Methods
Recommendations for Prevention and Control of Infections in NICU Patients: S. aureus (2020)
This guideline is based on a targeted systematic review of the best available evidence on the prevention and control of infections in NICUs.
a. Development of Key Questions
In order to inform the development of the Staphylococcus aureus (S. aureus) Key Questions, electronic searches were conducted to retrieve existing relevant guidelines and to identify gaps and areas where new evidence may have been published. The strategies used for the guideline searches and results can be found in the Appendixpdf icon[PDF – 142 pages]. (Appendix, Section 1.) The results of this initial review informed the development of a preliminary list of Key Questions. Clinical experts were surveyed at national infectious disease society meetings to provide feedback on potential Key Questions and to identify additional topics of interest. Key Questions were finalized after vetting them with HICPAC.
b. Literature Search
A list of search terms was developed to identify the literature most relevant to the Key Questions. The terms were incorporated into search strategies, and these searches were performed in MEDLINE, EMBASE, CINAHL, and the Cochrane Library.
At the outset of this effort in 2011, the literature base was deemed insufficient to formulate evidence-based recommendations for methicillin-sensitive Staphylococcus aureus (MSSA). Therefore, databases were searched through December 2011 for studies addressing methicillin-resistant Staphylococcus aureus (MRSA). When the MRSA-specific literature search was updated with results from December 2011 to August 2019, the MSSA literature base was deemed sufficient for review. The additional search encompassed S. aureus and included MSSA. The detailed search strategies for identifying primary literature and the search results are provided in the Appendixpdf icon[PDF – 142 pages]. (Appendix Section 1.B.) Subject matter experts supplemented the literature search results by recommending relevant references published since August 2019.
c. Study Selection
Titles and abstracts from references were screened by dual review (M.D., A.E., C.H., J.H., K.I., M.I., A.D.O., K.T.R., S.S., or E.C.S.). Full-text articles were retrieved if they were:
- Relevant to one or more Key Question;
- Primary research, systematic reviews, or meta-analyses; and
- Written in English.
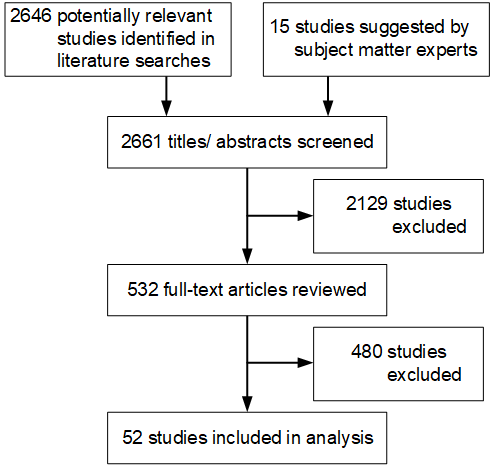
The Appendixpdf icon[PDF – 142 pages] presents the full list of exclusion criteria. (Appendix Section 2.) The full texts of selected articles were then screened by two independent reviewers, and disagreements were resolved by discussion (K.B., M.D., A.E., L.F., J.H., W.C.H., K.I., M.I., A.M., A.D.O., K.T.R., S.S., N.S., or E.C.S.). After the full-text screening was complete, a bibliography of the articles selected for inclusion was vetted with subject matter experts. Additional studies suggested by the subject matter experts were screened for inclusion as described above. The results of the study selection process are depicted in Figure 1.
Figure 1. Results of the Study Selection Process
d. Data Extraction and Synthesis
Data and results of clinically relevant outcomes from the studies meeting inclusion criteria were extracted into standardized evidence tables. Data and analyses were extracted as presented in the studies. For the purposes of this review, statistical significance was defined as p ≤ 0.05.
e. Grading of the Evidence
The risk of bias associated with each study was assessed using scales developed by the University of Pennsylvania Center for Evidence-based Practice, and scores were recorded in the evidence tables. The Appendixpdf icon[PDF – 142 pages] includes the questions used to assess the quality of each study design. (Appendix, Section 4.) The quality of the evidence base was then assessed using methods adapted from the Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group, which considers randomized controlled trials (RCTs) the gold standard. The GRADE approach[50,51] was applied to provide explicit links between the available evidence and the resulting recommendations.
The literature retrieved to answer Key Questions 3 and 4 did not include interventional studies that might target higher-risk NICU patients. As the literature retrieved for this question was insufficient to formulate recommendations for the prevention of S. aureus in NICU patients, the evidence for this Key Question was not assessed using GRADE. Rather, a qualitative summary of the results of these studies is included in the narrative.
f. Formulating Recommendations
The criteria used to formulate the strength of each recommendation are described in the HICPAC Update to the Recommendation Categorization Scheme, and in the Appendixpdf icon[PDF – 142 pages]. (Appendix Section 5.) The recommendation justification tables for each recommendation outline the factors weighed when determining the recommendation’s strength. The draft recommendations were cross-checked with the guidelines identified in the systematic literature search.
g. Reviewing and Finalizing the Guideline
A draft of the Guideline, including narrative evidence reviews, recommendations, GRADE tables, and evidence tables, was presented to HICPAC for review and input at public meetings in November 2017, May 2018, and November 2018. Following further revisions, the Guideline was in the Federal Register for a period of public comment from September 2 – November 4, 2019. The literature search was updated, and results of the updated search and the public comments received were reviewed at a HICPAC meeting on November 15, 2019. The draft was revised accordingly and finalized and is posted to CDC’s Infection Control Guidelines website.
h. Updating the Guideline
Future revisions to this Guideline will be guided by new research and technological advancements for preventing and managing infectious disease outbreaks in the NICU setting.
- Guyatt G, Oxman AD, Akl EA, et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. Journal of clinical epidemiology. 2011;64(4):383-394.
- Schunemann HJ, Oxman AD, Brozek J, et al. GRADE: assessing the quality of evidence for diagnostic recommendations. Evidence-based medicine. 2008;13(6):162-163.