Resources for Health Professionals
Updated November 10, 2021
Diagnosis
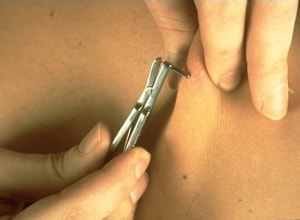
The gold standard test for the diagnosis of onchocerciasis remains the skin snip biopsy. The biopsy is performed using a sclerocorneal biopsy punch or by elevating a small cone of skin (3 mm in diameter) with a needle and shaving it off with a scalpel. This will result in the removal of around 2 mg of tissue. The tissue is then incubated in normal saline at room temperature for 24 hours to allow the microfilariae (larvae) to emerge. The microfilariae can then be identified microscopically. The sites for the skin snip are usually over the iliac crest, the scapula, and the lower extremities. Six snips provide the most diagnostic sensitivity. Skin snip sensitivity may be limited in the pre-patent stage of infection, which can last approximately 12–18 months, and in low intensity infections. Furthermore, the specificity of skin snips for the diagnosis of onchocerciasis in patients exposed in Loa loa-endemic areas may also be limited. Performing polymerase chain reaction (PCR) of the skin snip can increase the sensitivity and specificity in these two situations, though this is not commercially available.
If a patient has skin nodules caused by Onchocerca infection, nodulectomy allows for the identification of macrofilariae (adult worms) in the tissue. Slit lamp eye exam can be used to visualize microfilariae, or the lesions they cause, in individuals with eye disease.
There are antibody tests that can assist in the diagnosis of onchocerciasis, though many are not available outside the research setting. There is a general screen for any filarial infection (including Wuchereria, Brugia, Loa, and Mansonella infections) that is available in some specialty diagnostic labs. Because the test is highly sensitive, it is useful in determining if an individual has had filarial infection, but it is not specific enough to identify which filarial infection. As with any antibody test, the results indicate only that the patient has been exposed to a filarial disease, but it cannot determine if the patient has an active infection. This distinction is less important in symptomatic travelers, but it limits the usefulness of the test in persons from endemic areas. One advantage of the test is that it can pick up evidence of infection in the pre-patent stage of infection. There are several Onchocerca-specific serologic tests in existence, such as the OV-16 antigen antibody test and the OV luciferase immunoprecipitation system (LIPS) assay, but these are currently only available in research settings and are not approved for diagnosis in the United States.
In general, the diagnosis of O. volvulus infection should be made with skin snip. However, when skin snips are negative and clinical suspicion of infection is high, the general antibody test could be used in an attempt to exclude infection. If the general antibody test were positive, then it might be necessary to consider performing additional skin snips and/or seeking additional diagnostic information by enlisting the assistance of researchers who perform additional antibody tests.
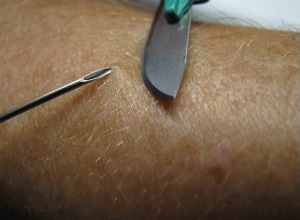
Left: A physician takes a skin sample from a patient for a skin snip biopsy by elevating a piece of skin with a needle and shaving it off with a scalpel.
Right: A physician takes a skin sample from a patient for a skin snip biopsy using a sclerocorneal biopsy punch.
Credit: Laboratory of Parasitic Diseases, NIAID, NIH, courtesy of T. Nutman, MD
Treatment
The treatment of choice for onchocerciasis is ivermectin, which has been shown to reduce the occurrence of blindness and to reduce the occurrence and severity of skin symptoms. Ivermectin kills the microfilariae (larvae), but not the macrofilariae (adult worms). There is no evidence that prolonged daily treatment provides any benefit over annual treatment, as one dose results in a significant decrease in microfilarial load that lasts a year or more. Treatment with higher than recommended doses has an increased incidence of side effects and may even be harmful. Although ivermectin does not kill the macrofilariae, it does sterilize the adult female worms. There is evidence that treating people with ivermectin more frequently than once a year facilitates more rapid sterilization of the female worms and that treating a person who no longer lives in an endemic area more frequently, such as every three to six months, could result in a shorter duration of symptoms. Treatment for a patient who will not be returning to live in an endemic area should be given every six months (and dosing as frequent as every three months could be considered) for as long as there is evidence of continued infection. Evidence of continued infection would include skin symptoms such as pruritus, microfilariae in skin biopsies, and microfilariae on eye exam. Finding adult worms in nodules would not necessarily constitute evidence of the need for continued treatment, as the adult worms do not cause symptoms and ivermectin does not kill the adult worms. Treatment with ivermectin can cause mild symptoms associated with death of the microfilariae, such as increase itching, but there is no worsening of eye symptoms. Severe adverse reactions to ivermectin in the absence of Loa loa co-infection are rare.
Doxycycline has been shown in studies to kill Wolbachia, an endosymbiotic rickettsia-like bacterium that appears to be required for the survival of the O. volvulus adult worm and for embryogenesis. Treatment with a 6-week course of doxycycline has been shown to kill more than 60% of the adult female worms and to sterilize 80 to 90% of the females 20 months after treatment. Doxycycline does not kill the microfilariae, so treatment with ivermectin would be needed to result in a more rapid decrease of symptoms. Most protocols that have examined the effectiveness of doxycycline had given treatment with ivermectin four to six months after treatment with doxycycline, so the safety of simultaneous treatment is not known. Treating with ivermectin one week prior to starting doxycycline would be reasonable. As doxycycline does not result in the rapid death of the parasites and as most of the mild side effects of ivermectin treatment are thought to be related to rapid release of Wolbachia antigens, the side effect profile of the medication when used for the treatment of onchocerciasis does not appear to be different than that of its use for other indications. There are some data to suggest that treatment of onchocerciasis with doxycycline in patients co-infected with Loa loa is safe, but this is limited to one randomized controlled trial where only people with Loa microfilarial loads < 8,000 per mL were treated and one community-based study of doxycycline treatment in co-endemic areas in which Loa microfilarial loads were not determined.
Older treatments for onchocerciasis, such as suramin and diethylcarbamazine, should not be used. Suramin has multiple systemic toxicities that limit its use in the presence of other less toxic and effective therapies. Diethylcarbamazine accelerates the development of onchocercal blindness.
Treatments for Onchocerca volvulus
| Usage/Drug | Adult Dose | Pediatric dose |
|---|---|---|
| To kill microfilariae: ivermectin |
150 mcg/kg orally in one dose every 6 months |
150 mcg/kg orally in one dose every 6 months |
| To kill macrofilariae: doxycycline* |
200 mg orally daily for 6 weeks |
200 mg orally daily for 6 weeks |
* Doxycycline is not standard therapy, but several studies support its use and safety. Treatment with ivermectin should be given one week prior to treatment with doxycycline in order to provide symptom relief to the patient. If the patient cannot tolerate 200 mg PO daily of doxycycline, 100mg PO daily is sufficient to sterilize female Onchocerca.
Oral ivermectin is available for human use in the United States.
Doxycycline is available for human use in the United States.
Note on treatment in patients co-infected with Loa loa: Patients with Loa loa co-infection should not be treated for onchocerciasis without consulting an expert on loiasis due to the risk of a fatal encephalitic reaction to ivermectin. Treatment of co-infected people with doxycycline has only been studied in persons with Loa counts of <8000 microfilariae per mL.
For questions regarding diagnostic or treatment considerations, contact the Division of Parasitic Diseases and Malaria: parasites@cdc.gov. Diagnostic assistance is also available via DPDx.