon Leukemia (May 22, 2021). Of 21 patients with myeloproliferative neoplasms given a single dose of the Pfizer/BioNTech BTN162b2 vaccine, 17 had spike-specific IgG (4 of whom had spike-specific IgG prior to vaccination) and 18 had neutralizing antibody titers. 15/20 patients had SARS-CoV-2-specific CD4+ T cells and 7/20 had SARS-CoV-2-specific CD8+ T cell responses.
Social, Behavioral, and Communication Science
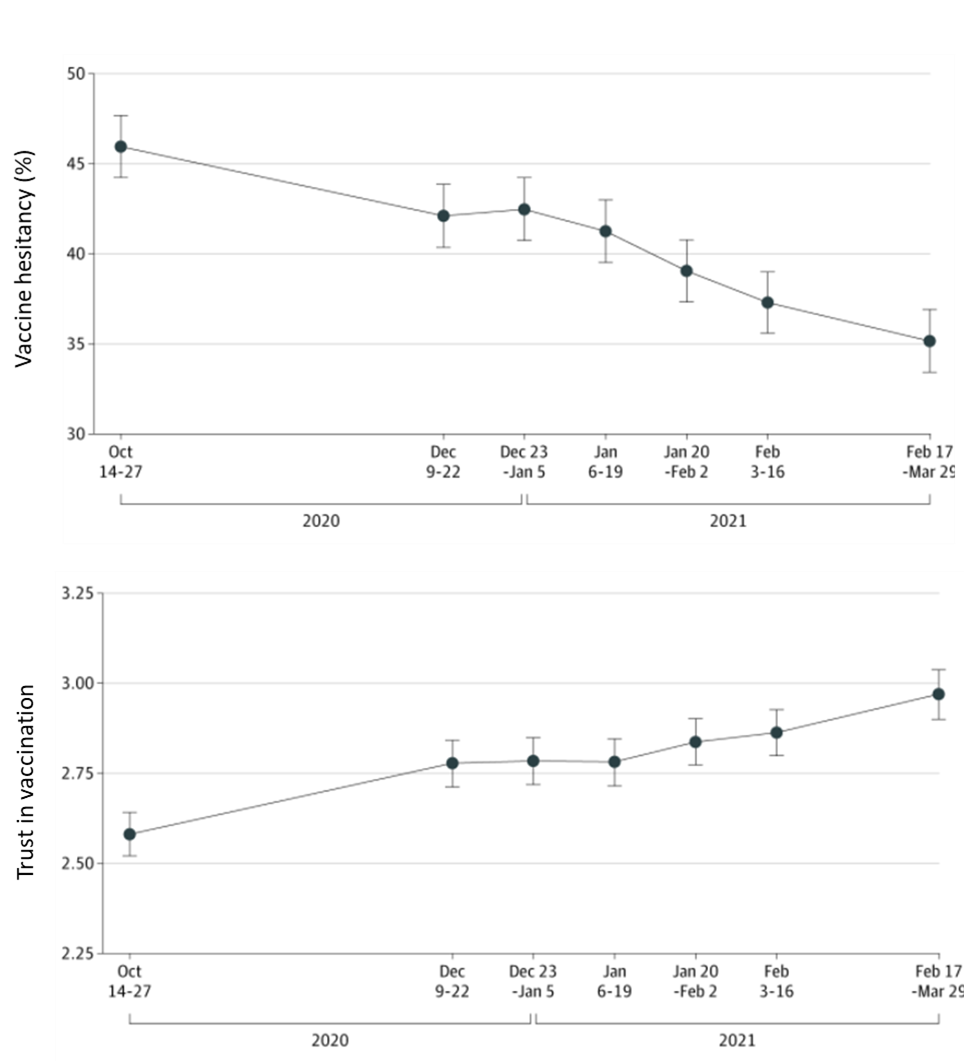
- Daly et al. Public trust and willingness to vaccinate against COVID-19 in the US from October 14, 2020, to March 29, 2021.external icon JAMA (May 24, 2021). Based on a sample of 7,420 participants in the internet panel Understanding America Study, vaccine hesitancy among US adults declined from 46% in October 2020 to 35.2% in March 2021. Hesitancy remained high among adults aged 18–39 years (44.1%), those without a college degree (42.9%), and households earning <$50 000 or less (43.7%).
Note: adapted from Daly et al. Top: percentage of people reporting hesitancy to receive a COVID-19 vaccine with 95% CIs. Bottom: Trust in COVID-19 vaccines using scale of 0 (do not trust development/approval processes) to 6 (fully trust processes). Reproduced with permission from JAMA, 2021. Published online May 24, 2021. https://doi.org/10.1001/jama.2021.8246. Copyright© 2021 American Medical Association. All rights reserved.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.