Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Prevention and Control of Seasonal Influenza with Vaccines
Recommendations of the Advisory Committee on Immunization Practices (ACIP), 2009
Please note: An erratum has been published for this article. To view the erratum, please click here.
Summary
This report updates the 2008 recommendations by CDC's Advisory Committee on Immunization Practices (ACIP) regarding the use of influenza vaccine for the prevention and control of seasonal influenza (CDC. Prevention and control of influenza: recommendations of the Advisory Committee on Immunization Practices [ACIP]. MMWR 2008;57[No. RR-7]). Information on vaccination issues related to the recently identified novel influenza A H1N1 virus will be published later in 2009. The 2009 seasonal influenza recommendations include new and updated information. Highlights of the 2009 recommendations include 1) a recommendation that annual vaccination be administered to all children aged 6 months--18 years for the 2009--10 influenza season; 2) a recommendation that vaccines containing the 2009--10 trivalent vaccine virus strains A/Brisbane/59/2007 (H1N1)-like, A/Brisbane/10/2007 (H3N2)-like, and B/Brisbane/60/2008-like antigens be used; and 3) a notice that recommendations for influenza diagnosis and antiviral use will be published before the start of the 2009--10 influenza season. Vaccination efforts should begin as soon as vaccine is available and continue through the influenza season. Approximately 83% of the United States population is specifically recommended for annual vaccination against seasonal influenza; however, <40% of the U.S. population received the 2008--09 influenza vaccine. These recommendations also include a summary of safety data for U.S. licensed influenza vaccines. These recommendations and other information are available at CDC's influenza website (http://www.cdc.gov/flu); any updates or supplements that might be required during the 2009--10 influenza season also can be found at this website. Vaccination and health-care providers should be alert to announcements of recommendation updates and should check the CDC influenza website periodically for additional information.
Introduction
In the United States, annual epidemics of seasonal influenza occur typically during the late fall through early spring. Influenza viruses can cause disease among persons in any age group, but rates of infection are highest among children (1--3). Rates of serious illness and death are highest among persons aged ≥65 years, children aged <2 years, and persons of any age who have medical conditions that place them at increased risk for complications from influenza (1,4,5). An annual average of approximately 36,000 deaths during 1990--1999 and 226,000 hospitalizations during 1979--2001 have been associated with influenza epidemics (6,7).
Annual influenza vaccination is the most effective method for preventing influenza virus infection and its complications. Influenza vaccine can be administered to any person aged >6 months who does not have contraindications to vaccination to reduce the likelihood of becoming ill with influenza or of transmitting influenza to others. Trivalent inactivated influenza vaccine (TIV) can be used for any person aged ≥6 months, including those with high-risk conditions (Boxes 1 and 2). Live, attenuated influenza vaccine (LAIV) may be used for healthy, nonpregnant persons aged 2--49 years. No preference is indicated for LAIV or TIV when considering vaccination of healthy, nonpregnant persons aged 2--49 years. Because the safety or effectiveness of LAIV has not been established in persons with underlying medical conditions that confer a higher risk for influenza complications, these persons should be vaccinated only with TIV. Influenza viruses undergo frequent antigenic change (i.e., antigenic drift); to gain immunity against viruses in circulation, patients must receive an annual vaccination against the influenza viruses that are predicted on the basis of viral surveillance data. . Although vaccination coverage has increased in recent years for many groups targeted for routine vaccination, coverage remains low among most of these groups, and strategies to improve vaccination coverage, including use of reminder/recall systems and standing orders programs, should be implemented or expanded.
Antiviral medications are an adjunct to vaccination and are effective when administered as treatment and when used for chemoprophylaxis after an exposure to influenza virus. However, the emergence since 2005 of resistance to one or more of the four licensed antiviral agents (oseltamivir, zanamivir, amantadine and rimantadine) among circulating strains has complicated antiviral treatment and chemoprophylaxis recommendations. Updated antiviral treatment and chemoprophylaxis recommendations will be provided in a separate set of guidelines later in 2009. CDC has issued interim recommendations for antiviral treatment and chemoprophylaxis of influenza (8), and these guidelines should be consulted pending issuance of new recommendations.
In April 2009, a novel influenza A (H1N1) virus that is similar to influenza viruses previously identified in swine was determined to be the cause of an influenza respiratory illness that spread across North America and was identified in many areas of the world by May 2009. The symptoms of novel influenza A (H1N1) virus infection are similar to those of seasonal influenza, and specific diagnostic testing is required to distinguish novel influenza A (H1N1) virus infection from seasonal influenza (9). The epidemiology of this illness is still being studied and prevention issues related to this newly emerging influenza virus will be published separately.
Methods
CDC's Advisory Committee on Immunization Practices (ACIP) provides annual recommendations for the prevention and control of influenza. The ACIP Influenza Vaccine Working Group* meets monthly throughout the year to discuss newly published studies, review current guidelines, and consider revisions to the recommendations. As they review the annual recommendations for ACIP consideration of the full committee, members of the working group consider a variety of issues, including burden of influenza illness, vaccine effectiveness, safety, and coverage in groups recommended for vaccination, feasibility, cost-effectiveness, and anticipated vaccine supply. Working group members also request periodic updates on vaccine and antiviral production, supply, safety and efficacy from vaccinologists, epidemiologists, and manufacturers. State and local vaccination program representatives are consulted. CDC's Influenza Division (available at http://www.cdc.gov/flu) provides influenza surveillance and antiviral resistance data. The Vaccines and Related Biological Products Advisory Committee provides advice on vaccine strain selection to the Food and Drug Administration (FDA), which selects the viral strains to be used in the annual trivalent influenza vaccines.
Published, peer-reviewed studies are the primary source of data used by ACIP in making recommendations for the prevention and control of influenza, but unpublished data that are relevant to issues under discussion also might be considered. Among studies discussed or cited, those of greatest scientific quality and those that measured influenza-specific outcomes are the most influential. For example, population-based estimates that use outcomes associated with laboratory-confirmed influenza virus infection outcomes contribute the most specific data for estimates of influenza burden. The best evidence for vaccine or antiviral efficacy and effectiveness comes from randomized controlled trials that assess laboratory-confirmed influenza infections as an outcome measure and consider factors such as timing and intensity of influenza circulation and degree of match between vaccine strains and wild circulating strains (10,11). Randomized, placebo-controlled trials cannot be performed ethically in populations for which vaccination already is recommended, but observational studies that assess outcomes associated with laboratory-confirmed influenza infection can provide important vaccine or antiviral effectiveness data. Randomized, placebo-controlled clinical trials are the best source of vaccine and antiviral safety data for common adverse events; however, such studies do not have the statistical power to identify rare but potentially serious adverse events. The frequency of rare adverse events that might be associated with vaccination is best assessed by reviewing computerized medical records from large linked clinical databases and medical charts of persons who are identified as having a potential adverse event after vaccination (12,13). Vaccine coverage data from a nationally representative, randomly selected population that includes verification of vaccination through health-care record review are superior to coverage data derived from limited populations or without verification of vaccination; however, these data rarely are available for older children or adults (14). Finally, studies that assess vaccination program practices that improve vaccination coverage are most influential in formulating recommendations if the study design includes a nonintervention comparison group. In cited studies that included statistical comparisons, a difference was considered to be statistically significant if the p-value was <0.05 or the 95% confidence interval (CI) around an estimate of effect allowed rejection of the null hypothesis (i.e., no effect).
These recommendations were presented to the full ACIP and approved in February 2009. Modifications were made to the ACIP statement during the subsequent review process at CDC to update and clarify wording in the document. Vaccine recommendations apply only to persons who do not have contraindications to vaccine use (see Contraindications and Precautions for use of TIV and Contraindications and Precautions for use of LAIV). Data presented in this report were current as of July 17, 2009. Further updates, if needed, will be posted at CDC's influenza website (http://www.cdc.gov/flu).
Primary Changes and Updates in the Recommendations
The 2009 recommendations include three principal changes or updates:
- Annual vaccination of all children aged 6 months--18 years should begin as soon as the 2009--10 influenza vaccine is available. Annual vaccination of all children aged 6 months--4 years (59 months) and older children with conditions that place them at increased risk for complications from influenza should continue to be a primary focus of vaccination efforts as providers and programs transition to routinely vaccinating all children.
- The 2009--10 trivalent vaccine virus strains are A/Brisbane/59/2007 (H1N1)-like, A/Brisbane/10/2007 (H3N2)-like, and B/Brisbane 60/2008-like antigens.
- Most seasonal influenza A (H1N1) virus strains tested from the United States and other countries are now resistant to oseltamivir. Recommendations for influenza diagnosis and antiviral use will be published later in 2009. CDC issued interim recommendations for antiviral treatment and chemoprophylaxis of influenza in December 2008, and these should be consulted for guidance pending recommendations from the ACIP (8).
Background and Epidemiology
Biology of Influenza
Influenza A and B are the two types of influenza viruses that cause epidemic human disease. Influenza A viruses are categorized into subtypes on the basis of two surface antigens: hemagglutinin and neuraminidase. Since 1977, influenza A (H1N1) viruses, influenza A (H3N2) viruses, and influenza B viruses have circulated globally. Influenza A (H1N2) viruses that probably emerged after genetic reassortment between human A (H3N2) and A (H1N1) viruses also have been identified in some influenza seasons. In April 2009, human infections with a novel influenza A (H1N1) virus were identified; as of June 2009, infections with the novel influenza A (H1N1) virus have been reported worldwide. This novel virus is derived partly from influenza A viruses that circulate in swine and is antigenically distinct from human influenza A (H1N1) viruses in circulation since 1977. Influenza A subtypes and B viruses are further separated into groups on the basis of antigenic similarities. New influenza virus variants result from frequent antigenic change (i.e., antigenic drift) resulting from point mutations and recombination events that occur during viral replication (15). Recent studies have begun to shed some light on the complex molecular evolution and epidemiologic dynamics of influenza A viruses (16--18).
Currently circulating influenza B viruses are separated into two distinct genetic lineages (Yamagata and Victoria) but are not categorized into subtypes. Influenza B viruses undergo antigenic drift less rapidly than influenza A viruses. Influenza B viruses from both lineages have circulated in most recent influenza seasons (19).
Immunity to the surface antigens, particularly the hemagglutinin, reduces the likelihood of infection (20). Antibody against one influenza virus type or subtype confers limited or no protection against another type or subtype of influenza virus. Furthermore, antibody to one antigenic type or subtype of influenza virus might not protect against infection with a new antigenic variant of the same type or subtype (21). Frequent emergence of antigenic variants through antigenic drift is the virologic basis for seasonal epidemics and is the reason for annually reassessing the need to change one or more of the recommended strains for influenza vaccines.
More dramatic changes, or antigenic shifts, occur less frequently. Antigenic shift occurs when a new subtype of influenza A virus appears and can result in the emergence of a novel influenza A virus with the potential to cause a pandemic. New influenza A subtypes have the potential to cause a pandemic when they are able to cause human illness and demonstrate efficient human-to-human transmission and little or no previously existing immunity has been identified among humans (15). Novel influenza A (H1N1) virus is not a new subtype, but because the large majority of humans appear to have no pre-existing antibody to key novel influenza A (H1N1) virus hemagglutinin epitopes, substantial potential exists for widespread infection (16).
Health-Care Use, Hospitalizations, and Deaths Attributed to Influenza
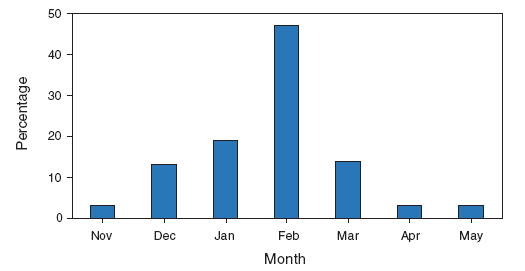
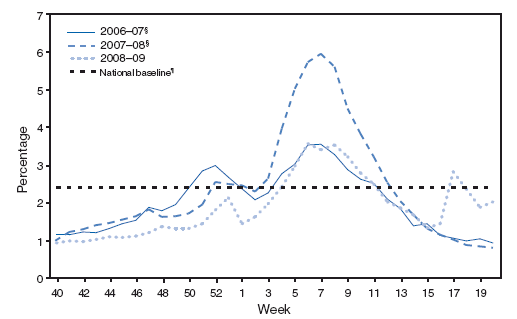
In the United States, annual epidemics of influenza typically occur during the fall or winter months, but the peak of influenza activity can occur as late as April or May (Figure 1). Influenza-related complications requiring urgent medical care, including hospitalizations or deaths, can result from the direct effects of influenza virus infection, from complications associated with age or pregnancy, or from complications of underlying cardiopulmonary conditions or other chronic diseases. Studies that have measured rates of a clinical outcome without a laboratory confirmation of influenza virus infection (e.g., respiratory illness requiring hospitalization during influenza season) to assess the effect of influenza can be difficult to interpret because of circulation of other respiratory pathogens (e.g., respiratory syncytial virus) during the same time as influenza viruses (22--24). However, increases in healthcare provider visits for acute febrile respiratory illness occur each year during the time when influenza viruses circulate. Data from the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet) demonstrate the annual increase in physician visits for influenza-like illness (ILI)† and for each influenza season; for 2009, the data also indicate the recent resurgence of respiratory illness associated with circulation of novel influenza A (H1N1) virus (Figure 2) (25,26).
During seasonal influenza epidemics from 1979--1980 through 2000--2001, the estimated annual overall number of influenza-associated hospitalizations in the United States ranged from approximately 55,000 to 431,000 per annual epidemic (mean: 226,000) (7). The estimated annual number of deaths attributed to influenza from the 1990--91 influenza season through the 1998--99 season ranged from 17,000 to 51,000 per epidemic (mean: 36,000) (6). In the United States, the estimated number of influenza-associated deaths increased during 1990--1999. This increase was attributed in part to the substantial increase in the number of persons aged ≥65 years who were at increased risk for death from influenza complications (6). In one study, an average of approximately 19,000 influenza-associated pulmonary and circulatory deaths per influenza season occurred during 1976--1990 compared with an average of approximately 36,000 deaths per season during 1990--1999 (6). In addition, influenza A (H3N2) viruses, which have been associated with higher mortality (27), predominated in 90% of influenza seasons during 1990--1999 compared with compared with 57% of seasons during 1976--1990 (6).
Influenza viruses cause disease among persons in all age groups (1--5). Rates of infection are highest among children, but the risks for complications, hospitalizations, and deaths from influenza are higher among persons aged ≥65 years, young children, and persons of any age who have medical conditions that place them at increased risk for complications from influenza (1,4,5,28--31). Estimated rates of influenza-associated hospitalizations and deaths varied substantially by age group in studies conducted during different influenza epidemics. During 1990--1999, estimated average rates of influenza-associated pulmonary and circulatory deaths per 100,000 persons were 0.4--0.6 among persons aged 0--49 years, 7.5 among persons aged 50--64 years, and 98.3 among persons aged ≥65 years (6).
Children
Among children aged <5 years, influenza-related illness is a common cause of visits to medical practices and emergency departments (EDs). During two influenza seasons (2002--03 and 2003--04), the percentage of visits among children aged <5 years with acute respiratory illness or fever caused by laboratory-confirmed influenza ranged from 10%--19% of medical office visits to 6%--29% of EDs visits during the influenza season. On the basis of these data, the rate of visits to medical clinics for influenza was estimated to be 50--95 per 1,000 children, and the rate of visits to EDs was estimated to be 6--27 per 1,000 children (32). A multiyear study in New York City used viral surveillance data to estimate influenza strain-specific illness rates among ED visits. In addition to the expected variation by season and age group, influenza B epidemics were found to be an important cause of illness among school-aged children in several seasons, and annual epidemics of both influenza A and B peaked among school-aged children before other age groups (33). Retrospective studies using medical records data have demonstrated similar rates of illness among children aged <5 years during other influenza seasons (29,34,35). During the influenza season, an estimated 7--12 additional outpatient visits and 5--7 additional antibiotic prescriptions per 100 children aged <15 years have been documented when compared with periods when influenza viruses are not circulating, with rates decreasing with increasing age of the child (35). During 1993--2004 in the Boston area, the rate of ED visits for respiratory illness that was attributed to influenza virus based on viral surveillance data among children aged ≤7 years during the winter respiratory illness season ranged from 22.0 per 1,000 children aged 6--23 months to 5.4 per 1,000 children aged 5--7 years (36).
Rates of influenza-associated hospitalization are substantially higher among infants and young children than among older children when influenza viruses are in circulation and are similar to rates for other groups considered at high risk for influenza-related complications (37--42), including persons aged ≥65 years (35,39). During 1979--2001, on the basis of data from a national sample of hospital discharges of influenza-associated hospitalizations among children aged <5 years, the estimated rate of influenza-associated hospitalizations in the United States was 108 hospitalizations per 100,000 person-years (7). Recent population-based studies that measured hospitalization rates for laboratory-confirmed influenza in young children have documented hospitalization rates that are similar to or higher than rates derived from studies that analyzed hospital discharge data (32,34,41,43,44). Annual hospitalization rates for laboratory-confirmed influenza decrease with increasing age, ranging from 240--720 per 100,000 children aged <6 months to approximately 20 per 100,000 children aged 2--5 years (32). Hospitalization rates for children aged <5 years with high-risk medical conditions are approximately 250--500 per 100,000 children (29,31,45).
Influenza-associated deaths are uncommon among children. An estimated annual average of 92 influenza-related deaths (0.4 deaths per 100,000 persons) occurred among children aged <5 years during the 1990s compared with 32,651 deaths (98.3 per 100,000 persons) among adults aged ≥65 years (6). Of 153 laboratory-confirmed influenza-related pediatric deaths reported during the 2003--04 influenza season, 96 (63%) deaths occurred among children aged <5 years and 61 (40%) among children aged <2 years. Among the 149 children who died and for whom information on underlying health status was available, 100 (67%) did not have an underlying medical condition that was an indication for vaccination at that time (46). In California during the 2003--04 and 2004--05 influenza seasons, 51% of children with laboratory-confirmed influenza who died and 40% of those who required admission to an intensive care unit had no underlying medical conditions (47). These data indicate that although children with risk factors for influenza complications are at higher risk for death, the majority of pediatric deaths occur among children with no known high-risk conditions. The annual number of influenza-associated deaths among children reported to CDC for the past four influenza seasons has ranged from 44 during 2004--05 to 84 during 2007--08 (48). As of July 8, 2009, a total of 17 deaths caused by novel influenza A (H1N1) virus infection have occurred in 2009 among children in the United States (CDC, unpublished data, 2009).
Death associated with laboratory-confirmed influenza virus infection among children (defined as persons aged <18 years) is a nationally reportable condition. Deaths among children that have been attributed to co-infection with influenza and Staphylococcus aureus, particularly methicillin-resistant S. aureus (MRSA), have increased during the preceding four influenza seasons (26,49). The reason for this increase is not established but might reflect an increasing prevalence within the general population of colonization with MRSA strains, some of which carry certain virulence factors (50,51).
Adults
Hospitalization rates during the influenza season are substantially increased for persons aged ≥65 years. One retrospective analysis based on data from managed-care organizations collected during 1996--2000 estimated that the risk during influenza season among persons aged ≥65 years with underlying conditions that put them at risk for influenza-related complications (i.e., one or more of the conditions listed as indications for vaccination) was approximately 560 influenza-associated hospitalizations per 100,000 persons compared with approximately 190 per 100,000 healthy persons. Persons aged 50--64 years with underlying medical conditions also were at substantially increased risk for hospitalizations during influenza season compared with healthy adults aged 50--64 years. No increased risk for influenza-related hospitalizations was demonstrated among healthy adults aged 50--64 years or among those aged 19--49 years, regardless of underlying medical conditions (28).
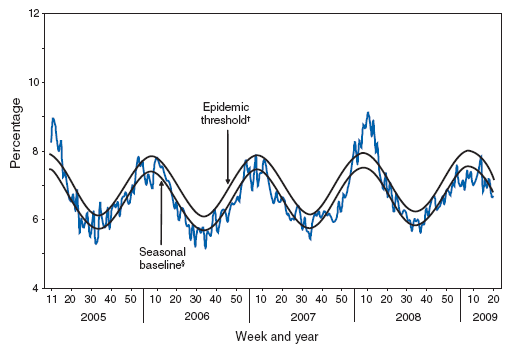
Influenza is an important contributor to the annual increase in deaths attributed to pneumonia and influenza that is observed during the winter months (Figure 3). During 1976--2001, an estimated yearly average of 32,651 (90%) influenza-related deaths occurred among adults aged ≥65 years (6). Risk for influenza-related death was highest among the oldest elderly, with persons aged ≥85 years 16 times more likely to die from an influenza-related illness than persons aged 65--69 years (6).
The duration of influenza symptoms is prolonged and the severity of influenza illness increased among persons with human immunodeficiency virus (HIV) infection (52--56). A retrospective study of young and middle-aged women enrolled in Tennessee's Medicaid program determined that the attributable risk for cardiopulmonary hospitalizations among women with HIV infection was higher during influenza seasons than it was either before or after influenza was circulating. The risk for hospitalization was higher for HIV-infected women than it was for women with other underlying medical conditions (57). Another study estimated that the risk for influenza-related death was 94--146 deaths per 100,000 persons with acquired immunodeficiency syndrome (AIDS) compared with 0.9--1.0 deaths per 100,000 persons aged 25--54 years and 64--70 deaths per 100,000 persons aged ≥65 years in the general population (58).
Influenza-related excess deaths among pregnant women were reported during the pandemics of 1918--1919 and 1957--1958 (59--63). Case reports and several epidemiologic studies also indicate that pregnancy increases the risk for influenza complications (64--69) for the mother. The majority of studies that have attempted to assess the effect of influenza on pregnant women have measured changes in excess hospitalizations for respiratory illness during influenza season but not laboratory-confirmed influenza hospitalizations. Pregnant women have an increased number of medical visits for respiratory illnesses during influenza season compared with nonpregnant women (70). Hospitalized pregnant women with respiratory illness during influenza season have increased lengths of stay compared with hospitalized pregnant women without respiratory illness. Rates of hospitalization for respiratory illness were twice as common during influenza season (71). A retrospective cohort study of approximately 134,000 pregnant women conducted in Nova Scotia during 1990--2002 compared medical record data for pregnant women to data from the same women during the year before pregnancy. Among pregnant women, 0.4% were hospitalized and 25% visited a clinician during pregnancy for a respiratory illness. The rate of third-trimester hospital admissions during the influenza season was five times higher than the rate during the influenza season in the year before pregnancy and more than twice as high as the rate during the noninfluenza season. An excess of 1,210 hospital admissions in the third trimester per 100,000 pregnant women with comorbidities and 68 admissions per 100,000 women without comorbidities was reported (72). In one study, pregnant women with respiratory hospitalizations did not have an increase in adverse perinatal outcomes or delivery complications (73); another study indicated an increase in delivery complications, including fetal distress, preterm labor, and cesarean delivery. However, infants born to women with laboratory-confirmed influenza during pregnancy do not have higher rates of low birth weight, congenital abnormalities, or lower Apgar scores compared with infants born to uninfected women (64,74).
Options for Controlling Influenza
The most effective strategy for preventing influenza is annual vaccination (10,15). Strategies that focus on providing routine vaccination to persons at higher risk for influenza complications have long been recommended, although coverage among the majority of these groups remains low. Routine vaccination of certain persons (e.g., children, contacts of persons at risk for influenza complications, and health-care personnel [HCP]) who serve as a source of influenza virus transmission might provide additional protection to persons at risk for influenza complications and reduce the overall influenza burden. However, coverage levels among these persons need to be increased before effects on transmission can be measured reliably. Antiviral drugs used for chemoprophylaxis or treatment of influenza are adjuncts to vaccine but are not substitutes for annual vaccination. However, antiviral drugs might be underused among those hospitalized with influenza (75). Nonpharmacologic interventions (e.g., advising frequent handwashing and improved respiratory hygiene) are reasonable and inexpensive; these strategies have been demonstrated to reduce respiratory diseases; reductions in detectable influenza A viruses on hands after handwashing also have been demonstrated (76--78). Few data are available to assess the effects of community-level respiratory disease mitigation strategies (e.g., closing schools, avoiding mass gatherings, or using respiratory protection) on reducing influenza virus transmission during typical seasonal influenza epidemics (79,80).
Influenza Vaccine Efficacy, Effectiveness, and Safety
Evaluating Influenza Vaccine Efficacy and Effectiveness Studies
The efficacy (i.e., prevention of illness among vaccinated persons in controlled trials) and effectiveness (i.e., prevention of illness in vaccinated populations) of influenza vaccines depend in part on the age and immunocompetence of the vaccine recipient, the degree of similarity between the viruses in the vaccine and those in circulation (see Effectiveness of Influenza Vaccination when Circulating Influenza Virus Strains Differ from Vaccine Strains), and the outcome being measured. Influenza vaccine efficacy and effectiveness studies have used multiple possible outcome measures, including the prevention of medically attended acute respiratory illness (MAARI), prevention of laboratory-confirmed influenza virus illness, prevention of influenza or pneumonia-associated hospitalizations or deaths, or prevention of seroconversion to circulating influenza virus strains. Efficacy or effectiveness for more specific outcomes such as laboratory-confirmed influenza typically will be higher than for less specific outcomes such as MAARI because the causes of MAARI include infections with other pathogens that influenza vaccination would not be expected to prevent (81). Observational studies that compare less-specific outcomes among vaccinated populations to those among unvaccinated populations are subject to biases that are difficult to control for during analyses. For example, an observational study that determines that influenza vaccination reduces overall mortality might be biased if healthier persons in the study are more likely to be vaccinated (82,83). Randomized controlled trials that measure laboratory-confirmed influenza virus infections as the outcome are the most persuasive evidence of vaccine efficacy, but such trials cannot be conducted ethically among groups recommended to receive vaccine annually.
Influenza Vaccine Composition
Both LAIV and TIV contain strains of influenza viruses that are antigenically equivalent to the annually recommended strains: one influenza A (H3N2) virus, one influenza A (H1N1) virus, and one influenza B virus. Each year, one or more virus strains in the vaccine might be changed on the basis of global surveillance for influenza viruses and the emergence and spread of new strains. For the 2009--10 influenza season, the influenza B vaccine virus strain was changed to B/Brisbane/60/2008, a representative of the B/Victoria lineage) compared with the 2008--09 season. The influenza A (H1N1 and H3N2 vaccine virus strains were not changed (84). Viruses for both types of currently licensed vaccines are grown in eggs. Both vaccines are administered annually to provide optimal protection against influenza virus infection (Table 1). Both TIV and LAIV are widely available in the United States. Although both types of vaccines are expected to be effective, the vaccines differ in several respects (Table 1).
Major Differences Between TIV and LAIV
During the preparation of TIV, the vaccine viruses are made noninfectious (i.e., inactivated or killed) (15). Only subvirion and purified surface antigen preparations of TIV (often referred to as "split" and subunit vaccines, respectively) are available in the United States. TIV contains killed viruses and thus cannot cause influenza. LAIV contains live, attenuated influenza viruses that have the potential to cause mild signs or symptoms (e.g., runny nose, nasal congestion, fever, or sore throat). LAIV is administered intranasally by sprayer, whereas TIV is administered intramuscularly by injection. LAIV is licensed for use among nonpregnant persons aged 2--49 years; safety has not been established in persons with underlying medical conditions that confer a higher risk for influenza complications. TIV is licensed for use among persons aged ≥6 months, including those who are healthy and those with chronic medical conditions (Table 1).
Correlates of Protection after Vaccination
Immune correlates of protection against influenza infection after vaccination include serum hemagglutination inhibition antibody and neutralizing antibody (20,85). Increased levels of antibody induced by vaccination decrease the risk for illness caused by strains that are antigenically similar to those strains of the same type or subtype included in the vaccine (86--89). The majority of healthy children and adults have high titers of antibody after vaccination (87,90). Although immune correlates such as achievement of certain antibody titers after vaccination correlate well with immunity on a population level, the significance of reaching or failing to reach a certain antibody threshold is not well understood on the individual level. Other immunologic correlates of protection that might best indicate clinical protection after receipt of an intranasal vaccine such as LAIV (e.g., mucosal antibody) are more difficult to measure (91,92). Laboratory measurements that correlate with protective immunity induced by LAIV have been described, including measurement of cell-mediated immunity with ELISPOT assays that measure gamma-interferon (89).
Immunogenicity, Efficacy, and Effectiveness of TIV
Children
Children aged ≥6 months typically have protective levels of anti-influenza antibody against specific influenza virus strains after receiving the recommended number of doses of influenza vaccine (85 90 93--97). In most seasons, one or more vaccine antigens are changed compared with the previous season. In consecutive years when vaccine antigens change, children aged <9 years who received only 1 dose of vaccine in their first year of vaccination are less likely to have protective antibody responses when administered only a single dose during their second year of vaccination compared with children who received 2 doses in their first year of vaccination (98--100).
When the vaccine antigens do not change from one season to the next, priming children aged 6--23 months with a single dose of vaccine in the spring followed by a dose in the fall engenders similar antibody responses compared with a regimen of 2 doses in the fall (101). However, one study conducted during a season when the vaccine antigens did not change compared with the previous season estimated 62% effectiveness against ILI for healthy children who had received only 1 dose in the previous influenza season and only 1 dose in the study season compared with 82% for those who received 2 doses separated by >4 weeks during the study season (102).
The antibody response among children at higher risk for influenza-related complications (e.g., children with chronic medical conditions) might be lower than those reported typically among healthy children (103,104). However, antibody responses among children with asthma are similar to those of healthy children and are not substantially altered during asthma exacerbations requiring short-term prednisone treatment (105).
Vaccine effectiveness studies also have indicated that 2 doses are needed to provide adequate protection during the first season that young children are vaccinated. Among children aged <5 years who have never received influenza vaccine previously or who received only 1 dose of influenza vaccine in their first year of vaccination, vaccine effectiveness is lower compared with children who received 2 doses in their first year of being vaccinated. Two large retrospective studies of young children who had received only 1 dose of TIV in their first year of being vaccinated determined that no decrease was observed in ILI-related office visits compared with unvaccinated children (102,106). Similar results were reported in a case-control study of children aged 6--59 months (107). These results, along with the immunogenicity data indicating that antibody responses are significantly higher when young children are given 2 doses, are the basis for the recommendation that all children aged <9 years who are being vaccinated for the first time should receive 2 vaccine doses separated by at least 4 weeks.
Estimates of vaccine efficacy or effectiveness among children aged ≥6 months have varied by season and study design. In a randomized trial conducted during five influenza seasons (1985--1990) in the United States among children aged 1--15 years, annual vaccination reduced laboratory-confirmed influenza A substantially (77%--91%) (87). A limited 1-year placebo-controlled study reported vaccine efficacy against laboratory-confirmed influenza illness of 56% among healthy children aged 3--9 years and 100% among healthy children and adolescents aged 10--18 years (108). A randomized, double-blind, placebo-controlled trial conducted during two influenza seasons among children aged 6--24 months indicated that efficacy was 66% against culture-confirmed influenza illness during the 1999--00 influenza season but did not reduce culture-confirmed influenza illness significantly during the 2000--20 influenza season (109).
A case-control study conducted during the 2003--04 season found vaccine effectiveness of 49% against laboratory-confirmed influenza (107). An observational study among children aged 6--59 months with laboratory-confirmed influenza compared with children who tested negative for influenza reported vaccine effectiveness of 44% in the 2003--04 influenza season and 57% during the 2004--05 season (110). Partial vaccination (only 1 dose for children being vaccinated for the first time) was not effective in either study. During an influenza season (2003--04) with a suboptimal vaccine match, a retrospective cohort study conducted among approximately 30,000 children aged 6 months--8 years indicated vaccine effectiveness of 51% against medically attended, clinically diagnosed pneumonia or influenza (i.e., no laboratory confirmation of influenza) among fully vaccinated children and 49% among approximately 5,000 children aged 6--23 months (106). Another retrospective cohort study of similar size conducted during the same influenza season in Denver but limited to healthy children aged 6--21 months estimated clinical effectiveness of 2 TIV doses to be 87% against pneumonia or influenza-related office visits (102). Among children, TIV effectiveness might increase with age (87,111). A systematic review of published studies estimated vaccine effectiveness at 59% for children aged >2 years but concluded that additional evidence was needed to demonstrate effectiveness among children aged 6 months--2 years (112).
Because of the recognized influenza-related disease burden among children with other chronic diseases or immunosuppression and the long-standing recommendation for vaccination of these children, randomized placebo-controlled studies to study efficacy in these children have not been conducted. In a nonrandomized controlled trial among children aged 2--6 years and 7--14 years who had asthma, vaccine efficacy was 54% and 78% against laboratory-confirmed influenza type A infection and 22% and 60% against laboratory-confirmed influenza type B infection, respectively. Vaccinated children aged 2--6 years with asthma did not have substantially fewer type B influenza virus infections compared with the control group in this study (113). The association between vaccination and prevention of asthma exacerbations is unclear. One study suggested that vaccination might provide protection against asthma exacerbations (114); however, other studies of children with asthma have not demonstrated decreased exacerbations (115).
TIV has been demonstrated to reduce acute otitis media in some studies. Two studies have reported that TIV decreases the risk for influenza-related otitis media by approximately 30% among children with mean ages of 20 and 27 months, respectively (116,117). However, a large study conducted among children with a mean age of 14 months indicated that TIV was not effective against acute otitis media (109). Influenza vaccine effectiveness against a nonspecific clinical outcome such as acute otitis media, which is caused by a variety of pathogens and is not typically diagnosed using influenza virus culture, would be expected to be relatively low.
Adults Aged <65 Years
One dose of TIV is highly immunogenic in healthy adults aged <65 years. Limited or no increase in antibody response is reported among adults when a second dose is administered during the same season (118--120). When the vaccine and circulating viruses are antigenically similar, TIV prevents laboratory-confirmed influenza illness among approximately 70%--90% of healthy adults aged <65 years in randomized controlled trials (121--124). Vaccination of healthy adults also has resulted in decreased work absenteeism and decreased use of health-care resources, including use of antibiotics, when the vaccine and circulating viruses are well-matched (121--123). Efficacy or effectiveness against laboratory-confirmed influenza illness was 47%--77% in studies conducted during different influenza seasons when the vaccine strains were antigenically dissimilar to the majority of circulating strains (117,119,121--124). However, effectiveness among healthy adults against influenza-related hospitalization, measured in the most recent of these studies, was 90% (125).
In certain studies, persons with certain chronic diseases have lower serum antibody responses after vaccination compared with healthy young adults and can remain susceptible to influenza virus infection and influenza-related upper respiratory tract illness (126,127). Vaccine effectiveness among adults aged <65 years who are at higher risk for influenza complications typically is lower than that reported for healthy adults. In a case-control study conducted during the 2003--04 influenza season, when the vaccine was a suboptimal antigenic match to many circulating virus strains, effectiveness for prevention of laboratory-confirmed influenza illness among adults aged 50--64 years with high-risk conditions was 48% compared with 60% for healthy adults (125). Effectiveness against hospitalization among adults aged 50--64 years with high-risk conditions was 36% compared with 90% effectiveness among healthy adults in that age range (125). A randomized controlled trial among adults in Thailand with chronic obstructive pulmonary disease (median age: 68 years) indicated a vaccine effectiveness of 76% in preventing laboratory-confirmed influenza during a season when viruses were well-matched to vaccine viruses. Effectiveness did not decrease with increasing severity of underlying lung disease (128).
Few randomized controlled trials have studied the effect of influenza vaccination on noninfluenza outcomes. A randomized controlled trial conducted in Argentina among 301 adults hospitalized with myocardial infarction or undergoing angioplasty for cardiovascular disease (56% of whom were aged ≥65 years) found that a significantly lower percentage (6%) of cardiovascular deaths occurred among vaccinated persons at 1 year after vaccination compared with unvaccinated persons (17%) (129). A randomized, double-blind, placebo-controlled study conducted in Poland among 658 persons with coronary artery disease indicated that significantly fewer vaccinated persons vaccinated persons had a cardiac ischemic event during the 9 months of follow up compared with unvaccinated persons (p <0.05) (130).
Observational studies that have measured clinical endpoints without laboratory confirmation of influenza virus infection, typically have demonstrated substantial reductions in hospitalizations or deaths among adults with risk factors for influenza complications. In a case-control study conducted during 1999--2000 in Denmark among adults aged <65 years with underlying medical conditions, vaccination reduced deaths attributable to any cause 78% and reduced hospitalizations attributable to respiratory infections or cardiopulmonary diseases 87% (131). A benefit was reported after the first vaccination and increased with subsequent vaccinations in subsequent years (132). Among patients with diabetes mellitus, vaccination was associated with a 56% reduction in any complication, a 54% reduction in hospitalizations, and a 58% reduction in deaths (133). Certain experts have noted that the substantial effects on morbidity and mortality among those who received influenza vaccination in these observational studies should be interpreted with caution because of the difficulties in ensuring that those who received vaccination had similar baseline health status as those who did not (82,83). One meta-analysis of published studies concluded that evidence was insufficient to demonstrate that persons with asthma benefit from vaccination (134). However, a meta-analysis that examined effectiveness among persons with chronic obstructive pulmonary disease identified evidence of benefit from vaccination (135).
Immunocompromised Persons
TIV produces adequate antibody concentrations against influenza among vaccinated HIV-infected persons who have minimal AIDS-related symptoms and normal or near-normal CD4+ T-lymphocyte cell counts (136--138). Among persons who have advanced HIV disease and low CD4+ T-lymphocyte cell counts, TIV might not induce protective antibody titers (138,139); a second dose of vaccine does not improve the immune response in these persons (139,140). A randomized, placebo-controlled trial determined that TIV was highly effective in preventing symptomatic, laboratory-confirmed influenza virus infection among HIV-infected persons with a mean of 400 CD4+ T-lymphocyte cells/mm3; however, a limited number of persons with CD4+ T-lymphocyte cell counts of <200 were included in that study (140). A nonrandomized study of HIV-infected persons determined that influenza vaccination was most effective among persons with >100 CD4+ cells and among those with <30,000 viral copies of HIV type-1/mL (53).
On the basis of certain limited studies, immunogenicity for persons with solid organ transplants varies according to transplant type. Among persons with kidney or heart transplants, the proportion who developed seroprotective antibody concentrations was similar or slightly reduced compared with healthy persons (141--143). However, a study among persons with liver transplants indicated reduced immunologic responses to influenza vaccination (144--146), especially if vaccination occurred within the 4 months after the transplant procedure (144).
Pregnant Women and Neonates
Pregnant women have protective levels of anti-influenza antibodies after vaccination (147,148). Passive transfer of anti-influenza antibodies that might provide protection from vaccinated women to neonates has been reported (147,149--151). A retrospective, clinic-based study conducted during 1998--2003 documented a nonsignificant trend toward fewer episodes of MAARI during one influenza season among vaccinated pregnant women compared with unvaccinated pregnant women and substantially fewer episodes of MAARI during the peak influenza season (148). However, a retrospective study conducted during 1997--2002 that used clinical records data did not indicate a reduction in ILI among vaccinated pregnant women or their infants (152). In another study conducted during 1995--2001, medical visits for respiratory illness among the infants were not substantially reduced (153). One randomized controlled trial conducted in Bangladesh that provided vaccination to pregnant women during the third trimester demonstrated a 29% reduction in respiratory illness with fever and a 36% reduction in respiratory illness with fever among their infants during the first 6 months after birth. In addition, infants born to vaccinated women had a 63% reduction in laboratory-confirmed influenza illness during the first 6 months of life (154). All women in this trial breastfed their infants (mean duration: 14 weeks).
Older Adults
Adults aged ≥65 years typically have a diminished immune response to influenza vaccination compared with young healthy adults, suggesting that immunity might be of shorter duration (although still extending through one influenza season) (155,156). However, a review of the published literature concluded that no clear evidence existed that immunity declined more rapidly in the elderly (157), and additional vaccine doses during the same season do not increase the antibody response (118,120). Infections among the vaccinated elderly might be associated with an age-related reduction in ability to respond to vaccination rather than reduced duration of immunity (127,128). One prospective cohort study found that immunogenicity among hospitalized persons who were either aged ≥65 years or who were aged 18--64 years and had one or more chronic medical conditions was similar compared with outpatients (158).
The only randomized controlled trial among community-dwelling persons aged ≥60 years reported a vaccine efficacy of 58% (CI = 26%--77%) against laboratory-confirmed influenza illness during a season when the vaccine strains were considered to be well-matched to circulating strains (159). Additional information from this trial published separately indicated that efficacy among those aged ≥70 years was 57% (CI = -36%--87%), similar to younger persons. However, few persons aged >75 years participated in this study, and the wide confidence interval for the estimate of efficacy among participants aged ≥70 years included 0 (160). Influenza vaccine effectiveness in preventing MAARI among the elderly in nursing homes has been estimated at 20%--40% (161,162), and reported outbreaks among well-vaccinated nursing home populations have suggested that vaccination might not have any significant effectiveness when circulating strains are drifted from vaccine strains (163,164). In contrast, some studies have indicated that vaccination can be up to 80% effective in preventing influenza-related death (161,165--167). Among elderly persons not living in nursing homes or similar long-term--care facilities, influenza vaccine is 27%--70% effective in preventing hospitalization for pneumonia and influenza (168--170). Influenza vaccination reduces the frequency of secondary complications and reduces the risk for influenza-related hospitalization and death among community-dwelling adults aged ≥65 years with and without high-risk medical conditions (e.g., heart disease and diabetes) (169--174). However, studies demonstrating large reductions in hospitalizations and deaths among the vaccinated elderly have been conducted using medical record databases and have not measured reductions in laboratory-confirmed influenza illness. These studies have been challenged because of concerns that they have not adequately controlled for differences in the propensity for healthier persons to be more likely than less healthy persons to receive vaccination (82,83,166,175--177).
TIV Dosage, Administration, and Storage
The composition of TIV varies according to manufacturer, and package inserts should be consulted. TIV formulations in multidose vials contain the vaccine preservative thimerosal; preservative-free, single-dose preparations also are available. TIV should be stored at 35°F--46°F (2°C--8°C) and should not be frozen. TIV that has been frozen should be discarded. Dosage recommendations and schedules vary according to age group (Table 2). Vaccine prepared for a previous influenza season should not be administered to provide protection for any subsequent season.
The intramuscular route is recommended for TIV. Adults and older children should be vaccinated in the deltoid muscle. A needle length of ≥1 inch (>25 mm) should be considered for persons in these age groups because needles of <1 inch might be of insufficient length to penetrate muscle tissue in certain adults and older children (178). When injecting into the deltoid muscle among children with adequate deltoid muscle mass, a needle length of 7/8--1.25 inches is recommended (179).
Infants and young children should be vaccinated in the anterolateral aspect of the thigh. A needle length of 7/8--1 inch should be used for children aged <12 months.
Adverse Events After Receipt of TIV
Children
Studies support the safety of annual TIV in children and adolescents. The largest published postlicensure population-based study assessed TIV safety in 251,600 children aged <18 years, (including 8,476 vaccinations in children aged 6--23 months) through the Vaccine Safety Datalink (VSD), who were enrolled in one of five health maintenance organizations (HMOs) during 1993--1999. This study indicated no increase in clinically important medically attended events during the 2 weeks after inactivated influenza vaccination compared with control periods 3--4 weeks before and after vaccination (180). A retrospective cohort study using VSD medical records data from 45,356 children aged 6--23 months provided additional evidence supporting overall safety of TIV in this age group. During the 2 weeks after vaccination, TIV was not associated with statistically significant increases in any clinically important medically attended events other than gastritis/duodenitis, and 13 diagnoses, including acute upper respiratory illness, otitis media and asthma, were significantly less common (181). On chart review, most children with a diagnosis of gastritis/duodenitis had self-limited vomiting or diarrhea. The positive or negative associations between TIV and any of these diagnoses do not necessarily indicate a causal relationship (181).
In a study of 791 healthy children aged 1--15 years, postvaccination fever was noted among 12% of those aged 1--5 years, 5% among those aged 6--10 years, and 5% among those aged 11--15 years (87). Fever, malaise, myalgia, and other systemic symptoms that can occur after vaccination with inactivated vaccine most often affect persons who have had no previous exposure to the influenza virus antigens in the vaccine (e.g., young children) (182,183). These reactions begin 6--12 hours after vaccination and can persist for 1--2 days. Data about potential adverse events among children after influenza vaccination are available from the Vaccine Adverse Event Reporting System (VAERS). Because of the limitations of passive reporting systems, determining causality for specific types of adverse events usually is not possible using VAERS data alone.
Published reviews of VAERS reports submitted after administration of TIV to children aged 6--23 months indicated that the most frequently reported adverse events were fever, rash, injection-site reactions, and seizures; the majority of the limited number of reported seizures appeared to be febrile (184,185). Seizure and fever were the leading serious adverse events (SAEs), defined using standard criteria, reported to VAERS in these studies (184,185); further investigation in VSD did not confirm an association with febrile seizures as identified in VAERS (181).
Adults
In placebo-controlled studies among adults, the most frequent side effect of vaccination was soreness at the vaccination site (affecting 10%--64% of patients) that lasted <2 days (186,187). These local reactions typically were mild and rarely interfered with the recipients' ability to conduct usual daily activities. Placebo-controlled trials demonstrate that among older persons and healthy young adults, administration of TIV is not associated with higher rates for systemic symptoms (e.g., fever, malaise, myalgia, and headache) when compared with placebo injections (121,134,186--188). One prospective cohort study found that the rate of adverse events was similar among hospitalized persons who either were aged ≥65 years or were aged 18--64 years and had one or more chronic medical conditions compared with outpatients (158). Adverse events in adults aged ≥18 years reported to VAERS during 1990--2005 were analyzed. The most common adverse events reported to VAERS in adults included injection-site reactions, pain, fever, myalgia, and headache. The VAERS review identified no new safety concerns. In clinical trials, SAEs were reported to occur after vaccination with TIV at a rate of <1%. A small proportion (14%) of the TIV VAERS reports in adults were classified as SAEs, without assessment of causality. The most common SAE reported after TIV in VAERS in adults was Guillain-Barré Syndrome (GBS) (189). The potential association between TIV and GBS has been an area of ongoing research (see Guillain-Barré Syndrome and TIV).
Pregnant Women and Neonates
FDA has classified TIV as a "Pregnancy Category C" medication, indicating that adequate animal reproduction studies have not been conducted.. Available data indicate that influenza vaccine does not cause fetal harm when administered to a pregnant woman or affect reproductive capacity. One study of approximately 2,000 pregnant women who received TIV during pregnancy demonstrated no adverse fetal effects and no adverse effects during infancy or early childhood (190). A matched case-control study of 252 pregnant women who received TIV within the 6 months before delivery determined no adverse events after vaccination among pregnant women and no difference in pregnancy outcomes compared with 826 pregnant women who were not vaccinated (148). During 2000--2003, an estimated 2 million pregnant women were vaccinated, and only 20 adverse events among women who received TIV were reported to VAERS during this time, including nine injection-site reactions and eight systemic reactions (e.g., fever, headache, and myalgias). In addition, three miscarriages were reported, but these were not known to be causally related to vaccination (191). Similar results have been reported in certain smaller studies (147,149,192), and a recent international review of data on the safety of TIV concluded that no evidence exists to suggest harm to the fetus (193). The rate of adverse events associated with TIV was similar to the rate of adverse events among pregnant women who received pneumococcal polysaccharide vaccine in one small randomized controlled trial in Bangladesh, and no severe adverse events were reported in any study group (154).
Persons with Chronic Medical Conditions
In a randomized cross-over study of children and adults with asthma, no increase in asthma exacerbations was reported for either age group (194), and two additional studies also have indicated no increase in wheezing among vaccinated asthmatic children (114) or adults (195). One study reported that 20%--28% of children with asthma aged 9 months--18 years had local pain and swelling at the site of influenza vaccination (104), and another study reported that 23% of children aged 6 months--4 years with chronic heart or lung disease had local reactions (93). A blinded, randomized, cross-over study of 1,952 adults and children with asthma demonstrated that only self-reported "body aches" were reported more frequently after TIV (25%) than placebo-injection (21%) (194). However, a placebo-controlled trial of TIV indicated no difference in local reactions among 53 children aged 6 months--6 years with high-risk medical conditions or among 305 healthy children aged 3--12 years (97).
Among children with high-risk medical conditions, one study of 52 children aged 6 months--3 years reported fever among 27% and irritability and insomnia among 25% (93); and a study among 33 children aged 6--18 months reported that one child had irritability and one had a fever and seizure after vaccination (196). No placebo comparison group was used in these studies.
Immunocompromised Persons
Data demonstrating safety of TIV for HIV-infected persons are limited, but no evidence exists that vaccination has a clinically important impact on HIV infection or immunocompetence. One study demonstrated a transient (i.e., 2--4 week) increase in HIV RNA (ribonucleic acid) levels in one HIV-infected person after influenza virus infection (197). Studies have demonstrated a transient increase in replication of HIV-1 in the plasma or peripheral blood mononuclear cells of HIV-infected persons after vaccine administration (138,198). However, more recent and better-designed studies have not documented a substantial increase in the replication of HIV (199--202). CD4+ T-lymphocyte cell counts or progression of HIV disease have not been demonstrated to change substantially after influenza vaccination among HIV-infected persons compared with unvaccinated HIV-infected persons (138,203). Limited information is available about the effect of antiretroviral therapy on increases in HIV RNA levels after either natural influenza virus infection or influenza vaccination (52,204).
Data are similarly limited for persons with other immunocompromising conditions. In small studies, vaccination did not affect allograft function or cause rejection episodes in recipients of kidney transplants (141,142), heart transplants (143), or liver transplants (144).
Immediate Hypersensitivity Reactions after Influenza Vaccines
Vaccine components can rarely cause allergic reactions, also called immediate hypersensitivity reactions, among certain recipients. Immediate hypersensitivity reactions are mediated by preformed immunoglobulin E (IgE) antibodies against a vaccine component and usually occur within minutes to hours of exposure (205). Symptoms of immediate hypersensitivity range from mild urticaria (hives) and angioedema to anaphylaxis. Anaphylaxis is a severe life-threatening reaction that involves multiple organ systems and can progress rapidly. Symptoms and signs of anaphylaxis can include but are not limited to generalized urticaria, wheezing, swelling of the mouth and throat, difficulty breathing, vomiting, hypotension, decreased level of consciousness, and shock. Minor symptoms such as red eyes or hoarse voice also might be present (179,205--208).
Allergic reactions might be caused by the vaccine antigen, residual animal protein, antimicrobial agents, preservatives, stabilizers, or other vaccine components (209). Manufacturers use a variety of compounds to inactivate influenza viruses and add antibiotics to prevent bacterial growth. Package inserts for specific vaccines of interest should be consulted for additional information. ACIP has recommended that all vaccine providers should be familiar with the office emergency plan and be certified in cardiopulmonary resuscitation (179). The Clinical Immunization Safety Assessment (CISA) network, a collaboration between CDC and six medical research centers with expertise in vaccination safety, has developed an algorithm to guide evaluation and revaccination decisions for persons with suspected immediate hypersensitivity after vaccination (205).
Immediate hypersensitivity reaction after TIV and LAIV are rare. A VSD study of children aged <18 years in four HMOs during 1991--1997 estimated the overall risk of postvaccination anaphylaxis to be less than 1 case per 500,000 doses administered and in this study no cases were identified in TIV recipients (210). Reports of anaphylaxis occurring after receipt of TIV and LAIV in adults have rarely been reported to VAERS (189).
Some immediate hypersensitivity reactions after TIV or LAIV are caused by the presence of residual egg protein in the vaccines (211). Although influenza vaccines contain only a limited quantity of egg protein, this protein can induce immediate hypersensitivity reactions among persons who have severe egg allergy. Asking persons if they can eat eggs without adverse effects is a reasonable way to determine who might be at risk for allergic reactions from receiving influenza vaccines (179). Persons who have had symptoms such as hives or swelling of the lips or tongue, or who have experienced acute respiratory distress after eating eggs, should consult a physician for appropriate evaluation to help determine if future influenza vaccine should be administered. Persons who have documented (IgE)-mediated hypersensitivity to eggs, including those who have had occupational asthma related to egg exposure or other allergic responses to egg protein, also might be at increased risk for allergic reactions to influenza vaccine, and consultation with a physician before vaccination should be considered (212--214). A regimen has been developed for administering influenza vaccine to asthmatic children with severe disease and egg hypersensitivity (213).
Hypersensitivity reactions to other vaccine components also can rarely occur. Although exposure to vaccines containing thimerosal can lead to hypersensitivity (215), the majority of patients do not have reactions to thimerosal when it is administered as a component of vaccines, even when patch or intradermal tests for thimerosal indicate hypersensitivity (216,217). When reported, hypersensitivity to thimerosal typically has consisted of local delayed hypersensitivity reactions (216).
Ocular and Respiratory Symptoms after TIV
Ocular or respiratory symptoms have occasionally been reported within 24 hours after TIV administration, but these symptoms typically are mild and resolve quickly without specific treatment. In some trials conducted in the United States, ocular or respiratory symptoms included red eyes (<1%--6%), cough (1%--7%), wheezing (1%), and chest tightness (1%--3%) (207,208,218--220). However, most of these trials were not placebo-controlled, and causality cannot be determined. In addition, ocular and respiratory symptoms are features of a variety of respiratory illnesses and seasonal allergies that would be expected to occur coincidentally among vaccine recipients unrelated to vaccination. A placebo-controlled vaccine effectiveness study among young adults found that 2% of persons who received the 2006--07 formulation of Fluzone (Sanofi Pasteur) reported red eyes compared with none of the controls (p = 0.03) (221). A similar trial conducted during the 2005--06 influenza season found that 3% of Fluzone recipients reported red eyes compared with 1% of placebo recipients; however the difference was not statistically significant (222) .
Oculorespiratory syndrome (ORS), an acute, self-limited reaction to TIV with prominent ocular and respiratory symptoms, was first described during the 2000--01 influenza season in Canada. The initial case-definition for ORS was the onset of one or more of the following within 2--24 hours after receiving TIV: bilateral red eyes and/or facial edema and/or respiratory symptoms (coughing, wheezing, chest tightness, difficulty breathing, sore throat, hoarseness or difficulty swallowing, cough, wheeze, chest tightness, difficulty breathing, sore throat, or facial swelling) (223). ORS was first described in Canada and strongly associated with one vaccine preparation (Fluviral S/F, Shire Biologics, Quebec, Canada) not available in the United States during the 2000--01 influenza season (224). Subsequent investigations identified persons with ocular or respiratory symptoms meeting an ORS case-definition in safety monitoring systems and trials that had been conducted before 2000 in Canada, the United States, and several European countries (225--227).
The cause of ORS has not been established; however studies suggest the reaction is not IgE-mediated (228). After changes in the manufacturing process of the vaccine preparation associated with ORS during 2000--01, the incidence of ORS in Canada was greatly reduced (226). In one placebo-controlled study, only hoarseness, cough, and itchy or sore eyes (but not red eyes) were significantly associated with a reformulated Fluviral preparation. These findings indicated that ORS symptoms following use of the reformulated vaccine were mild, resolved within 24 hours, and might not typically be of sufficient concern to cause vaccine recipients to seek medical care (229).
Ocular and respiratory symptoms reported after TIV administration, including ORS, have some similarities with immediate hypersensitivity reactions. One study indicated that the risk for ORS recurrence with subsequent vaccination is low, and persons with ocular or respiratory symptoms (e.g., bilateral red eyes, cough, sore throat, or hoarseness) after TIV that did not involve the lower respiratory tract have been revaccinated without reports of SAEs after subsequent exposure to TIV (230). VAERS routinely monitors for adverse events such as ocular or respiratory symptoms after receipt of TIV.
Contraindications and Precautions for Use of TIV
TIV is contraindicated and should not be administered to persons known to have anaphylactic hypersensitivity to eggs or to other components of the influenza vaccine unless the recipient has been desensitized. Prophylactic use of antiviral agents is an option for preventing influenza among such persons. Information about vaccine components is located in package inserts from each manufacturer. Persons with moderate to severe acute febrile illness usually should not be vaccinated until their symptoms have abated. Moderate or severe acute illness with or without fever is a precaution§ for TIV. GBS within 6 weeks following a previous dose of influenza vaccine is considered to be a precaution for use of influenza vaccines.
Revaccination in Persons Who Experienced Ocular or Respiratory Symptoms After TIV
When assessing whether a patient who experienced ocular and respiratory symptoms should be revaccinated, providers should determine if concerning signs and symptoms of Ig-E mediated immediate hypersensitivity are present (see Immediate Hypersensitivity after Influenza Vaccines). Health-care providers who are unsure whether symptoms reported or observed after TIV represent an IgE-mediated hypersensitivity immune response should seek advice from an allergist/immunologist. Persons with symptoms of possible IgE-mediated hypersensitivity after TIV should not receive influenza vaccination unless hypersensitivity is ruled out or revaccination is administered under close medical supervision (205).
Ocular or respiratory symptoms observed after TIV often are coincidental and unrelated to TIV administration, as observed among placebo recipients in some randomized controlled studies. Determining whether ocular or respiratory symptoms are coincidental or related to possible ORS might not be possible. Persons who have had red eyes, mild upper facial swelling, or mild respiratory symptoms (e.g., sore throat, cough, or hoarseness) after TIV without other concerning signs or symptoms of hypersensitivity can receive TIV in subsequent seasons without further evaluation. Two studies showed that persons who had symptoms of ORS after TIV were at a higher risk for ORS after subsequent TIV administration; however, these events usually were milder than the first episode (230,231).
Guillain-Barré Syndrome and TIV
The annual incidence of GBS is 10--20 cases per 1 million adults (232). Substantial evidence exists that multiple infectious illnesses, most notably Campylobacter jejuni gastrointestinal infections and upper respiratory tract infections, are associated with GBS (233--235). A recent study identified serologically confirmed influenza virus infection as a trigger of GBS, with time from onset of influenza illness to GBS of 3--30 days. The estimated frequency of influenza-related GBS was four to seven times higher than the frequency that has been estimated for influenza-vaccine--associated GBS (236).
The 1976 swine influenza vaccine was associated with an increased frequency of GBS, estimated at one additional case of GBS per 100,000 persons vaccinated (237,238). The risk for influenza-vaccine--associated GBS was higher among persons aged ≥25 years than among persons aged <25 years (239). However, obtaining epidemiologic evidence for a small increase in risk for a rare condition with multiple causes is difficult, and no evidence consistently exists for a causal relation between subsequent vaccines prepared from other influenza viruses and GBS.
None of the studies conducted using influenza vaccines other than the 1976 swine influenza vaccine has demonstrated a substantial increase in GBS associated with influenza vaccines. During three of four influenza seasons studied during 1977--1991, the overall relative risk estimates for GBS after influenza vaccination were not statistically significant in any of these studies (240--242). However, in a study of the 1992--93 and 1993--94 seasons, the overall relative risk for GBS was 1.7 (CI = 1.0--2.8; p = 0.04) during the 6 weeks after vaccination, representing approximately one additional case of GBS per 1 million persons vaccinated; the combined number of GBS cases peaked 2 weeks after vaccination (238). Results of a study that examined health-care data from Ontario, Canada, during 1992--2004 demonstrated a small but statistically significant temporal association between receiving influenza vaccination and subsequent hospital admission for GBS. However, no increase in cases of GBS at the population level was reported after introduction of a mass public influenza vaccination program in Ontario beginning in 2000 (243). Data from VAERS have documented decreased reporting of GBS occurring after vaccination across age groups over time, despite overall increased reporting of other non-GBS conditions occurring after administration of influenza vaccine (237). Published data from the United Kingdom's General Practice Research Database (GPRD) found influenza vaccine to be associated with a decreased risk for GBS, although whether this was associated with protection against influenza or confounding because of a "healthy vaccinee" (e.g., healthier persons might be more likely to be vaccinated and also be at lower risk for GBS) (244) is unclear. A separate GPRD analysis found no association between vaccination and GBS for a 9-year period; only three cases of GBS occurred within 6 weeks after administration of influenza vaccine (245). A third GPRD analysis found that GBS was associated with recent ILI, but not influenza vaccination (246).
The estimated risk for GBS (on the basis of the few studies that have demonstrated an association between vaccination and GBS) is low (i.e., approximately one additional case per 1 million persons vaccinated). The potential benefits of influenza vaccination in preventing serious illness, hospitalization, and death substantially outweigh these estimates of risk for vaccine-associated GBS. No evidence indicates that the case-fatality ratio for GBS differs among vaccinated persons and those not vaccinated.
Use of TIV Among Patients with a History of GBS
The incidence of GBS among the general population is low, but persons with a history of GBS have a substantially greater likelihood of subsequently experiencing GBS than persons without such a history (232). Thus, the likelihood of coincidentally experiencing GBS after influenza vaccination is expected to be greater among persons with a history of GBS than among persons with no history of this syndrome. Whether influenza vaccination specifically might increase the risk for recurrence of GBS is unknown. Among 311 patients with GBS who responded to a survey, 11 (4%) reported some worsening of symptoms after influenza vaccination; however, some of these patients had received other vaccines at the same time, and recurring symptoms were generally mild (247). However, as a precaution, persons who are not at high risk for severe influenza complications and who are known to have experienced GBS within 6 weeks generally should not be vaccinated. As an alternative, physicians might consider using influenza antiviral chemoprophylaxis for these persons. Although data are limited, the established benefits of influenza vaccination might outweigh the risks for many persons who have a history of GBS and who also are at high risk for severe complications from influenza.
Vaccine Preservative (Thimerosal) in Multidose Vials of TIV
Thimerosal, a mercury-containing antibacterial compound, has been used as a preservative in vaccines and other medications since the 1930s (248) and is used in multidose vial preparations of TIV to reduce the likelihood of bacterial growth. No scientific evidence indicates that thimerosal in vaccines, including influenza vaccines, is a cause of adverse events other than occasional local hypersensitivity reactions in vaccine recipients. In addition, no scientific evidence exists that thimerosal-containing vaccines are a cause of adverse events among children born to women who received vaccine during pregnancy. The weight of accumulating evidence does not suggest an increased risk for neurodevelopment disorders from exposure to thimerosal-containing vaccines (249--258). The U.S. Public Health Service and other organizations have recommended that efforts be made to eliminate or reduce the thimerosal content in vaccines as part of a strategy to reduce mercury exposures from all sources (249,250,259) Also, continuing public concerns about exposure to mercury in vaccines has been viewed as a potential barrier to achieving higher vaccine coverage levels and reducing the burden of vaccine-preventable diseases. Since mid-2001, vaccines routinely recommended for infants aged <6 months in the United States have been manufactured either without or with greatly reduced (trace) amounts of thimerosal. As a result, a substantial reduction in the total mercury exposure from vaccines for infants and children already has been achieved (179)). ACIP and other federal agencies and professional medical organizations continue to support efforts to provide thimerosal-preservative--free vaccine options.
The benefits of influenza vaccination for all recommended groups, including pregnant women and young children, outweigh concerns on the basis of a theoretical risk from thimerosal exposure through vaccination. The risks for severe illness from influenza virus infection are elevated among both young children and pregnant women, and vaccination has been demonstrated to reduce the risk for severe influenza illness and subsequent medical complications. In contrast, no scientifically conclusive evidence has demonstrated harm from exposure to vaccine containing thimerosal preservative. For these reasons, persons recommended to receive TIV may receive any age- and risk factor--appropriate vaccine preparation, depending on availability. An analysis of VAERS reports found no difference in the safety profile of preservative-containing compared with preservative-free TIV vaccines in infants aged 6--23 months (184).
Nonetheless, as of May 2009, some states have enacted legislation banning the administration of vaccines containing mercury; the provisions defining mercury content vary (260). LAIV and many of the single-dose vial or syringe preparations of TIV are thimerosal-free, and the number of influenza vaccine doses that do not contain thimerosal as a preservative is expected to increase (Table 2). However, these laws might present a barrier to vaccination unless influenza vaccines that do not contain thimerosal as a preservative are easily available in those states.
The U.S. vaccine supply for infants and pregnant women is in a period of transition as manufacturers expand the availability of thimerosal-reduced or thimerosal-free vaccine to reduce the cumulative exposure of infants to mercury. Other environmental sources of mercury exposure are more difficult or impossible to avoid or eliminate (249).
LAIV Dosage, Administration, and Storage
Each dose of LAIV contains the same three vaccine antigens used in TIV. However, the antigens are constituted as live, attenuated, cold-adapted, temperature-sensitive vaccine viruses. Providers should refer to the package insert, which contains additional information about the formulation of this vaccine and other vaccine components. LAIV does not contain thimerosal. LAIV is made from attenuated viruses that are able to replicate efficiently only at temperatures present in the nasal mucosa. LAIV does not cause systemic symptoms of influenza in vaccine recipients, although a minority of recipients experience nasal congestion or fever, which is probably a result of effects of intranasal vaccine administration or local viral replication or fever (261).
LAIV is intended for intranasal administration only and should not be administered by the intramuscular, intradermal, or intravenous route. LAIV is not licensed for vaccination of children aged <2 years or adults aged >49 years. LAIV is supplied in a prefilled, single-use sprayer containing 0.2 mL of vaccine. Approximately 0.1 mL (i.e., half of the total sprayer contents) is sprayed into the first nostril while the recipient is in the upright position. An attached dose-divider clip is removed from the sprayer to administer the second half of the dose into the other nostril. LAIV is shipped at 35°F--46°F (2°C--8°C). LAIV should be stored at 35°F--46°F (2°C--8°C) on receipt and can remain at that temperature until the expiration date is reached (261). Vaccine prepared for a previous influenza season should not be administered to provide protection for any subsequent season.
Shedding, Transmission, and Stability of Vaccine Viruses
Available data indicate that both children and adults vaccinated with LAIV can shed vaccine viruses after vaccination, although in lower amounts than occur typically with shedding of wild-type influenza viruses. In rare instances, shed vaccine viruses can be transmitted from vaccine recipients to unvaccinated persons. However, serious illnesses have not been reported among unvaccinated persons who have been infected inadvertently with vaccine viruses.
One study of 197 children aged 8--36 months in a child care center assessed transmissibility of vaccine viruses from 98 vaccinated children to the other 99 unvaccinated children; 80% of vaccine recipients shed one or more virus strains (mean duration: 7.6 days). One influenza type B vaccine strain isolate was recovered from a placebo recipient and was confirmed to be vaccine-type virus. The type B isolate retained the cold-adapted, temperature-sensitive, attenuated phenotype, and it possessed the same genetic sequence as a virus shed from a vaccine recipient who was in the same play group. The placebo recipient from whom the influenza type B vaccine strain was isolated had symptoms of a mild upper respiratory illness but did not experience any serious clinical events. The estimated probability of acquiring vaccine virus after close contact with a single LAIV recipient in this child care population was 1%--2% (262).
Studies assessing whether vaccine viruses are shed have been based on viral cultures or PCR detection of vaccine viruses in nasal aspirates from persons who have received LAIV. Among 345 subjects aged 5--49 years, 30% had detectable virus in nasal secretions obtained by nasal swabbing after receiving LAIV. The duration of virus shedding and the amount of virus shed was inversely correlated with age, and maximal shedding occurred within 2 days of vaccination. Symptoms reported after vaccination, including runny nose, headache, and sore throat, did not correlate with virus shedding (263). Other smaller studies have reported similar findings (264,265). Vaccine strain virus was detected from nasal secretions in one (2%) of 57 HIV-infected adults who received LAIV, none of 54 HIV-negative participants (266), and three (13%) of 23 HIV-infected children compared with seven (28%) of 25 children who were not HIV-infected (267). No participants in these studies had detectable virus beyond 10 days after receipt of LAIV. The possibility of person-to-person transmission of vaccine viruses was not assessed in these studies (264--267).
In clinical trials, viruses isolated from vaccine recipients have retained attenuated phenotypes. In one study, nasal and throat swab specimens were collected from 17 study participants for 2 weeks after vaccine receipt (268). Virus isolates were analyzed by multiple genetic techniques. All isolates retained the LAIV genotype after replication in the human host, and all retained the cold-adapted and temperature-sensitive phenotypes. A study conducted in a child care setting demonstrated that limited genetic change occurred in the LAIV strains following replication in the vaccine recipients (269).
Immunogenicity, Efficacy, and Effectiveness of LAIV
LAIV virus strains replicate primarily in nasopharyngeal epithelial cells. The protective mechanisms induced by vaccination with LAIV are not understood completely but appear to involve both serum and nasal secretory antibodies. The immunogenicity of the approved LAIV has been assessed in multiple studies conducted among children and adults (270--276).
Healthy Children
A randomized, double-blind, placebo-controlled trial among 1,602 healthy children aged 15--71 months assessed the efficacy of LAIV against culture-confirmed influenza during two seasons (277,278). This trial included a subset of children aged 60--71 months who received 2 doses in the first season. During season one (1996--97), when vaccine and circulating virus strains were well-matched, efficacy against culture-confirmed influenza was 94% for participants who received 2 doses of LAIV separated by ≥6 weeks, and 89% for those who received 1 dose. During season two (1997--98), when the A (H3N2) component in the vaccine was not well-matched with circulating virus strains, efficacy (1 dose) was 86%, for an overall efficacy for two influenza seasons of 92%. Receipt of LAIV also resulted in 21% fewer febrile illnesses and a significant decrease in acute otitis media requiring antibiotics (277,279). Other randomized, placebo-controlled trials demonstrating the efficacy of LAIV in young children against culture-confirmed influenza include a study conducted among children aged 6--35 months attending child care centers during consecutive influenza seasons (280) in which 85%--89% efficacy was observed, and a study conducted among children aged 12--36 months living in Asia during consecutive influenza seasons in which 64%--70% efficacy was documented (281). In one community-based, nonrandomized open-label study, reductions in MAARI were observed among children who received 1 dose of LAIV during the 1990--00 and 2000--01 influenza seasons even though antigenically drifted influenza A/H1N1 and B viruses were circulating during that season (282). LAIV efficacy in preventing laboratory-confirmed influenza also has been demonstrated in studies comparing the efficacy of LAIV with TIV rather than with a placebo (see Comparisons of LAIV and TIV Efficacy or Effectiveness).
Healthy Adults
A randomized, double-blind, placebo-controlled trial of LAIV effectiveness among 4,561 healthy working adults aged 18--64 years assessed multiple endpoints, including reductions in self-reported respiratory tract illness without laboratory confirmation, work loss, health-care visits, and medication use during influenza outbreak periods . The study was conducted during the 1997--98 influenza season, when the vaccine and circulating A (H3N2) strains were not well-matched. The frequency of febrile illnesses was not significantly decreased among LAIV recipients compared with those who received placebo. However, vaccine recipients had significantly fewer severe febrile illnesses (19% reduction) and febrile upper respiratory tract illnesses (24% reduction), and significant reductions in days of illness, days of work lost, days with health-care--provider visits, and use of prescription antibiotics and over-the-counter medications (283). Efficacy against culture-confirmed influenza in a randomized, placebo-controlled study was 57% in the 2004--05 influenza season and 43% in the 2005--06 influenza season, although efficacy in these studies was not demonstrated to be significantly greater than placebo (221,222).
Adverse Events after Receipt of LAIV
Healthy Children Aged 2--18 Years
In a subset of healthy children aged 60--71 months from one clinical trial, certain signs and symptoms were reported more often after the first dose among LAIV recipients (n = 214) than among placebo recipients (n = 95), including runny nose (48% and 44%, respectively); headache (18% and 12%, respectively); vomiting (5% and 3%, respectively); and myalgias (6% and 4%, respectively) (277). However, these differences were not statistically significant. In other trials, signs and symptoms reported after LAIV administration have included runny nose or nasal congestion (20%--75%), headache (2%--46%), fever (0--26%), vomiting (3%--13%), abdominal pain (2%), and myalgias (0--21%) (270,272,273,280,284--287). These symptoms were associated more often with the first dose and were self-limited. A placebo-controlled trial in 9,689 children aged 1--17 years assessed prespecified medically attended outcomes during the 42 days after vaccination (286). Following >1,500 statistical analyses in the 42 days after LAIV, elevated risks that were biologically plausible were observed for the following conditions: asthma, upper respiratory infection, musculoskeletal pain, otitis media with effusion, and adenitis/adenopathy. The increased risk for wheezing events after LAIV was observed among children aged 18--35 months (RR: 4.06; 90% CI = 1.3--17.9). In this study, the rate of SAEs was 0.2% in LAIV and placebo recipients; none of the SAEs was judged to be related to the vaccine by the study investigators (286).
In a randomized trial published in 2007, LAIV and TIV were compared among children aged 6--59 months (288). Children with medically diagnosed or treated wheezing within 42 days before enrollment or with a history of severe asthma were excluded from this study. Among children aged 24--59 months who received LAIV, the rate of medically significant wheezing, using a prespecified definition, was not greater compared with those who received TIV (288). Wheezing was observed more frequently among younger LAIV recipients aged 6--23 months in this study; LAIV is not licensed for this age group. In a previous randomized placebo-controlled safety trial among children aged 12 months--17 years without a history of asthma by parental report, an elevated risk for asthma events (RR: 4.1; CI = 1.3--17.9) was documented among 728 children aged 18--35 months who received LAIV. Of the 16 children with asthma-related events in this study, seven had a history of asthma on the basis of subsequent medical record review. None required hospitalization, and elevated risks for asthma were not observed in other age groups (286).
Another study was conducted among >11,000 children aged 18 months--18 years in which 18,780 doses of vaccine were administered for 4 years. For children aged 18 months--4 years, no increase was reported in asthma visits 0--15 days after vaccination compared with the prevaccination period. A significant increase in asthma events was reported 15--42 days after vaccination, but only in vaccine year 1 (289). A 4-year, open-label field trial study assessed LAIV safety of more than 2000 doses administered to children aged 18 months--18 years with a history of intermittent wheeze who were otherwise healthy. Among these children, no increased risk was reported for medically attended acute respiratory illnesses, including acute asthma exacerbation, during the 0--14 or 0--42 days after LAIV compared with the pre- and postvaccination reference periods (290).
Initial data from VAERS during 2007--2008, following ACIP's recommendation for LAIV use in healthy children aged 2--4 years, did not suggest a concern for wheezing after LAIV in young children. However data also suggest uptake of LAIV was limited, and safety monitoring for wheezing events after LAIV is ongoing (CDC, unpublished data, 2008).
Adults Aged 19--49 Years
Among adults, runny nose or nasal congestion (28%--78%), headache (16%--44%), and sore throat (15%--27%) have been reported more often among vaccine recipients than placebo recipients (277,291). In one clinical trial among a subset of healthy adults aged 18--49 years, signs and symptoms reported significantly more often (p<0.05) among LAIV recipients (n = 2,548) than placebo recipients (n = 1,290) within 7 days after each dose included cough (14% and 11%, respectively), runny nose (45% and 27%, respectively), sore throat (28% and 17%, respectively), chills (9% and 6%, respectively), and tiredness/weakness (26% and 22%, respectively) (92). A review of 460 reports to VAERS after distribution of approximately 2.5 million doses during the 2003--04 and 2004--05 influenza seasons did not indicate any new safety concerns (292). Few of the LAIV VAERS reports (9%) were SAEs; respiratory events were the most common conditions reported.
Persons at Higher Risk for Influenza-Related Complications
Limited data assessing the safety of LAIV use for certain groups at higher risk for influenza-related complications are available. In one study of 54 HIV-infected persons aged 18--58 years and with CD4+ counts ≥200 cells/mm3 who received LAIV, no SAEs were reported during a 1-month follow-up period (266). Similarly, one study demonstrated no significant difference in the frequency of adverse events or viral shedding among HIV-infected children aged 1--8 years on effective antiretroviral therapy who were administered LAIV compared with HIV-uninfected children receiving LAIV (267). LAIV was well-tolerated among adults aged ≥65 years with chronic medical conditions (293). These findings suggest that persons at risk for influenza complications who have inadvertent exposure to LAIV would not have significant adverse events or prolonged viral shedding and that persons who have contact with persons at higher risk for influenza-related complications may receive LAIV.
Comparisons of LAIV and TIV Efficacy or Effectiveness
Both TIV and LAIV have been demonstrated to be effective in children and adults. However, data directly comparing the efficacy or effectiveness of these two types of influenza vaccines are limited and insufficient to identify whether one vaccine might offer a clear advantage over the other in certain settings or populations. Studies comparing the efficacy of TIV to that of LAIV have been conducted in a variety of settings and populations using several different outcomes. One randomized, double-blind, placebo-controlled challenge study that was conducted among 92 healthy adults aged 18--41 years assessed the efficacy of both LAIV and TIV in preventing influenza infection when challenged with wild-type strains that were antigenically similar to vaccine strains (294). The overall efficacy in preventing laboratory-documented influenza from all three influenza strains combined was 85% and 71%, respectively, when challenged 28 days after vaccination by viruses to which study participants were susceptible before vaccination. The difference in efficacy between the two vaccines was not statistically significant in this limited study. No additional challenges were conducted to assess efficacy at time points later than 28 days (294). In a randomized, double-blind, placebo-controlled trial that was conducted among young adults during the 2004--05 influenza season, when the majority of circulating H3N2 viruses were antigenically drifted from that season's vaccine viruses, the efficacy of LAIV and TIV against culture-confirmed influenza was 57% and 77%, respectively. The difference in efficacy was not statistically significant and was attributable primarily to a difference in efficacy against influenza B (222). A similar study conducted during the 2005--06 influenza season found no significant difference in vaccine efficacy (221).
A randomized controlled clinical trial conducted among children aged 6--59 months during the 2004--05 influenza season demonstrated a 55% reduction in cases of culture-confirmed influenza among children who received LAIV compared with those who received TIV (288). In this study, LAIV efficacy was higher compared with TIV against antigenically drifted viruses and well-matched viruses (288). An open-label, nonrandomized, community-based influenza vaccine trial conducted during an influenza season when circulating H3N2 strains were poorly matched with strains contained in the vaccine also indicated that LAIV, but not TIV, was effective against antigenically drifted H3N2 strains during that influenza season. In this study, children aged 5--18 years who received LAIV had significant protection against laboratory-confirmed influenza (37%) and pneumonia and influenza events (50%) (295). A recent observational study conducted among military personnel aged 17--49 years over three influenza seasons indicated that persons who received TIV had a significantly lower incidence of health-care encounters resulting in diagnostic coding for pneumonia and influenza compared with those who received LAIV. However, among new recruits being vaccinated for the first time, the incidence of pneumonia- and influenza-coded health-care encounters among those received LAIV was similar to those receiving TIV (296).
Although LAIV is not licensed for use in persons with risk factors for influenza complications, certain studies have compared the efficacy of LAIV to TIV in these groups. LAIV provided 32% increased protection in preventing culture-confirmed influenza compared with TIV in one study conducted among children aged ≥6 years and adolescents with asthma (297) and 52% increased protection compared with TIV among children aged 6--71 months with recurrent respiratory tract infections (298).
Effectiveness of Vaccination for Decreasing Transmission to Contacts
Decreasing transmission of influenza from caregivers and household contacts to persons at high risk might reduce ILI and complications among persons at high risk. Influenza virus infection and ILI are common among HCP (299--301). Influenza outbreaks have been attributed to low vaccination rates among HCP in hospitals and long-term--care facilities (302--304). One serosurvey demonstrated that 23% of HCP had serologic evidence of influenza virus infection during a single influenza season; the majority had mild illness or subclinical infection (299). Observational studies have demonstrated that vaccination of HCP is associated with decreased deaths among nursing home patients (305,306). In one cluster-randomized controlled trial that included 2,604 residents of 44 nursing homes, significant decreases in mortality, ILI, and medical visits for ILI care were demonstrated among residents in nursing homes in which staff were offered influenza vaccination (coverage rate: 48%) compared with nursing homes in which staff were not provided with vaccination (coverage rate: 6%) (307). A review concluded that vaccination of HCP in settings in which patients also were vaccinated provided significant reductions in deaths among elderly patients from all causes and deaths from pneumonia (308).
Epidemiologic studies of community outbreaks of influenza demonstrate that school-aged children typically have the highest influenza illness attack rates, suggesting routine universal vaccination of children might reduce transmission to their household contacts and possibly others in the community. Results from certain studies have indicated that the benefits of vaccinating children might extend to protection of their adult contacts and to persons at risk for influenza complications in the community. However, these data are limited, and studies have not used laboratory-confirmed influenza as an outcome measure. A single-blinded, randomized controlled study conducted as part of a 1996--1997 vaccine effectiveness study demonstrated that vaccinating preschool-aged children with TIV reduced influenza-related morbidity among some household contacts (309). A randomized, placebo-controlled trial among children with recurrent respiratory tract infections demonstrated that members of families with children who had received LAIV were significantly less likely to have respiratory tract infections and reported significantly fewer workdays lost compared with families with children who received placebo (310). In nonrandomized community-based studies, administration of LAIV has been demonstrated to reduce MAARI (311,312) and ILI-related economic and medical consequences (e.g., workdays lost and number of health-care provider visits) among contacts of vaccine recipients (312). Households with children attending schools in which school-based LAIV vaccination programs had been established reported less ILI and fewer physician visits during peak influenza season compared with households with children in schools in which no LAIV vaccination had been offered. However a decrease in the overall rate of school absenteeism was not reported in communities in which LAIV vaccination was offered (312). During an influenza outbreak during the 2005--06 influenza season, countywide school-based influenza vaccination was associated with reduced absenteeism among elementary and high school students in one county that implemented a school based vaccination program compared with another county without such a program (313). These community-based studies have not used laboratory-confirmed influenza as an outcome.
Some studies also have documented reductions in influenza illness among persons living in communities where focused programs for vaccinating children have been conducted. A community-based observational study conducted during the 1968 pandemic using a univalent inactivated vaccine reported that a vaccination program targeting school-aged children (coverage rate: 86%) in one community reduced influenza rates within the community among all age groups compared with another community in which aggressive vaccination was not conducted among school-aged children (314). An observational study conducted in Russia demonstrated reductions in ILI among the community-dwelling elderly after implementation of a vaccination program using TIV for children aged 3--6 years (57% coverage achieved) and children and adolescents aged 7--17 years (72% coverage achieved) (315). In a nonrandomized community-based study conducted over three influenza seasons, 8%--18% reductions in the incidence of MAARI during the influenza season among adults aged ≥35 years were observed in communities in which LAIV was offered to all children aged ≥18 months (estimated coverage rate: 20%--25%) compared with communities that did not provide routine influenza vaccination programs for all children (311). In a subsequent influenza season, the same investigators documented a 9% reduction in MAARI rates during the influenza season among persons aged 35--44 years in intervention communities, where coverage was estimated at 31% among school children. However, MAARI rates among persons aged ≥45 years were lower in the intervention communities regardless of the presence of influenza in the community, suggesting that lower rates could not be attributed to vaccination of school children against influenza (295).
The largest study to examine the community effects of increasing overall vaccine coverage was an ecologic study that described the experience in Ontario, Canada, which was the only province to implement a universal influenza vaccination program beginning in 2000. On the basis of models developed from administrative and viral surveillance data, influenza-related mortality, hospitalizations, ED use, and physicians' office visits decreased significantly more in Ontario after program introduction than in other provinces, with the largest reductions observed in younger age groups (316).
Effectiveness of Influenza Vaccination When Circulating Influenza Virus Strains Differ from Vaccine Strains
Manufacturing trivalent influenza virus vaccines is a challenging process that takes 6--8 months to complete. Vaccination can provide reduced but substantial cross-protection against drifted strains in some seasons, including reductions in severe outcomes such as hospitalization. Usually one or more circulating viruses with antigenic changes compared with the vaccine strains are identified in each influenza season. In addition, two distinct lineages of influenza B viruses have co-circulated in recent years, and limited cross-protection is observed against the lineage not represented in the vaccine (48). However, assessment of the clinical effectiveness of influenza vaccines cannot be determined solely by laboratory evaluation of the degree of antigenic match between vaccine and circulating strains. In some influenza seasons, circulating influenza viruses with significant antigenic differences predominate, and reductions in vaccine effectiveness sometimes are observed compared with seasons when vaccine and circulating strains are well-matched,(107,121,125,173,222). However, even during years when vaccine strains were not antigenically well matched to circulating strains (the result of antigenic drift), substantial protection has been observed against severe outcomes, presumably because of vaccine-induced cross-reacting antibodies (121,125,222,283). For example, in one study conducted during the 2003--04 influenza season, when the predominant circulating strain was an influenza A (H3N2) virus that was antigenically different from that season's vaccine strain, effectiveness against laboratory-confirmed influenza illness among persons aged 50--64 years was 60% among healthy persons and 48% among persons with medical conditions that increased the risk for influenza complications (125). An interim, within-season analysis during the 2007--08 influenza season indicated that vaccine effectiveness was 44% overall, 54% among healthy persons aged 5--49 years, and 58% against influenza A, despite the finding that viruses circulating in the study area were predominately a drifted influenza A (H3N2) and an influenza B strain from a different lineage compared with vaccine strains (317). Among children, both TIV and LAIV provide protection against infection even in seasons when vaccines and circulating strains are not well-matched. Vaccine effectiveness against ILI was 49%--69% in two observational studies, and 49% against medically attended, laboratory-confirmed influenza in a case-control study conducted among young children during the 2003--04 influenza season, when a drifted influenza A (H3N2) strain predominated, based on viral surveillance data (102,106). However, continued improvements in collecting representative circulating viruses and use of surveillance data to forecast antigenic drift are needed. Shortening manufacturing time to increase the time to identify good vaccine candidate strains from among the most recent circulating strains also is important. Data from multiple seasons that are collected in a consistent manner are needed to better understand vaccine effectiveness during seasons when circulating and vaccine virus strains are not well-matched.
Seasonal influenza vaccines are not expected to provide protection against novel influenza A (H1N1) virus infection because this novel strain hemagglutinin is substantially different from seasonal influenza A (H1N1). Preliminary immunologic data indicate that few persons have antibody that shows evidence of cross-reactivity against novel influenza A (H1N1) virus, and few show increases in antibody titer to novel influenza A (H1N1) virus after vaccination with the 2007--08 or the 2008--09 seasonal influenza vaccines (318). Vaccines currently are being developed that are specific to novel influenza A (H1N1) virus.
Cost-Effectiveness of Influenza Vaccination
Economic studies of influenza vaccination are difficult to compare because they have used different measures of both costs and benefits (e.g., cost-only, cost-effectiveness, cost-benefit, or cost-utility). However, most studies find that vaccination reduces or minimizes health care, societal, and individual costs and the productivity losses and absenteeism associated with influenza illness. One national study estimated the annual economic burden of seasonal influenza in the United States (using 2003 population and dollars) to be $87.1 billion, including $10.4 billion in direct medical costs (319).
Studies of influenza vaccination in the United States among persons aged ≥65 years have estimated substantial reductions in hospitalizations and deaths and overall societal cost savings (168,169). Studies comparing adults in different age groups also find that vaccination is economically beneficial. One study that compared the economic impact of vaccination among persons aged ≥65 years with those aged 15--64 years indicated that vaccination resulted in a net savings per quality-adjusted life year (QALY) and that the Medicare program saved costs of treating illness by paying for vaccination (320). A study of a larger population comparing persons aged 50--64 years with those aged ≥65 years estimated the cost-effectiveness of influenza vaccination to be $28,000 per QALY saved (in 2000 dollars) in persons aged 50--64 years compared with $980 per QALY saved among persons aged ≥65 years (321).
Economic analyses among adults aged <65 years have reported mixed results regarding influenza vaccination. Two studies in the United States found that vaccination can reduce both direct medical costs and indirect costs from work absenteeism and reduced productivity (322,323). However, another U.S. study indicated no productivity and absentee savings in a strategy to vaccinate healthy working adults, although vaccination was still estimated to be cost-effective (324).
Cost analyses have documented the considerable financial burden of illness among children. In a study of 727 children conducted at a medical center during 2000--2004, the mean total cost of hospitalization for influenza-related illness was $13,159 ($39,792 for patients admitted to an intensive care unit and $7,030 for patients cared for exclusively on the wards) (325). A strategy that focuses on vaccinating children with medical conditions that confer a higher risk for influenza complications are more cost-effective than a strategy of vaccinating all children (324). An analysis that compared the costs of vaccinating children of varying ages with TIV and LAIV indicated that costs per QALY saved increased with age for both vaccines. In 2003 dollars per QALY saved, costs for routine vaccination using TIV were $12,000 for healthy children aged 6--23 months and $119,000 for healthy adolescents aged 12--17 years compared with $9,000 and $109,000 using LAIV, respectively (326). Economic evaluations of vaccinating children have demonstrated a wide range of cost estimates, but have generally found this strategy to be either cost-saving or cost-beneficial (327--330).
Economic analyses are sensitive to the vaccination venue, with vaccination in medical care settings incurring higher projected costs. In a published model, the mean cost (year 2004 values) of vaccination was lower in mass vaccination ($17.04) and pharmacy ($11.57) settings than in scheduled doctor's office visits ($28.67) (331). Vaccination in nonmedical settings was projected to be cost saving for healthy adults aged ≥50 years and for high-risk adults of all ages. For healthy adults aged 18--49 years, preventing an episode of influenza would cost $90 if vaccination were delivered in a pharmacy setting, $210 in a mass vaccination setting, and $870 during a scheduled doctor's office visit (331). Medicare payment rates in recent years have been less than the costs associated with providing vaccination in a medical practice (332).
Vaccination Coverage Levels
Continued annual monitoring is needed to determine the effects on vaccination coverage of vaccine supply delays and shortages, changes in influenza vaccination recommendations and target groups for vaccination, reimbursement rates for vaccine and vaccine administration, and other factors related to vaccination coverage among adults and children. One of the Healthy People 2010 objectives (objective no. 14-29a) includes achieving an influenza vaccination coverage level of 90% for persons aged ≥65 years and among nursing home residents (333,334); new strategies to improve coverage are needed to achieve this objective (335,336). Increasing vaccination coverage among persons who have high-risk conditions and are aged <65 years, including children at high risk, is the highest priority for expanding influenza vaccine use.
On the basis of the 2007 final data and the 2008 early release data from the National Health Interview Survey (NHIS), estimated national influenza vaccine coverage during the 2006--07 and 2007--08 influenza seasons increased minimally among persons aged ≥65 years and those aged 50--64 years (Table 3) and are only slightly lower than coverage levels observed before the 2004--05 vaccine shortage year (337--339). In the 2006--07 and 2007--08 influenza seasons, estimated vaccination coverage levels among adults with high-risk conditions aged 18--49 years were 25% and 30%, respectively, substantially lower than the Healthy People 2000 and Healthy People 2010 objectives of 60% (Table 3) (333,334).
Studies conducted among children and adults indicate that opportunities to vaccinate persons at risk for influenza complications (e.g., during hospitalizations for other causes) often are missed. In one study, 23% of children hospitalized with influenza and a comorbidity had a previous hospitalization during the preceding influenza vaccination season (340). In a study of hospitalized Medicare patients, only 31.6% were vaccinated before admission, 1.9% during admission, and 10.6% after admission (341). A study in New York City conducted during 2001--2005 among 7,063 children aged 6--23 months indicated that 2-dose vaccine coverage increased from 1.6% to 23.7% over time; however, although the average number of medical visits during which an opportunity to be vaccinated decreased during the course of the study from 2.9 to 2.0 per child, 55% of all visits during the final year of the study still represented a missed vaccination opportunity (342). Using standing orders in hospitals increases vaccination rates among hospitalized persons (343), and vaccination of hospitalized patients is safe and stimulates an appropriate immune response (158). In one survey, the strongest predictor of receiving vaccination was the survey respondent's belief that he or she was in a high-risk group, based on data from one survey; however, many persons in high-risk groups did not know that they were in a group recommended for vaccination (344).
Reducing racial/ethnic health disparities, including disparities in influenza vaccination coverage, is an overarching national goal that is not being met (334). Estimated vaccination coverage levels in 2007 among persons aged ≥65 years were 70% for non-Hispanic whites, 58% for non-Hispanic blacks, and 54% for Hispanics (345). Among Medicare beneficiaries, other key factors that contribute to disparities in coverage include variations in the propensity of patients to actively seek vaccination and variations in the likelihood that providers recommend vaccination (346,347). One study estimated that eliminating these disparities in vaccination coverage would have an impact on mortality similar to the impact of eliminating deaths attributable to kidney disease among blacks or liver disease among Hispanics (348).
Reported vaccination levels are low among children at increased risk for influenza complications. Coverage among children aged 2--17 years with asthma for the 2004--05 influenza season was estimated to be 29% (349). One study reported 79% vaccination coverage among children attending a cystic fibrosis treatment center (350). During the first season for which ACIP recommended that all children aged 6 months--23 months receive vaccination, 33% received 1 or more doses of influenza vaccine, and 18% received 2 doses if they were unvaccinated previously (351). Among children enrolled in HMOs who had received a first dose during 2001--2004, second dose coverage varied from 29% to 44% among children aged 6--23 months and from 12% to 24% among children aged 2--8 years (352). A rapid analysis of influenza vaccination coverage levels among members of an HMO in Northern California demonstrated that during the 2004--05 influenza season, the first year of the recommendation for vaccination of children aged 6--23 months, 1-dose coverage was 57% (353). During the 2006--07 influenza season, the second season for which ACIP recommended that all children aged 6 months--23 months receive vaccination, coverage remained low and did not increase substantially from the 2004--05 season. Data collected in 2007 by the National Immunization Survey indicated that for the 2006--07 season, 32% of children aged 6--23 months received at least 1 dose of influenza vaccine and 21% were fully vaccinated (i.e., received 1 or 2 doses depending on previous vaccination history); however, results varied substantially among states (354). As has been reported for older adults, a physician recommendation for vaccination and the perception that having a child be vaccinated "is a smart idea" were associated positively with likelihood of vaccination of children aged 6--23 months (355). Similarly, children with asthma were more likely to be vaccinated if their parents recalled a physician recommendation to be vaccinated or believed that the vaccine worked well (356). Implementation of a reminder/recall system in a pediatric clinic increased the percentage of children with asthma receiving vaccination from 5% to 32% (357).
Although annual vaccination is recommended for HCP and is a high priority for reducing morbidity associated with influenza in health-care settings and for expanding influenza vaccine use (358--360), national survey data demonstrated a vaccination coverage level of only 42% among HCP during the 2005--06 season, and 44% during the 2006--07 season (Table 3). Vaccination of HCP has been associated with reduced work absenteeism (300) and with fewer deaths among nursing home patients (305,307) and elderly hospitalized patients (308). Factors associated with a higher rate of influenza vaccination among HCP include older age, being a hospital employee, having employer-provided health-care insurance, having had pneumococcal or hepatitis B vaccination in the past, or having visited a health-care professional during the preceding year. Non-Hispanic black HCP were less likely than non-Hispanic white HCP to be vaccinated (361). HCP who decline vaccination frequently express doubts about the risk for influenza and the need for vaccination, are concerned about vaccine effectiveness and side effects, and dislike injections (362).
Vaccine coverage among pregnant women increased during the 2007--08 influenza season with 24% of pregnant women reporting vaccination, excluding pregnant women who reported diabetes, heart disease, lung disease, and other selected high-risk conditions (Table 3). However, the sample size is small, and the increase in coverage compared with previous seasons was not statistically significant. In a study of influenza vaccine acceptance by pregnant women, 71% of those who were offered the vaccine chose to be vaccinated (363). However, a 1999 survey of obstetricians and gynecologists determined that only 39% administered influenza vaccine to obstetric patients in their practices, although 86% agreed that pregnant women's risk for influenza-related morbidity and mortality increases during the last two trimesters (364).
Influenza vaccination coverage in all groups recommended for vaccination remains suboptimal. Despite the timing of the peak of influenza disease, administration of vaccine decreases substantially after November. According to results from the NHIS regarding the two most recent influenza seasons for which these data are available, approximately 84% of all influenza vaccination were administered during September--November. Among persons aged ≥65 years, the percentage of September--November vaccinations was 92% (365). Because many persons recommended for vaccination remain unvaccinated at the end of November, CDC encourages public health partners and health-care providers to conduct vaccination clinics and other activities that promote seasonal influenza vaccination annually during National Influenza Vaccination Week (December 6--12, 2009) and throughout the remainder of the influenza season.
Self-report of influenza vaccination among adults compared with determining vaccination status from the medical record, is a sensitive and specific source of information (366,367). Patient self-reports should be accepted as evidence of influenza vaccination in clinical practice (367). However, information on the validity of parents' reports of pediatric influenza vaccination is not yet available.
Recommendations for Using TIV and LAIV During the 2009--10 Influenza Season
Both TIV and LAIV prepared for the 2009--10 season will include A/Brisbane/59/2007 (H1N1)-like, A/Brisbane/10/2007 (H3N2)-like, and B/Brisbane/60/2008-like antigens. The influenza B virus component of the 2009--10 vaccine is from the Victoria lineage (368). These viruses will be used because they are representative of seasonal influenza viruses that are predicted to be circulating in the United States during the 2009--10 influenza season and have favorable growth properties in eggs. Seasonal influenza vaccines are not expected to provide substantial protection against infection with the recently identified novel influenza A (H1N1) (318), and guidance for the prevention of infection against this virus will be published separately.
TIV and LAIV can be used to reduce the risk for influenza virus infection and its complications. Vaccination providers should administer influenza vaccine to any person who wishes to reduce the likelihood of becoming ill with influenza or transmitting influenza to others should they become infected.
Healthy, nonpregnant persons aged 2--49 years can choose to receive either vaccine. Some TIV formulations are FDA-licensed for use in persons as young as age 6 months (see Recommended Vaccines for Different Age Groups). TIV is licensed for use in persons with high-risk conditions (Table 2). LAIV is FDA-licensed for use only for persons aged 2--49 years. In addition, FDA has indicated that the safety of LAIV has not been established in persons with underlying medical conditions that confer a higher risk for influenza complications. All children aged 6 months--8 years who have not been vaccinated previously at any time with at least 1 dose of either LAIV (if appropriate) or TIV should receive 2 doses of age-appropriate vaccine in the same season, with a single dose during subsequent seasons.
Target Groups for Protection Through Vaccination
Influenza vaccine should be provided to all persons who want to reduce the risk for becoming ill with influenza or of transmitting it to others. However, emphasis on providing routine vaccination annually to certain groups at higher risk for influenza infection or complications is advised, including all children aged 6 months--18 years, all persons aged ≥50 years, and other adults at risk for medical complications from influenza. In addition, all persons who live with or care for persons at high risk for influenza-related complications, including contacts of children aged <6 months, should receive influenza vaccine annually (Boxes 1 and 2). Approximately 85% of the U.S. population is included in one or more of these target groups; however, <40% of the U.S. population received an influenza vaccination during the 2008--09 influenza season.
Children Aged 6 Months--18 Years
Beginning with the 2008--09 influenza season, annual vaccination for all children aged 6 months--18 years was recommended. Children and adolescents at high risk for influenza complications should continue to be a focus of vaccination efforts as providers and programs transition to routinely vaccinating all children.
Healthy children aged 2--18 years can receive either LAIV or TIV. Children aged 6--23 months, and those aged 2--4 years who have evidence of asthma wheezing or who have medical conditions that put them at higher risk for influenza complications should receive TIV (see Considerations When Using LAIV). All children aged 6 months--8 years who have not received vaccination against influenza previously should receive 2 doses of vaccine the first year they are vaccinated.
Persons at Risk for Medical Complications
Vaccination to prevent influenza is particularly important for the following persons, who are at increased risk for severe complications from influenza, or at higher risk for influenza-related outpatient, ED, or hospital visits:
- all children aged 6 months--4 years (59 months);
- all persons aged ≥50 years;
- children and adolescents (aged 6 months--18 years) who are receiving long-term aspirin therapy and who might be at risk for experiencing Reye syndrome after influenza virus infection;
- women who will be pregnant during the influenza season;
- adults and children who have chronic pulmonary (including asthma) or cardiovascular (except hypertension), renal, hepatic, neurological/neuromuscular, hematologic, or metabolic disorders (including diabetes mellitus);
- adults and children who have immunosuppression (including immunosuppression caused by medications or by HIV); and
- residents of nursing homes and other long-term--care facilities.
For children, the risk for severe complications from seasonal influenza is highest among those aged <2 years, who have much higher rates of hospitalization for influenza-related complications compared with older children (7,32,39). Medical care and ED visits attributable to influenza are increased among children aged <5 years compared with older children (32). Chronic neurologic and neuromuscular conditions include any condition (e.g., cognitive dysfunction, spinal cord injuries, seizure disorders, or other neuromuscular disorders) that can compromise respiratory function or the handling of respiratory secretions or that can increase the risk for aspiration (30).
Persons Who Live With or Care for Persons at High Risk for Influenza-Related Complications
To prevent transmission to persons identified above, vaccination with TIV or LAIV (unless contraindicated) also is recommended for the following persons. When vaccine supply is limited, vaccination efforts should focus on delivering vaccination to these persons:
- HCP;
- household contacts (including children) and caregivers of children aged ≤59 months (i.e., aged <5 years) and adults aged ≥50 years; and
- household contacts (including children) and caregivers of persons with medical conditions that put them at higher risk for severe complications from influenza.
Children Aged <6 Months
Children aged <6 months are not recommended for vaccination, and antivirals are not licensed for use among infants. Protection of young infants, who have hospitalization rates similar to those observed among the elderly, depends on vaccination of the infants' close contacts. A recent study conducted in Bangladesh demonstrated that infants born to vaccinated women have significant protection from laboratory-confirmed influenza, either through transfer of influenza-specific maternal antibodies or by reducing the risk for exposure to influenza that might occur through vaccination of the mother (154). All household contacts, health-care and day care providers, and other close contacts of young infants should be vaccinated.
Vaccination of Specific Populations
Children Aged 6 Months--18 Years
All children aged 6 months--18 years should be vaccinated against influenza annually. In 2004, ACIP recommended routine vaccination for all children aged 6--23 months, and in 2006, ACIP expanded the recommendation to include all children aged 24--59 months. Recommendations to provide routine influenza vaccination to all children and adolescents aged 6 months--18 years are made on the basis of 1) accumulated evidence that influenza vaccine is effective and safe for children (see Influenza Vaccine Efficacy, Effectiveness, and Safety); 2) increased evidence that influenza has substantial adverse impacts among children and their contacts (e.g., school absenteeism, increased antibiotic use, medical care visits, and parental work loss) (see Health-Care Use, Hospitalizations, and Deaths Attributed to Influenza); and 3) an expectation that a simplified age-based influenza vaccine recommendation for all children and adolescents will improve vaccine coverage levels among children who already have a risk- or contact-based indication for annual influenza vaccination.
Children typically have the highest attack rates during community outbreaks of influenza and serve as a major source of transmission within communities (1,2). If sufficient vaccination coverage among children can be achieved, potential benefits include the indirect effect of reducing influenza among persons who have close contact with children and reducing overall transmission within communities. Achieving and sustaining community-level reductions in influenza will require mobilization of community resources and development of sustainable annual vaccination campaigns to assist health-care providers and vaccination programs in providing influenza vaccination services to children of all ages. In many areas, innovative community-based efforts, which might include mass vaccination programs in school or other community settings, will be needed to supplement vaccination services provided in health-care providers' offices or public health clinics. In nonrandomized community-based controlled trials, reductions in ILI-related symptoms and medical visits among household contacts have been demonstrated in communities where vaccination programs among school-aged children were established compared with communities without such vaccination programs (295,314,315).Reducing influenza-related illness among children who are at high risk for influenza complications should continue to be a primary focus of influenza-prevention efforts. Children who should be vaccinated because they are at high risk for influenza complications include all children aged 6--59 months, children with certain medical conditions, children who are contacts of children aged <5 years (60 months) or of persons aged ≥50 years, and children who are contacts of persons at high risk for influenza complications because of medical conditions.
All children aged 6 months--8 years who have not received vaccination against influenza previously should receive 2 doses of vaccine the first influenza season that they are vaccinated. The second dose should be administered 4 or more weeks after the initial dose. When only 1 dose is administered to children aged 6 months--8 years during their first year of vaccination, 2 doses should be administered in the following season. However, 2 doses should only be administered in the first season of vaccination, or in the season that immediately follows if only 1 dose is administered in the first season. For example, children aged 6 months--8 years who were vaccinated for the first time with the 2008--09 influenza vaccine but received only 1 dose should receive 2 doses of the 2009--10 influenza vaccine. All other children aged 6 months--8 years who have previously received 1 or more doses of influenza vaccine at any time should receive 1 dose of the 2009--10 influenza vaccine. Children aged 6 months--8 years who received only a single vaccination during a season before 2007--08 should receive 1 dose of the 2009--10 influenza vaccine. If possible, both doses should be administered before onset of influenza season. However, vaccination, including the second dose, is recommended even after influenza virus begins to circulate in a community.
HCP and Other Persons Who Can Transmit Influenza to Those at High Risk
Healthy persons who are infected with influenza virus, including those with subclinical infection, can transmit influenza virus to persons at higher risk for complications from influenza. In addition to HCP, groups that can transmit influenza to high-risk persons and that should be vaccinated include
- employees of assisted living and other residences for persons in groups at high risk;
- persons who provide home care to persons in groups at high risk; and
- household contacts of persons in groups at high risk, including contacts such as children or mothers of newborns.
In addition, because children aged <5 years are at increased risk for influenza-related hospitalization (7,31,39,369,370) compared with older children, vaccination is recommended for their household contacts and out-of-home caregivers. Because influenza vaccines have not been licensed by FDA for use among children aged <6 months, emphasis should be placed on vaccinating contacts of these children.
Healthy HCP and persons aged 2--49 years who are contacts of persons in these groups and who are not contacts of severely immunosuppressed persons (see Close Contacts of Immunocompromised Persons) should receive either LAIV or TIV when indicated or requested. All other persons, including pregnant women, should receive TIV.
All HCP and persons in training for health-care professions should be vaccinated annually against influenza. Persons working in health-care settings who should be vaccinated include physicians, nurses, and other workers in both hospital and outpatient-care settings, medical emergency-response workers (e.g., paramedics and emergency medical technicians), employees of nursing home and long-term--care facilities who have contact with patients or residents, and students in these professions who will have contact with patients (359,360,371).
Facilities that employ HCP should provide vaccine to workers by using approaches that have been demonstrated to be effective in increasing vaccination coverage. Health-care administrators should consider the level of vaccination coverage among HCP to be one measure of a patient safety quality program and consider obtaining signed declinations from personnel who decline influenza vaccination for reasons other than medical contraindications (360,372,373). Influenza vaccination rates among HCP within facilities should be regularly measured and reported, and ward-, unit-, and specialty-specific coverage rates should be provided to staff and administration (360). Studies have demonstrated that organized campaigns can attain higher rates of vaccination among HCP with moderate effort and by using strategies that increase vaccine acceptance (358,360,374).
Efforts to increase vaccination coverage among HCP are supported by various national accrediting and professional organizations and in certain states by statute. The Joint Commission on Accreditation of Health-Care Organizations has approved an infection-control standard that requires accredited organizations to offer influenza vaccinations to staff, including volunteers and licensed independent practitioners with close patient contact. The standard became an accreditation requirement beginning January 1, 2007 (375). In addition, the Infectious Diseases Society of America has recommended mandatory vaccination for HCP, with a provision for declination of vaccination based on religious or medical reasons (376). Some states have regulations regarding vaccination of HCP in long-term--care facilities (377), require that health-care facilities offer influenza vaccination to HCP, or require that HCP either receive influenza vaccination or indicate a religious, medical, or philosophic reason for not being vaccinated (378,379).
Close Contacts of Immunocompromised Persons
Immunocompromised persons are at risk for influenza complications but might have inadequate protection after vaccination. Close contacts of immunocompromised persons, including HCP, should be vaccinated to reduce the risk for influenza transmission. TIV is recommended for vaccinating household members, HCP, and others who have close contact with severely immunosuppressed persons (e.g., patients with hematopoietic stem cell transplants) during those periods in which the immunosuppressed person requires care in a protective environment (typically defined as a specialized patient-care area with a positive airflow relative to the corridor, high-efficiency particulate air filtration, and frequent air changes) (360,380).
LAIV transmission from a recently vaccinated person causing clinically important illness in an immunocompromised contact has not been reported. The rationale for avoiding use of LAIV among HCP or other close contacts of severely immunocompromised patients is the theoretical risk that a live, attenuated vaccine virus could be transmitted to the severely immunosuppressed person. As a precautionary measure, HCP who receive LAIV should avoid providing care for severely immunosuppressed patients requiring a protected environment for 7 days after vaccination. Hospital visitors who have received LAIV should avoid contact with severely immunosuppressed persons in protected environments for 7 days after vaccination but should not be restricted from visiting less severely immunosuppressed patients.
No preference is indicated for TIV use by persons who have close contact with persons with lesser degrees of immunosuppression (e.g., persons with diabetes, persons with asthma who take corticosteroids, persons who have recently received chemotherapy or radiation but who are not being cared for in a protective environment as defined above, or persons infected with HIV) or for TIV use by HCP or other healthy nonpregnant persons aged 2--49 years in close contact with persons in all other groups at high risk.
Pregnant Women
Pregnant women and newborns are at risk for influenza complications, and all women who are pregnant or will be pregnant during influenza season should be vaccinated. The American College of Obstetricians and Gynecologists and the American Academy of Family Physicians also have recommended routine vaccination of all pregnant women (381). No preference is indicated for use of TIV that does not contain thimerosal as a preservative (see Vaccine Preservative [Thimerosal] in Multidose Vials of TIV) for any group recommended for vaccination, including pregnant women. LAIV is not licensed for use in pregnant women. However, pregnant women do not need to avoid contact with persons recently vaccinated with LAIV.
Breastfeeding Mothers
Vaccination is recommended for all persons, including breastfeeding women, who are contacts of infants or children aged <5 years because infants and young children are at high risk for influenza complications and are more likely to require medical care or hospitalization if infected. Breastfeeding does not affect the immune response adversely and is not a contraindication for vaccination (179). Unless contraindicated because of other medical conditions, women who are breastfeeding can receive either TIV or LAIV. In one randomized controlled trial conducted in Bangladesh, infants born to women vaccinated during pregnancy had a lower risk for laboratory-confirmed influenza. However, the contribution to protection from influenza of breastfeeding compared with passive transfer of maternal antibodies during pregnancy was not determined (154).
Travelers
The risk for exposure to influenza during travel depends on the time of year and destination. In the temperate regions of the Southern Hemisphere, influenza activity occurs typically during April--September. In temperate climate zones of the Northern and Southern Hemispheres, travelers also can be exposed to influenza during the summer, especially when traveling as part of large tourist groups (e.g., on cruise ships) that include persons from areas of the world in which influenza viruses are circulating (382,383). In the tropics, influenza occurs throughout the year. In a study among Swiss travelers to tropical and subtropical countries, influenza was the most frequently acquired vaccine-preventable disease (384).
Any traveler who wants to reduce the risk for influenza infection should consider influenza vaccination, preferably at least 2 weeks before departure. In particular, persons at high risk for complications of influenza and who were not vaccinated with influenza vaccine during the preceding fall or winter should consider receiving influenza vaccine before travel if they plan to travel
- to the tropics,
- with organized tourist groups at any time of year, or
- to the Southern Hemisphere during April--September.
No information is available about the benefits of revaccinating persons before summer travel who already were vaccinated during the preceding fall, and revaccination is not recommended. Persons at high risk who receive the previous season's vaccine before travel should be revaccinated with the current vaccine the following fall or winter. Persons at higher risk for influenza complications should consult with their health-care practitioner to discuss the risk for influenza or other travel-related diseases before embarking on travel during the summer.
General Population
Vaccination is recommended for any persons who wish to reduce the likelihood of their becoming ill with influenza or transmitting influenza to others should they become infected. Healthy, nonpregnant persons aged 2--49 years might choose to receive either TIV or LAIV. All other persons aged ≥6 months should receive TIV. Persons who provide essential community services should be considered for vaccination to minimize disruption of essential activities during influenza outbreaks. Students or other persons in institutional settings (e.g., those who reside in dormitories or correctional facilities) should be encouraged to receive vaccine to minimize morbidity and the disruption of routine activities during influenza epidemics (385,386).
Recommended Vaccines for Different Age Groups
When vaccinating children aged 6--35 months with TIV, health-care providers should use TIV that has been licensed by the FDA for this age group (i.e., TIV manufactured by Sanofi Pasteur [FluZone]) (219). TIV from Novartis (Fluvirin) is FDA-approved in the United States for use among persons aged ≥4 years (220). TIV from GlaxoSmithKline (Fluarix and FluLaval) or CSL Biotherapies (Afluria) is labeled for use in persons aged ≥18 years because data to demonstrate immunogenicity or efficacy among younger persons have not been provided to FDA (207,208,218). LAIV from MedImmune (FluMist) is recommended for use by healthy nonpregnant persons aged 2--49 years (Table 2) (291). If a pediatric vaccine dose (0.25mL) is administered to an adult, an additional pediatric dose (0.25 mL) should be given to provide a full adult dose (0.5mL). If the error is discovered later (after the patient has left the vaccination setting), an adult dose should be administered as soon as the patient can return. No action needs to be taken if an adult dose is administered to a child. Several new vaccine formulations are being evaluated in immunogenicity and efficacy trials; when licensed, these new products will increase the influenza vaccine supply and provide additional vaccine choices for practitioners and their patients.
Influenza Vaccines and Use of Influenza Antiviral Medications
Unvaccinated persons who are receiving antiviral medications for treatment or chemoprophylaxis often also are recommended for vaccination. Administration of TIV to persons receiving influenza antivirals is acceptable. The effect on safety and effectiveness of LAIV coadministration with influenza antiviral medications has not been studied. However, because influenza antivirals reduce replication of influenza viruses, LAIV should not be administered until 48 hours after cessation of influenza antiviral therapy, and influenza antiviral medications should not be administered for 2 weeks after receipt of LAIV. Persons receiving antivirals within the period 2 days before to 14 days after vaccination with LAIV should be revaccinated at a later date with any approved vaccine formulation (179,291).
Considerations When Using LAIV
LAIV is an option for vaccination of healthy, nonpregnant persons aged 2--49 years, including HCP and other close contacts of high-risk persons (excepting severely immunocompromised persons who require care in a protected environment). No preference is indicated for LAIV or TIV when considering vaccination of healthy,¶ nonpregnant persons aged 2--49 years. Possible advantages of LAIV include its potential to induce a broad mucosal and systemic immune response in children, its ease of administration, and the possibly increased acceptability of an intranasal rather than intramuscular route of administration.
If the vaccine recipient sneezes after administration, the dose should not be repeated. However, if nasal congestion is present that might impede delivery of the vaccine to the nasopharyngeal mucosa, deferral of administration should be considered until resolution of the illness, or TIV should be administered instead. No data exist about concomitant use of nasal corticosteroids or other intranasal medications (261).
Although FDA licensure of LAIV excludes children aged 2--4 years with a history of asthma or recurrent wheezing, the precise risk, if any, of wheezing caused by LAIV among these children is unknown because experience with LAIV among these young children is limited. Young children might not have a history of recurrent wheezing if their exposure to respiratory viruses has been limited because of their age. Certain children might have a history of wheezing with respiratory illnesses but have not had asthma diagnosed.
The following screening recommendations should be used to assist persons who administer influenza vaccines in providing the appropriate vaccine for children aged 2--4 years.
- Clinicians and vaccination programs should screen for asthma or wheezing illness (or history of wheezing illness) when considering use of LAIV for children aged 2--4 years, and should avoid use of this vaccine in children with asthma or a recent wheezing episode within the previous 12 months. Health-care providers should consult the medical record, when available, to identify children aged 2--4 years with asthma or recurrent wheezing that might indicate asthma. In addition, to identify children who might be at greater risk for asthma and possibly at increased risk for wheezing after receiving LAIV, parents or caregivers of children aged 2--4 years should be asked: "In the past 12 months, has a health-care provider ever told you that your child had wheezing or asthma?" Children whose parents or caregivers answer "yes" to this question and children who have asthma or who had a wheezing episode noted in the medical record during the preceding 12 months should not receive LAIV. TIV is available for use in children with asthma or wheezing (387).LAIV can be administered to persons with minor acute illnesses (e.g., diarrhea or mild upper respiratory tract infection with or without fever). However, if nasal congestion is present that might impede delivery of the vaccine to the nasopharyngeal mucosa, deferral of administration should be considered until resolution of the illness.
Contraindications and Precautions for Use of LAIV
The effectiveness or safety of LAIV is not known for the following groups and administration of LAIV is contraindicated:
- persons with a history of hypersensitivity, including anaphylaxis, to any of the components of LAIV or to eggs.
- persons aged <2 years or those aged ≥50 years;
- adults and children who have chronic pulmonary (including asthma), cardiovascular (except hypertension), renal, hepatic, neurological/neuromuscular, hematological, or metabolic disorders (including diabetes mellitus);
- adults and children who have immunosuppression (including immunosuppression caused by medications or by HIV);
- children aged 2--4 years whose parents or caregivers report that a health-care provider has told them during the preceding 12 months that their child had wheezing or asthma, or whose medical record indicates a wheezing episode has occurred during the preceding 12 months;
- children or adolescents aged 6 months--18 years receiving aspirin or other salicylates (because of the association of Reye syndrome with wild-type influenza virus infection); or
- pregnant women.
A moderate or severe illness with or without fever is a precaution for use of LAIV. GBS within 6 weeks following a previous dose of influenza vaccine is considered to be a precaution for use of influenza vaccines. LAIV should not be administered to close contacts of immunosuppressed persons who require a protected environment.
Personnel Who Can Administer LAIV
Low-level introduction of vaccine viruses into the environment probably is unavoidable when administering LAIV. The risk for acquiring vaccine viruses from the environment is unknown but is probably low. Severely immunosuppressed persons should not administer LAIV. However, other persons at higher risk for influenza complications can administer LAIV. These include persons with underlying medical conditions placing them at higher risk or who are likely to be at risk, including pregnant women, persons with asthma, and persons aged ≥50 years.
Concurrent Administration of Influenza Vaccine with Other Vaccines
Use of LAIV concurrently with measles, mumps, rubella (MMR) alone and MMR and varicella vaccine among children aged 12--15 months has been studied, and no interference with the immunogenicity to antigens in any of the vaccines was observed (261,388). Among adults aged ≥50 years, the safety and immunogenicity of zoster vaccine and TIV was similar whether administered simultaneously or spaced 4 weeks apart (389). In the absence of specific data indicating interference, following ACIP's general recommendations for vaccination is prudent (179). Inactivated vaccines do not interfere with the immune response to other inactivated vaccines or to live vaccines. Inactivated or live vaccines can be administered simultaneously with LAIV. However, after administration of a live vaccine, at least 4 weeks should pass before another live vaccine is administered.
Recommendations for Vaccination Administration and Vaccination Programs
Although influenza vaccination levels increased substantially during the 1990s, little progress has been made since 2000 toward achieving national health objectives, and further improvements in vaccine coverage levels are needed to reduce the annual impact of influenza substantially. Strategies to improve vaccination levels, including using reminder/recall systems and standing orders programs (335,336,345), should be implemented whenever feasible. Vaccination efforts should begin as soon as vaccine is available and continue through the influenza season. Vaccination coverage can be increased by administering vaccine before and during the influenza season to persons during hospitalizations or routine health-care visits. Vaccinations can be provided in alternative settings (e.g., pharmacies, grocery stores, workplaces, or other locations in the community), thereby making special visits to physicians' offices or clinics unnecessary. Coordinated campaigns such as the National Influenza Vaccination Week (December 6--12, 2009) provide opportunities to refocus public attention on the benefits, safety, and availability of influenza vaccination throughout the influenza season. When educating patients about adverse events, clinicians should provide access to Vaccine Information Sheets (available at http://www.cdc.gov/vaccines/pubs/vis), and emphasize that 1) TIV contains noninfectious killed viruses and cannot cause influenza, 2) LAIV contains weakened influenza viruses that cannot replicate outside the upper respiratory tract and are unlikely to infect others, and 3) concomitant symptoms or respiratory disease unrelated to vaccination with either TIV or LAIV can occur after vaccination. Adverse events after influenza vaccination should be reported promptly to VAERS at http://vaers.hhs.gov even if the health-care professional is not certain that the vaccine caused the event.
Information About the Vaccines for Children Program
The Vaccines for Children (VFC) program supplies vaccine to all states, territories, and the District of Columbia for use by participating providers. These vaccines are to be provided to eligible children without vaccine cost to the patient or the provider, although the provider might charge a vaccine administration fee. All routine childhood vaccines recommended by ACIP are available through this program, including influenza vaccines. The program saves parents and providers out-of-pocket expenses for vaccine purchases and provides cost savings to states through CDC's vaccine contracts. The program results in lower vaccine prices and ensures that all states pay the same contract prices. Detailed information about the VFC program is available at http://www.cdc.gov/vaccines/programs/vfc/default.htm.
Influenza Vaccine Supply Considerations
The annual supply of influenza vaccine and the timing of its distribution cannot be guaranteed in any year. During the 2008--09 influenza season, 113 million doses of influenza vaccine were distributed in the United States. For the 2009--10 season, total production of seasonal influenza vaccine for the United States is anticipated to be >130 million doses, depending on demand and production yields. However, influenza vaccine distribution delays or vaccine shortages remain possible. One factor that affects production is the inherent critical time constraints in manufacturing the vaccine given the annual updating of the influenza vaccine strains. Multiple manufacturing and regulatory issues, including the anticipated need to produce a separate vaccine against novel influenza A (H1N1), also might affect the production schedule. To ensure optimal use of available doses of influenza vaccine, health-care providers, persons planning organized campaigns, and state and local public health agencies should develop plans for expanding outreach and infrastructure to vaccinate more persons in targeted groups and others who wish to reduce their risk for influenza. They also should develop contingency plans for the timing and prioritization of administering influenza vaccine if the supply of vaccine is delayed or reduced.
If supplies of TIV are not adequate, vaccination should be carried out in accordance with local circumstances of supply and demand based on the judgment of state and local health officials and health-care providers. Guidance for tiered use of TIV during prolonged distribution delays or supply shortfalls is available at http://www.cdc.gov/flu/professionals/vaccination/vax_priority.htm and will be modified as needed in the event of shortage. CDC and other public health agencies will assess the vaccine supply on a continuing basis throughout the manufacturing period and will inform both providers and the general public if any indication exists of a substantial delay or an inadequate supply.
Because LAIV is recommended for use only in healthy nonpregnant persons aged 2--49 years, no recommendations for prioritization of LAIV use are made. Either LAIV or TIV can be used when considering vaccination of healthy, nonpregnant persons aged 2--49 years. However, during shortages of TIV, LAIV should be used preferentially when feasible for all healthy nonpregnant persons aged 2--49 years (including HCP) who desire or are recommended for vaccination to increase the availability of inactivated vaccine for persons at high risk.
Timing of Vaccination
Vaccination efforts should be structured to ensure the vaccination of as many persons as possible over the course of several months, with emphasis on vaccinating before influenza activity in the community begins. Even if vaccine distribution begins before October, distribution probably will not be completed until December or January. The following recommendations reflect this phased distribution of vaccine.
In any given year, the optimal time to vaccinate patients cannot be determined precisely because influenza seasons vary in their timing and duration, and more than one outbreak might occur in a single community in a single year. In the United States, localized outbreaks that indicate the start of seasonal influenza activity can occur as early as October. However, in >80% of influenza seasons since 1976, peak influenza activity (which often is close to the midpoint of influenza activity for the season) has not occurred until January or later, and in >60% of seasons, the peak was in February or later (Figure 1). In general, health-care providers should begin offering vaccination soon after vaccine becomes available and if possible by October. To avoid missed opportunities for vaccination, providers should offer vaccination during routine health-care visits or during hospitalizations whenever vaccine is available. The potential for addition of a novel influenza A (H1N1) vaccine program to the current burden on vaccination programs and providers underscores the need for careful planning of seasonal vaccination programs. Beginning use of seasonal vaccine as soon as available, including in September or earlier, might reduce the overlap of seasonal and novel influenza vaccination efforts.
Vaccination efforts should continue throughout the season, because the duration of the influenza season varies, and influenza might not appear in certain communities until February or March. Providers should offer influenza vaccine routinely, and organized vaccination campaigns should continue throughout the influenza season, including after influenza activity has begun in the community. Vaccine administered in December or later, even if influenza activity has already begun, is likely to be beneficial in the majority of influenza seasons. The majority of adults have antibody protection against influenza virus infection within 2 weeks after vaccination (390,391).
All children aged 6 months--8 years who have not received vaccination against influenza previously should receive their first dose as soon after vaccine becomes available as is feasible and should receive the second dose ≥4 weeks later. This practice increases the opportunity for both doses to be administered before or shortly after the onset of influenza activity.
Vaccination clinics should be scheduled through December, and later if feasible, with attention to settings that serve children aged ≥6 months, pregnant women, other persons aged <50 years at increased risk for influenza-related complications, persons aged ≥50 years, HCP, and persons who are household contacts of children aged ≤59 months or other persons at high risk. Planners are encouraged to develop the capacity and flexibility to schedule at least one vaccination clinic in December. Guidelines for planning large-scale vaccination clinics are available at http://www.cdc.gov/flu/professionals/vaccination/vax_clinic.htm.
During a vaccine shortage or delay, substantial proportions of TIV doses might not be released and distributed until November and December or later. When the vaccine is substantially delayed or disease activity has not subsided, providers should consider offering vaccination clinics into January and beyond as long as vaccine supplies are available. Campaigns using LAIV also can extend into January and beyond.
Strategies for Implementing Vaccination Recommendations in Health-Care Settings
Successful vaccination programs combine publicity and education for HCP and other potential vaccine recipients, a plan for identifying persons recommended for vaccination, use of reminder/recall systems, assessment of practice-level vaccination rates with feedback to staff, and efforts to remove administrative and financial barriers that prevent persons from receiving the vaccine, including use of standing orders programs (336,392). The use of standing orders programs by long-term--care facilities (e.g., nursing homes and skilled nursing facilities), hospitals, and home health agencies ensures that vaccination is offered. Standing orders programs for influenza vaccination should be conducted under the supervision of a licensed practitioner according to a physician-approved facility or agency policy by HCP trained to screen patients for contraindications to vaccination, administer vaccine, and monitor for adverse events. The Centers for Medicare and Medicaid Services (CMS) has removed the physician signature requirement for the administration of influenza and pneumococcal vaccines to Medicare and Medicaid patients in hospitals, long-term--care facilities, and home health agencies (393). To the extent allowed by local and state law, these facilities and agencies can implement standing orders for influenza and pneumococcal vaccination of Medicare- and Medicaid-eligible patients. Payment for influenza vaccine under Medicare Part B is available (394,395) Other settings (e.g., outpatient facilities, managed care organizations, assisted living facilities, correctional facilities, pharmacies, and adult workplaces) are encouraged to introduce standing orders programs (396). In addition, physician reminders (e.g., flagging charts) and patient reminders are recognized strategies for increasing rates of influenza vaccination. Persons for whom influenza vaccine is recommended can be identified and vaccinated in the settings described in the following sections.
Outpatient Facilities Providing Ongoing Care
Staff in facilities providing ongoing medical care (e.g., physicians' offices, public health clinics, employee health clinics, hemodialysis centers, hospital specialty-care clinics, and outpatient rehabilitation programs) should identify and label the medical records of patients who should receive vaccination. Vaccine should be offered during visits throughout the influenza season. The offer of vaccination and its receipt or refusal should be documented in the medical record or vaccination information system. Patients for whom vaccination is recommended and who do not have regularly scheduled visits during the fall should be reminded by mail, telephone, or other means of the need for vaccination.
Outpatient Facilities Providing Episodic or Acute Care
Acute health-care facilities (e.g., EDs and walk-in clinics) should offer vaccinations throughout the influenza season to persons for whom vaccination is recommended or provide written information regarding why, where, and how to obtain the vaccine. This written information should be available in languages appropriate for the populations served by the facility.
Nursing Homes and Other Long-Term--Care Facilities
Vaccination should be provided routinely to all residents of long-term--care facilities. If possible, all residents should be vaccinated at one time before influenza season. In the majority of seasons, TIV will become available to long-term--care facilities in October or November, and vaccination should commence as soon as vaccine is available. As soon as possible after admission to the facility, the benefits and risks of vaccination should be discussed and education materials provided (397). Signed consent is not required (398). Residents admitted after completion of the vaccination program at the facility should be vaccinated at the time of admission.
Since October 2005, CMS has required nursing homes participating in the Medicare and Medicaid programs to offer all residents influenza and pneumococcal vaccines and to document the results. According to the requirements, each resident is to be vaccinated unless contraindicated medically, the resident or a legal representative refuses vaccination, or the vaccine is not available because of shortage. This information is to be reported as part of the CMS Minimum Data Set, which tracks nursing home health parameters (395,399).
Acute-Care Hospitals
Hospitals should serve as a key setting for identifying persons at increased risk for influenza complications. Unvaccinated persons of all ages (including children) with high-risk conditions and persons aged 6 months--18 years or ≥50 years who are hospitalized at any time during the period when vaccine is available should be offered and strongly encouraged to receive influenza vaccine before they are discharged. Standing orders to offer influenza vaccination to all hospitalized persons should be considered.
Visiting Nurses and Others Providing Home Care to Persons at High Risk
Nursing-care plans should identify patients for whom vaccination is recommended, and vaccine should be administered in the home if necessary as soon as influenza vaccine is available and throughout the influenza season. Caregivers and other persons in the household (including children) should be referred for vaccination.
Other Facilities Providing Services to Persons Aged ≥50 Years
Facilities providing services to persons aged ≥50 years (e.g., assisted living housing, retirement communities, and recreation centers) should offer unvaccinated residents, attendees, and staff annual on-site vaccination before the start of the influenza season. Continuing to offer vaccination throughout the fall and winter months is appropriate. Efforts to vaccinate newly admitted patients or new employees also should be continued, both to prevent illness and to avoid having these persons serve as a source of new influenza infections. Staff education should emphasize the benefits for self, staff and patients of protection from influenza through vaccination.
Health-Care Personnel
Health-care facilities should offer influenza vaccinations to all HCP, including night, weekend, and temporary staff. Particular emphasis should be placed on providing vaccinations to workers who provide direct care for persons at high risk for influenza complications. Efforts should be made to educate HCP regarding the benefits of vaccination and the potential health consequences of influenza illness for their patients, themselves, and their family members. All HCP should be provided convenient access to influenza vaccine at the work site, free of charge, as part of employee health programs (360,374,375).
Future Directions for Research and Recommendations Related to Influenza Vaccine
Although available influenza vaccines are effective and safe, additional research is needed to improve prevention efforts. Most mortality from influenza occurs among persons aged ≥65 years (6), and more immunogenic influenza vaccines are needed for this age group and other groups at high risk for mortality. Additional research also is needed to understand potential biases in estimating the benefits of vaccination among older adults in reducing hospitalizations and deaths (82,175,400). Additional studies of the relative cost-effectiveness and cost utility of influenza vaccination among children and adults, especially those aged <65 years, are needed and should be designed to account for year-to-year variations in influenza attack rates, illness severity, hospitalization costs and rates, and vaccine effectiveness when evaluating the long-term costs and benefits of annual vaccination (401). Additional data on indirect effects of vaccination also are needed to quantify the benefits of influenza vaccination of HCP in protecting their patients (308) and the benefits of vaccinating children to reduce influenza complications among those at risk. Because expansions in ACIP recommendations for vaccination will lead to more persons being vaccinated, much larger research networks are needed that can identify and assess the causality of very rare events that occur after vaccination, including GBS. Ongoing studies of safety in pediatric populations with expanded recommendations are needed and are underway. These research networks also could provide a platform for effectiveness and safety studies in the event of a pandemic. A recent study showed that influenza vaccines contain structures that can induce anti-GM1 antibodies after inoculation into mice (402). Further research on potential biologic or genetic risk factors for GBS in humans also is needed (397). In addition, a better understanding is needed of how to motivate persons at risk to seek annual influenza vaccination.
ACIP continues to review new vaccination strategies to protect against influenza, including the possibility of expanding routine influenza vaccination recommendations toward universal vaccination or other approaches that will help reduce or prevent the transmission of influenza and reduce the burden of severe disease (403--408). The 2009 ACIP expansion of annual vaccination recommendations to include all children aged 6 months--18 years will require a substantial increase in resources for epidemiologic research to develop long-term studies capable of assessing the possible effects on community-level transmission. Additional planning to improve surveillance systems capable of monitoring effectiveness, safety and vaccine coverage, and further development of implementation strategies will also be necessary. In addition, as noted by the National Vaccine Advisory Committee, strengthening the U.S. influenza vaccination system will require improving vaccine financing and demand and implementing systems to help better understand the burden of influenza in the United States (409). Vaccination programs capable of delivering annual influenza vaccination to a broad range of the population could potentially serve as a resilient and sustainable platform for delivering vaccines and monitoring outcomes for other urgently required public health interventions (e.g., vaccines for pandemic influenza or medications to prevent or treat illnesses caused by acts of terrorism).
Seasonal Influenza Vaccine and Influenza Viruses of Animal Origin
Human infection with novel or nonhuman influenza A virus strains, including influenza A viruses of animal origin, is a nationally notifiable disease (410). Human infections with nonhuman or novel human influenza A virus should be identified quickly and investigated to determine possible sources of exposure, identify additional cases, and evaluate the possibility of human-to-human transmission because transmission patterns could change over time with variations in these influenza A viruses.
Sporadic severe and fatal human cases of infection with highly pathogenic avian influenza A (H5N1) virus have been identified in Asia, Africa, Europe, and the Middle East, primarily among persons who have had direct or close unprotected contact with sick or dead birds associated with the ongoing H5N1 panzootic among birds (411--419). Limited, nonsustained human-to-human transmission of H5N1 virus has likely occurred in some case clusters (420,421). To date, no evidence exists of genetic reassortment between human influenza A and H5N1 viruses. However, influenza viruses derived from strains circulating among poultry (e.g., the H5N1 virus that has caused outbreaks of avian influenza and occasionally have infected humans) have the potential to recombine with human influenza A viruses (422,423). To date, highly pathogenic H5N1 virus has not been identified in wild or domestic birds or in humans in the United States. Guidance for testing suspected cases of H5N1 virus infection among persons in the U.S. and follow-up of contacts is available (424,425).
Human illness from infection with different avian influenza A subtype viruses also have been documented, including infections with low pathogenic and highly pathogenic viruses. A range of clinical illness has been reported for human infection with low pathogenic avian influenza viruses, including conjunctivitis with influenza A (H7N7) virus in the United Kingdom, lower respiratory tract disease and conjunctivitis with influenza A (H7N2) virus in the United Kingdom, and uncomplicated ILI with influenza A (H9N2) virus in Hong Kong and China (426--432). Two human cases of infection with low pathogenic influenza A (H7N2) were reported in the United States (429). Although human infections with highly pathogenic A (H7N7) virus infections typically have ILI or conjunctivitis, severe infections, including one fatal case in the Netherlands, have been reported (433,434). Conjunctivitis also has been reported because of human infection with highly pathogenic influenza A (H7N3) virus in Canada and low pathogenic A (H7N3) in the United Kingdom (426,434). In contrast, sporadic infections with highly pathogenic avian influenza A (H5N1) virus have caused severe illness in many countries, with an overall case-fatality proportion of >60% (421,435).
Swine influenza A (H1N1), A (H1N2), and A (H3N2) viruses, including reassortant viruses, are endemic among pig populations in the United States (436). Two clusters of influenza A (H2N3) virus infections among pigs have been reported recently (437). Outbreaks among pigs normally occur in colder weather months (late fall and winter) and sometimes with the introduction of new pigs into susceptible herds. An estimated 30% of the pig population in the United States has serologic evidence of having had swine influenza A (H1N1) virus infection. Sporadic human infections with a variety of swine influenza A viruses occur in the United States, but the incidence of these human infections is unknown (438--443). Persons infected with swine influenza A viruses typically report direct contact with ill pigs or places where pigs have been present (e.g., agricultural fairs or farms), and have symptoms that are clinically indistinguishable from infection with other respiratory viruses (440,441,444,445). Clinicians should consider swine influenza A virus infection in the differential diagnosis of patients with ILI who have had recent contact with pigs. The sporadic cases identified in recent years have not resulted in sustained human-to-human transmission of swine influenza A viruses or community outbreaks (368,445). Although immunity to swine influenza A viruses appears to be low (<2%) in the overall human population, 10%--20% of persons exposed occupationally to pigs (e.g., pig farmers or pig veterinarians) have been documented in certain studies to have antibody evidence of prior swine influenza A (H1N1) virus infection (438,446).
In April 2009, a novel influenza A (H1N1) virus similar to influenza viruses previously identified in swine was determined to the cause of an influenza-like respiratory illness among humans that spread across North America and throughout most of the world by May 2009 (9,447). The epidemiology of influenza caused by this novel influenza virus is still being studied, and whether this virus will achieve long-term circulation among humans or even replace one of the other seasonal influenza viruses as the cause of annual epidemics is unknown.
Current seasonal influenza vaccines are not expected to provide protection against human infection with avian influenza A viruses, including influenza A (H5N1) viruses, or to provide protection against currently circulating swine influenza A or the novel influenza A (H1N1) viruses (318,448). However, reducing seasonal influenza risk through influenza vaccination of persons who might be exposed to nonhuman influenza viruses (e.g., H5N1 virus) might reduce the theoretical risk for recombination of influenza A viruses of animal origin and human influenza A viruses by preventing seasonal influenza A virus infection within a human host.
CDC has recommended that persons who are charged with responding to avian influenza outbreaks among poultry receive seasonal influenza vaccination (448,449). As part of preparedness activities, the Occupational Safety and Health Administration (OSHA) has issued an advisory notice regarding poultry worker safety that is intended for implementation in the event of a suspected or confirmed avian influenza outbreak at a poultry facility in the United States. OSHA guidelines recommend that poultry workers in an involved facility receive vaccination against seasonal influenza; OSHA also has recommended that HCP involved in the care of patients with documented or suspected avian influenza should be vaccinated with the most recent seasonal human influenza vaccine to reduce the risk for co-infection with human influenza A viruses (449).
Recommendations for Using Antiviral Agents for Seasonal Influenza
Annual vaccination is the primary strategy for preventing complications of influenza virus infections. Antiviral medications with activity against influenza viruses are useful adjuncts in the prevention of influenza, and effective when used early in the course of illness for treatment. Four influenza antiviral agents are licensed in the United States: amantadine, rimantadine, zanamivir, and oseltamivir.
During the 2007--08 influenza season, influenza A (H1N1) viruses with a mutation that confers resistance to oseltamivir became more common in the United States and other countries (450--452). As of July 2009, in the United States, approximately 99% of human influenza A (H1N1) viruses tested, and none of the influenza A (H3N2) or influenza B viruses tested have been resistant to oseltamivir. As of July 2, 2009, with few exceptions, novel influenza A (H1N1) viruses that began circulating in April 2009 remained sensitive to oseltamivir (453). Oseltamivir resistance among circulating seasonal influenza A (H1N1) virus strains presents challenges for the selection of antiviral medications for treatment and chemoprophylaxis of influenza, and provides additional reasons for clinicians to test patients for influenza virus infection and to consult surveillance data when evaluating persons with acute respiratory illnesses during influenza season. CDC has published interim guidelines to provide options for treatment or chemoprophylaxis of influenza in the United States if oseltamivir-resistant seasonal influenza A (H1N1) viruses are circulating widely in a community or if the prevalence of oseltamivir-resistant influenza A (H1N1) viruses is uncertain (8). Updated guidance on antiviral use will be available from ACIP before the start of the 2009--10 influenza season. This guidance will include a summary of antiviral resistance data from the 2008--09 influenza season, and will be published separately from the vaccination recommendations. Until the ACIP recommendations for use of antivirals against influenza are published, CDC's previously published recommendations for use of influenza antiviral medications should be consulted for guidance on antiviral use (8). New guidance on clinical management of influenza, including use of antivirals, also is available from the Infectious Diseases Society of America (454).
Sources of Information Regarding Influenza and its Surveillance
Information regarding influenza surveillance, prevention, detection, and control is available at http://www.cdc.gov/flu. During October--May, surveillance information is updated weekly. In addition, periodic updates regarding influenza are published in MMWR (http://www.cdc.gov/mmwr). Additional information regarding influenza vaccine can be obtained by calling 1-800-CDC-INFO (1-800-232-4636). State and local health departments should be consulted about availability of influenza vaccine, access to vaccination programs, information related to state or local influenza activity, reporting of influenza outbreaks and influenza-related pediatric deaths, and advice concerning outbreak control.
Vaccine Adverse Event Reporting System (VAERS)
Adverse events after influenza vaccination should be reported promptly to VAERS at http://vaers.hhs.gov, even if the reporter is unsure whether vaccine caused the event. Clinically significant adverse events that follow vaccination should be reported to VAERS at http://www.vaers.hhs.gov. Reports may be filed securely online or by telephone at 1-800-822-7967 to request reporting forms or other assistance.
National Vaccine Injury Compensation Program
The National Vaccine Injury Compensation Program (VICP), established by the National Childhood Vaccine Injury Act of 1986, as amended, provides a mechanism through which compensation can be paid on behalf of a person determined to have been injured or to have died as a result of receiving a vaccine covered by VICP. The Vaccine Injury Table lists the vaccines covered by VICP and the injuries and conditions (including death) for which compensation might be paid. If the injury or condition is not on the Table, or does not occur within the specified time period on the Table, persons must prove that the vaccine caused the injury or condition.
For a person to be eligible for compensation, the general filing deadlines for injuries require claims to be filed within 3 years after the first symptom of the vaccine injury; for a death, claims must be filed within 2 years of the vaccine-related death and not more than 4 years after the start of the first symptom of the vaccine-related injury from which the death occurred. When a new vaccine is covered by VICP or when a new injury/condition is added to the Table, claims that do not meet the general filing deadlines must be filed within 2 years from the date the vaccine or injury/condition is added to the Table for injuries or deaths that occurred up to 8 years before the Table change. Persons of all ages who receive a VICP-covered vaccine might be eligible to file a claim. Both the intranasal (LAIV) and injectable (TIV) trivalent influenza vaccines are covered under VICP. Additional information about VICP is available at http//www.hrsa.gov/vaccinecompensation or by calling 1-800-338-2382.
Additional Information Regarding Influenza Virus Infection Control Among Specific Populations
Each year, ACIP provides general, annually updated information regarding control and prevention of influenza. Other reports related to controlling and preventing influenza among specific populations (e.g., immunocompromised persons, HCP, hospital patients, pregnant women, children, and travelers) also are available in the following publications:
- CDC. General recommendations on immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP) and the American Academy of Family Physicians (AAFP). MMWR 2006;55(No. RR-15).
- CDC. Influenza vaccination of health-care personnel: recommendations of the Healthcare Infection Control Practices Advisory Committee (HICPAC) and the Advisory Committee on Immunization Practices (ACIP). MMWR 2006;55(No. RR-2).
- CDC. Recommended immunization schedules for persons aged 0--18 years---United States, 2009. MMWR 2009;57:Q1--4.
- CDC. Recommended adult immunization schedule---United States, 2009. MMWR 2009;57:Q1--4.
- CDC. Guidelines for preventing health-care--associated pneumonia, 2003: recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee. MMWR 2003;53(No. RR-3).
- CDC. Respiratory hygiene/cough etiquette in health-care settings. Atlanta, GA: US Department of Health and Human Services, CDC; 2003. Available at http://www.cdc.gov/flu/professionals/infectioncontrol/resphygiene.htm.
- CDC. Prevention and control of vaccine-preventable diseases in long-term--care facilities. Atlanta, GA: US Department of Health and Human Services, CDC; 2006. Available at http://www.cdc.gov/flu/professionals/infection control/longtermcare.htm.
- CDC. Vaccine safety. Atlanta, GA: US Department of Health and Human Services, CDC; 2009. Available at http://www.cdc.gov/vaccinesafety/index.htm.
- American College of Obstetricians and Gynecologists. Influenza vaccination and treatment during pregnancy. ACOG committee opinion no. 305. Obstet Gynecol 2004;104:1125--6.
- American Academy of Pediatrics. 2009 red book: report of the Committee on Infectious Diseases. 29th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2009.
- Bodnar UR, Maloney SA, Fielding KL, et al. Preliminary guidelines for the prevention and control of ILI among passengers and crew members on cruise ships. Atlanta, GA: US Department of Health and Human Services, CDC; 1999. Available at http://wwwn.cdc.gov/travel/files/pre-guidelines-flu-cruise-ships1999.ashx.
- CDC. Infection control guidance for the prevention and control of influenza in acute-care facilities. Atlanta, GA: US Department of Health and Human Services, CDC; 2007. Available at http://www.cdc.gov/flu/professionals/infectioncontrol/health-carefacilities.htm.
- Food and Drug Administration. FDA pandemic influenza preparedness strategic plan. Washington, DC: Food and Drug Administration; 2007. Available at http://www.fda.gov/oc/op/pandemic/strategicplan03_07.html.
- World Health Organization. Recommendations for influenza vaccines. Geneva, Switzerland: World Health Organization; 2007. Available at http://www.who.int/csr/disease/influenza/vaccinerecommendations/en/index.html.
- American Heart Association and American College of Cardiology. Influenza vaccination as secondary prevention for cardiovascular disease. Circulation 2006;114:1549--53. Available at http://circ.ahajournals.org/cgi/content/full/114/14/1549.
Acknowledgments
Assistance in the preparation of this report was provided by Carolyn Bridges, MD, Larisa Gubareva, MD, PhD, Lyn Finelli, DrPH, Amanda Zongrone, Influenza Division; Margaret Coleman, PhD, Gary L. Euler, DrPH, Peng-jun Lu, PhD, Jeanne Santoli, MD, Abigail Shefer, MD, Immunization Services Division; Penina Haber, PhD, Barbara Slade, MD, Immunization Safety Office, National Center for Preparedness, Detection and Control of Infectious Diseases, CDC. Neal Halsey, MD, The Johns Hopkins School of Public Health, and David Hrncir, MD, Southwest Allergy & Asthma Center, San Antonio, Texas, contributed to the hypersensitivity section of this document.
References
- Monto AS, Kioumehr F. The Tecumseh Study of Respiratory Illness. IX. Occurrence of influenza in the community, 1966--1971. Am J Epidemiol 1975;102:553--63.
- Glezen WP, Couch RB. Interpandemic influenza in the Houston area, 1974--76. N Engl J Med 1978;298:587--92.
- Glezen WP, Greenberg SB, Atmar RL, et al. Impact of respiratory virus infections on persons with chronic underlying conditions. JAMA 2000;283:499--505.
- Barker WH. Excess pneumonia and influenza associated hospitalization during influenza epidemics in the United States, 1970--78. Am J Public Health 1986;76:761--5.
- Barker WH, Mullooly JP. Impact of epidemic type A influenza in a defined adult population. Am J Epidemiol 1980;112:798--811.
- Thompson WW, Shay DK, Weintraub E, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA 2003;289:179--86.
- Thompson WW, Shay DK, Weintraub E, et al. Influenza-associated hospitalizations in the United States. JAMA 2004;292:1333--40.
- CDC. CDC issues interim recommendations for the use of influenza antiviral medications in the setting of oseltamivir resistance among circulating influenza A (H1N1) viruses, 2008--09 influenza season. Atlanta, GA: US Department of Health and Human Services, CDC; 2008. Available at http://www2a.cdc.gov/HAN/ArchiveSys/ViewMsgV.asp?AlertNum=00279.
- Dawood FS, Jain S, Finelli L, et al. Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N Engl J Med 2009;360:2605--15.
- Nichol KL, Treanor JJ. Vaccines for seasonal and pandemic influenza. J Infect Dis 2006;194(Suppl 2):S111--8.
- Smith NM, Shay DK. Influenza vaccination for elderly people and their care workers. Lancet 2006;368:1752--3.
- Ellenberg SS, Foulkes MA, Midthun K, et al. Evaluating the safety of new vaccines: summary of a workshop. Am J Public Health 2005;95:800--7.
- Institute of Medicine. Vaccine safety research, data access, and public trust. Washington, DC: National Academies Press; 2005.
- Bartlett DL, Ezzati-Rice TM, Stokley S, et al. Comparison of NIS and NHIS/NIPRCS vaccination coverage estimates. National Immunization Survey. National Health Interview Survey/National Immunization Provider Record Check Study. Am J Prev Med 2001;20:25--7.
- Cox NJ, Subbarao K. Influenza. Lancet 1999;354:1277--82.
- Garten RJ, Davis CT, Russell CA, et al. Antigenic and genetic characteristics of swine-origin 2009 A (H1N1) influenza viruses circulating in humans. Science 2009. Available at http://www.sciencemag.org/cgi/content/abstract/1176225.
- Nelson MI, Viboud C, Simonsen L, et al. Multiple reassortment events in the evolutionary history of H1N1 influenza A virus since 1918. PLoS Pathog 2008;4:e1000012.
- Russell CA, Jones TC, Barr IG, et al. The global circulation of seasonal influenza A (H3N2) viruses. Science 2008;320:340--6.
- Xu X, Lindstrom SE, Shaw MW, et al. Reassortment and evolution of current human influenza A and B viruses. Virus Res 2004;103:55--60.
- Clements ML, Betts RF, Tierney EL, et al. Serum and nasal wash antibodies associated with resistance to experimental challenge with influenza A wild-type virus. J Clin Microbiol 1986;24:157--60.
- Couch RB. Kasel JA. Immunity to influenza in man. Annu Rev Microbiol 1983;37:529--49.
- Cooney MK, Fox JP, Hall CE. The Seattle Virus Watch. VI. Observations of infections with and illness due to parainfluenza, mumps and respiratory syncytial viruses and Mycoplasma pneumoniae. Am J Epidemiol 1975;101:532--51.
- Glezen WP, Taber LH, Frank AL, et al. Risk of primary infection and reinfection with respiratory syncytial virus. Am J Dis Child 1986;140:543--6.
- Glezen WP. Morbidity associated with the major respiratory viruses. Pediatr Ann 1990;19:535--40.
- CDC. Update: novel influenza A (H1N1) virus infections---worldwide, May 6, 2009. MMWR 2009;58:453--8.
- CDC. Update: influenza activity---United States, September 28, 2008--April 4, 2009, and composition of the 2009--10 influenza vaccine. MMWR 2009;58:369--74.
- Simonsen L, Clarke MJ, Williamson GD, et al. The impact of influenza epidemics on mortality: introducing a severity index. Am J Public Health 1997;87:1944--50.
- Mullooly JP, Bridges CB, Thompson WW, et al. Influenza- and RSV-associated hospitalizations among adults. Vaccine 2007;25:846--55.
- O'Brien MA, Uyeki TM, Shay DK, et al. Incidence of outpatient visits and hospitalizations related to influenza in infants and young children. Pediatrics 2004;113:585--93.
- Keren R, Zaoutis TE, Bridges CB, et al. Neurological and neuromuscular disease as a risk factor for respiratory failure in children hospitalized with influenza infection. JAMA 2005;294:2188--94.
- Neuzil KM, Wright PF, Mitchel EF Jr, et al. The burden of influenza illness in children with asthma and other chronic medical conditions. J Pediatr 2000;137:856--64.
- Poehling KA, Edwards KM, Weinberg GA, et al. The underrecognized burden of influenza in young children. N Engl J Med 2006;355:31--40.
- Olson DR, Heffernan RT, Paladini M, et al. Monitoring the impact of influenza by age: emergency department fever and respiratory complaint surveillance in New York City. PLoS Medicine 2007;4:e247.
- Neuzil KM, Zhu Y, Griffin MR, et al. Burden of interpandemic influenza in children younger than 5 years: a 25-year prospective study. J Infect Dis 2002;185:147--52.
- Neuzil KM, Mellen BG, Wright PF, et al. The effect of influenza on hospitalizations, outpatient visits, and courses of antibiotics in children. N Engl J Med 2000;342:225--31.
- Bourgeois FT, Valim C, Wei JC, et al. Influenza and other respiratory virus-related emergency department visits among young children. Pediatrics 2006;118:e1--8.
- Simonsen L, Fukuda K, Schonberger LB, et al. The impact of influenza epidemics on hospitalizations. J Infect Dis 2000;181:831--7.
- Glezen WP, Decker M, Perrotta DM. Survey of underlying conditions of persons hospitalized with acute respiratory disease during influenza epidemics in Houston, 1978--1981. Am Rev Respir Dis 1987;136:550--5.
- Izurieta HS, Thompson WW, Kramarz P, et al. Influenza and the rates of hospitalization for respiratory disease among infants and young children. N Engl J Med 2000;342:232--9.
- Mullooly JP, Barker WH. Impact of type A influenza on children: a retrospective study. Am J Public Health 1982;72:1008--16.
- Ampofo K, Gesteland PH, Bender J, et al. Epidemiology, complications, and cost of hospitalization in children with laboratory-confirmed influenza infection. Pediatrics 2006;118:2409--17.
- Coffin SE, Zaoutis TE, Rosenquist AB, et al. Incidence, complications, and risk factors for prolonged stay in children hospitalized with community-acquired influenza. Pediatrics 2007;119:740--8.
- Iwane MK, Edwards KM, Szilagyi PG, et al. Population-based surveillance for hospitalizations associated with respiratory syncytial virus, influenza virus, and parainfluenza viruses among young children. Pediatrics 2004;113:1758--64.
- Schrag SJ, Shay DK, Gershman K, et al. Multistate surveillance for laboratory-confirmed, influenza-associated hospitalizations in children: 2003--2004. Pediatr Infect Dis J 2006;25:395--400.
- Miller EK, Griffin MR, Edwards KM, et al. Influenza burden for children with asthma. Pediatrics 2008;121:1--8.
- Bhat N, Wright JG, Broder KR, et al. Influenza-associated deaths among children in the United States, 2003--2004. N Engl J Med 2005;353:2559--67.
- Louie JK, Schechter R, Honarmand S, et al. Severe pediatric influenza in California, 2003--2005: implications for immunization recommendations. Pediatrics 2006;117:e610--8.
- CDC. Influenza activity---United States and worldwide, 2007--08 season. MMWR 2008;57:692--7.
- Finelli L, Fiore A, Dhara R, et al. Influenza-associated pediatric mortality in the United States: increase of Staphylococcus aureus coinfection. Pediatrics 2008;122:805--11.
- Creech CB 2nd, Kernodle DS, Alsentzer A, et al. Increasing rates of nasal carriage of methicillin-resistant Staphylococcus aureus in healthy children. Pediatr Infect Dis J 2005;24:617--21.
- CDC. Severe methicillin-resistant Staphylococcus aureus community-acquired pneumonia associated with influenza---Louisiana and Georgia, December 2006--January 2007. MMWR 2007;56:325--9.
- Couch RB. Influenza, influenza virus vaccine, and human immunodeficiency virus infection. Clin Infect Dis 1999;28:548--51.
- Fine AD, Bridges CB, De Guzman AM, et al. Influenza A among patients with human immunodeficiency virus: an outbreak of infection at a residential facility in New York City. Clin Infect Dis 2001;32:1784--91.
- Radwan HM, Cheeseman SH, Lai KK, et al. Influenza in human immunodeficiency virus-infected patients during the 1997--1998 influenza season. Clin Infect Dis 2000;31:604--6.
- Safrin S, Rush JD, Mills J. Influenza in patients with human immunodeficiency virus infection. Chest 1990;98:33--7.
- Tasker SA, O'Brien WA, Treanor JJ, et al. Effects of influenza vaccination in HIV-infected adults: a double-blind, placebo-controlled trial. Vaccine 1998;16:1039--42.
- Neuzil KM, Reed GW, Mitchel EF Jr, et al. Influenza-associated morbidity and mortality in young and middle-aged women. JAMA 1999;281:901--7.
- Lin JC, Nichol KL. Excess mortality due to pneumonia or influenza during influenza seasons among persons with acquired immunodeficiency syndrome. Arch Intern Med 2001;161:441--6.
- Harris J. Influenza occurring in pregnant women: a statistical study of thirteen hundred and fifty cases. JAMA 1919;72:978--80.
- Freeman DW, Barno A. Deaths from Asian influenza associated with pregnancy. Am J Obstet Gynecol 1959;78:1172--5.
- Naleway AL, Smith WJ, Mullooly JP. Delivering influenza vaccine to pregnant women. Epidemiol Rev 2006;28:47--53.
- Widelock D, Csizmas L, Klein S. Influenza, pregnancy, and fetal outcome. Public Health Rep 1963;78:1--11.
- Louria DB, Blumenfeld HL, Ellis JT, et al. Studies on influenza in the pandemic of 1957--1958. II. Pulmonary complications of influenza. J Clin Invest 1959;38:213--65.
- Irving WL, James DK, Stephenson T, et al. Influenza virus infection in the second and third trimesters of pregnancy: a clinical and seroepidemiological study. BJOG 2000;107:1282--9.
- Kirshon B, Faro S, Zurawin RK, et al. Favorable outcome after treatment with amantadine and ribavirin in a pregnancy complicated by influenza pneumonia. A case report. J Reprod Med 1988;33:399--401.
- Neuzil KM, Reed GW, Mitchel EF, et al. Impact of influenza on acute cardiopulmonary hospitalizations in pregnant women. Am J Epidemiol 1998;148:1094--102.
- Schoenbaum SC, Weinstein L. Respiratory infection in pregnancy. Clin Obstet Gynecol 1979;22:293--300.
- Shahab S, Glezen W. Influenza virus. In: Gonik B, ed. Viral diseases in pregnancy. New York, NY: Springer-Verlag; 194:215--23.
- Kort BA, Cefalo RC, Baker VV. Fatal influenza A pneumonia in pregnancy. Am J Perinatol 1986;3:179--82.
- Mullooly JP, Barker WH, Nolan TF Jr. Risk of acute respiratory disease among pregnant women during influenza A epidemics. Public Health Rep 1986;101:205--11.
- Cox S, Posner SF, McPheeters M, et al. Hospitalizations with respiratory illness among pregnant women during influenza season. Obstet Gynecol 2006;107:1315--22.
- Dodds L, McNeil SA, Fell DB, et al. Impact of influenza exposure on rates of hospital admissions and physician visits because of respiratory illness among pregnant women. CMAJ 2007;176:463--8.
- Hartert TV, Neuzil KM, Shintani AK, et al. Maternal morbidity and perinatal outcomes among pregnant women with respiratory hospitalizations during influenza season. Am J Obstet Gynecol 2003;189:1705--12.
- Griffiths PD, Ronalds CJ, Heath RB. A prospective study of influenza infections during pregnancy. J Epidemiol Community Health 1980;34:124--8.
- McGeer A, Green KA, Plevneshi A, et al. Antiviral therapy and outcomes of influenza requiring hospitalization in Ontario, Canada. Clin Infect Dis 2007;45:1568--75.
- Grayson ML, Melvani S, Druce J, et al. Efficacy of soap and water and alcohol-based hand-rub preparations against live H1N1 influenza virus on the hands of human volunteers. Clin Infect Dis 2009;48:285--91.
- Jefferson T, Foxlee R, Del Mar C, et al. Interventions for the interruption or reduction of the spread of respiratory viruses. Cochrane Database Syst Rev 2007:CD006207.
- Luby SP, Agboatwalla M, Feikin DR, et al. Effect of handwashing on child health: a randomised controlled trial. Lancet 2005;366:225--33.
- Inglesby TV, Nuzzo JB, O'Toole T, et al. Disease mitigation measures in the control of pandemic influenza. Biosecur Bioterror 2006;4:366--75.
- Bell DM. Non-pharmaceutical interventions for pandemic influenza, national and community measures. Emerg Infect Dis 2006;12:88--94.
- Nichol KL. Heterogeneity of influenza case definitions and implications for interpreting and comparing study results. Vaccine 2006;24:6726--8.
- Jackson LA, Jackson ML, Nelson JC, et al. Evidence of bias in estimates of influenza vaccine effectiveness in seniors. Int J Epidemiol 2006;35:337--44.
- Simonsen L, Taylor RJ, Viboud C, et al. Mortality benefits of influenza vaccination in elderly people: an ongoing controversy. Lancet Infect Dis 2007;7:658--66.
- WHO. Recommended composition of influenza virus vaccines for use in the 2009--2010 influenza season (northern hemisphere winter). Wkly Epidemiol Rec 2009;84:65--72.
- Kilbourne E. Influenza. New York, NY: Plenum Medical Book Company; 1987.
- Oxford JS, Schild GC, Potter CW, et al. The specificity of the anti-haemagglutinin antibody response induced in man by inactivated influenza vaccines and by natural infection. J Hyg (Lond) 1979;82:51--61.
- Neuzil KM, Dupont WD, Wright PF, et al. Efficacy of inactivated and cold-adapted vaccines against influenza A infection, 1985 to 1990: the pediatric experience. Pediatr Infect Dis J 2001;20:733--40.
- Potter CW, Oxford JS. Determinants of immunity to influenza infection in man. Br Med Bull 1979;35:69--75.
- Hirota Y, Kaji M, Ide S, et al. Antibody efficacy as a keen index to evaluate influenza vaccine effectiveness. Vaccine 1997;15:962--7.
- La Montagne JR, Noble GR, Quinnan GV, et al. Summary of clinical trials of inactivated influenza vaccine---1978. Rev Infect Dis 1983;5:723--36.
- Treanor JWright PF. Immune correlates of protection against influenza in the human challenge model. Dev Biol (Basel) 2003;115:97--104.
- Belshe RB, Nichol KL, Black SB, et al. Safety, efficacy, and effectiveness of live, attenuated, cold-adapted influenza vaccine in an indicated population aged 5--49 years. Clin Infect Dis 2004;39:920--7.
- Daubeney P, Taylor CJ, McGaw J, et al. Immunogenicity and tolerability of a trivalent influenza subunit vaccine (Influvac) in high-risk children aged 6 months to 4 years. Br J Clin Pract 1997;51:87--90.
- Gonzalez M, Pirez MC, Ward E, et al. Safety and immunogenicity of a paediatric presentation of an influenza vaccine. Arch Dis Child 2000;83:488--91.
- Negri E, Colombo C, Giordano L, et al. Influenza vaccine in healthy children: a meta-analysis. Vaccine 2005;23:2851--61.
- Wright PF, Cherry JD, Foy HM, et al. Antigenicity and reactogenicity of influenza A/USSR/77 virus vaccine in children---a multicentered evaluation of dosage and safety. Rev Infect Dis 1983;5:758--64.
- Wright PF, Thompson J, Vaughn WK, et al. Trials of influenza A/New Jersey/76 virus vaccine in normal children: an overview of age-related antigenicity and reactogenicity. J Infect Dis 1977;136(Suppl)S731--41.
- Neuzil KM, Jackson LA, Nelson J, et al. Immunogenicity and reactogenicity of 1 versus 2 doses of trivalent inactivated influenza vaccine in vaccine-naive 5--8--year-old children. J Infect Dis 2006;194:1032--9.
- Walter EB, Neuzil KM, Zhu Y, et al. Influenza vaccine immunogenicity in 6-- to 23--month-old children: are identical antigens necessary for priming? Pediatrics 2006;118:e570--8.
- Englund JA, Walter EB, Gbadebo A, et al. Immunization with trivalent inactivated influenza vaccine in partially immunized toddlers. Pediatrics 2006;118:e579--85.
- Englund JA, Walter EB, Fairchok MP, et al. A comparison of 2 influenza vaccine schedules in 6-- to 23--month-old children. Pediatrics 2005;115:1039--47.
- Allison MA, Daley MF, Crane LA, et al. Influenza vaccine effectiveness in healthy 6-- to 21--month-old children during the 2003--2004 season. J Pediatr 2006;149:755--762.
- Bell TD, Chai H, Berlow B, et al. Immunization with killed influenza virus in children with chronic asthma. Chest 1978;73:140--5.
- Groothuis JR, Lehr MV, Levin MJ. Safety and immunogenicity of a purified haemagglutinin antigen in very young high-risk children. Vaccine 1994;12:139--41.
- Park CL, Frank AL, Sullivan M, et al. Influenza vaccination of children during acute asthma exacerbation and concurrent prednisone therapy. Pediatrics 1996;98:196--200.
- Ritzwoller DP, Bridges CB, Shetterly S, et al. Effectiveness of the 2003--2004 influenza vaccine among children 6 months to 8 years of age, with 1 vs 2 doses. Pediatrics 2005;116:153--9.
- Shuler CM, Iwamoto M, Bridges CB, et al. Vaccine effectiveness against medically attended, laboratory-confirmed influenza among children aged 6 to 59 months, 2003--2004. Pediatrics 2007;119:e587--95.
- Clover RD, Crawford S, Glezen WP, et al. Comparison of heterotypic protection against influenza A/Taiwan/86 (H1N1) by attenuated and inactivated vaccines to A/Chile/83--like viruses. J Infect Dis 1991;163:300--4.
- Hoberman A, Greenberg DP, Paradise JL, et al. Effectiveness of inactivated influenza vaccine in preventing acute otitis media in young children: a randomized controlled trial. JAMA 2003;290:1608--16.
- Eisenberg KW, Szilagyi PG, Fairbrother G, et al. Vaccine effectiveness against laboratory-confirmed influenza in children 6 to 59 months of age during the 2003--2004 and 2004--2005 influenza seasons. Pediatrics 2008;122:911--9.
- Zangwill KM, Belshe RB. Safety and efficacy of trivalent inactivated influenza vaccine in young children: a summary for the new era of routine vaccination. Pediatr Infect Dis J 2004;23:189--97.
- Jefferson T, Rivetti A, Harnden A, et al. Vaccines for preventing influenza in healthy children. Cochrane Database Syst Rev 2008:CD004879.
- Sugaya N, Nerome K, Ishida M, et al. Efficacy of inactivated vaccine in preventing antigenically drifted influenza type A and well-matched type B. JAMA 1994;272:1122--6.
- Kramarz P, Destefano F, Gargiullo PM, et al. Does influenza vaccination prevent asthma exacerbations in children? J Pediatr 2001;138:306--10..
- Bueving HJ, Bernsen RM, de Jongste JC, et al. Influenza vaccination in children with asthma: randomized double-blind placebo-controlled trial. Am J Respir Crit Care Med 2004;169:488--93.
- Clements DA, Langdon L, Bland C, et al. Influenza A vaccine decreases the incidence of otitis media in 6-- to 30--month-old children in day care. Arch Pediatr Adolesc Med 1995;149:1113--7.
- Heikkinen T, Ruuskanen O, Waris M, et al. Influenza vaccination in the prevention of acute otitis media in children. Am J Dis Child 1991;145:445--8.
- Gross PA, Weksler ME, Quinnan GV Jr, et al. Immunization of elderly people with two doses of influenza vaccine. J Clin Microbiol 1987;25:1763--5.
- Feery BJ, Cheyne IM, Hampson AW, et al. Antibody response to one and two doses of influenza virus subunit vaccine. Med J Aust 1976;1:186, 188--9.
- Levine M, Beattie BL, McLean DM. Comparison of one- and two-dose regimens of influenza vaccine for elderly men. CMAJ 1987;137:722--6.
- Bridges CB, Thompson WW, Meltzer MI, et al. Effectiveness and cost-benefit of influenza vaccination of healthy working adults: a randomized controlled trial. JAMA 2000;284:1655--63.
- Jefferson TO, Rivetti D, Di Pietrantonj C, et al. Vaccines for preventing influenza in healthy adults. Cochrane Database Syst Rev 2007:CD001269.
- Nichol KL, Lind A, Margolis KL, et al. The effectiveness of vaccination against influenza in healthy, working adults. N Engl J Med 1995;333:889--93.
- Campbell DS, Rumley MH. Cost-effectiveness of the influenza vaccine in a healthy, working-age population. J Occup Environ Med 1997;39:408--14.
- Herrera GA, Iwane MK, Cortese M, et al. Influenza vaccine effectiveness among 50--64--year-old persons during a season of poor antigenic match between vaccine and circulating influenza virus strains: Colorado, United States, 2003--2004. Vaccine 2007;25:154--60.
- Blumberg EA, Albano C, Pruett T, et al. The immunogenicity of influenza virus vaccine in solid organ transplant recipients. Clin Infect Dis 1996;22:295--302.
- Dorrell L, Hassan I, Marshall S, et al. Clinical and serological responses to an inactivated influenza vaccine in adults with HIV infection, diabetes, obstructive airways disease, elderly adults and healthy volunteers. Int J STD AIDS 1997;8:776--9.
- Wongsurkiat P, Maranetra KN, Wasi C, et al. Acute respiratory illness in patients with COPD and the effectiveness of influenza vaccination. Chest 2004;125:2011--20.
- Gurfinkel EP, Leon de la Fuente R, Mendiz O, et al. Flu vaccination in acute coronary syndromes and planned percutaneous coronary interventions (FLUVACS) Study. Eur Heart J 2004;25:25--31.
- Ciszewski A, Bilinska ZT, Brydak LB, et al. Influenza vaccination in secondary prevention from coronary ischaemic events in coronary artery disease: FLUCAD study. Eur Heart J 2008;29:1350--8.
- Hak E, Buskens E, van Essen GA, et al. Clinical effectiveness of influenza vaccination in persons younger than 65 years with high-risk medical conditions: the PRISMA study. Arch Intern Med 2005;165:274--80.
- Hak E, Buskens E, Nichol KL, et al. Do recommended high-risk adults benefit from a first influenza vaccination? Vaccine 2006;24:2799--802.
- Looijmans-Van den Akker I, Verheij TJ, Buskens E, et al. Clinical effectiveness of first and repeat influenza vaccination in adult and elderly diabetic patients. Diabetes Care 2006;29:1771--6.
- Cates CJ, Jefferson TO, Rowe BH. Vaccines for preventing influenza in people with asthma. Cochrane Database Syst Rev 2008:CD000364.
- Poole PJ, Chacko E, Wood-Baker RW, et al. Influenza vaccine for patients with chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2006:CD002733.
- Chadwick EG, Chang G, Decker MD, et al. Serologic response to standard inactivated influenza vaccine in human immunodeficiency virus-infected children. Pediatr Infect Dis J 1994;13:206--11.
- Huang KL, Ruben FL, Rinaldo CR Jr, et al. Antibody responses after influenza and pneumococcal immunization in HIV-infected homosexual men. JAMA 1987;257:2047--50.
- Staprans SI, Hamilton BL, Follansbee SE, et al. Activation of virus replication after vaccination of HIV-1--infected individuals. J Exp Med 1995;182:1727--37.
- Kroon FP, van Dissel JT, de Jong JC, et al. Antibody response after influenza vaccination in HIV-infected individuals: a consecutive 3--year study. Vaccine 2000;18:3040--9.
- Miotti PG, Nelson KE, Dallabetta GA, et al. The influence of HIV infection on antibody responses to a two-dose regimen of influenza vaccine. JAMA 1989;262:779--83.
- Scharpe J, Evenepoel P, Maes B, et al. Influenza vaccination is efficacious and safe in renal transplant recipients. Am J Transplant 2008;8:332--7.
- Edvardsson VO, Flynn JT, Deforest A, et al. Effective immunization against influenza in pediatric renal transplant recipients. Clin Transplant 1996;10:556--60.
- Fraund S, Wagner D, Pethig K, et al. Influenza vaccination in heart transplant recipients. J Heart Lung Transplant 1999;18:220--5.
- Lawal A, Basler C, Branch A, et al. Influenza vaccination in orthotopic liver transplant recipients: absence of post administration ALT elevation. Am J Transplant 2004;4:1805--9.
- Madan RP, Tan M, Fernandez-Sesma A, et al. A prospective, comparative study of the immune response to inactivated influenza vaccine in pediatric liver transplant recipients and their healthy siblings. Clin Infect Dis 2008;46:712--8.
- Duchini A, Hendry RM, Nyberg LM, et al. Immune response to influenza vaccine in adult liver transplant recipients. Liver Transpl 2001;7:311--3.
- Sumaya CV, Gibbs RS. Immunization of pregnant women with influenza A/New Jersey/76 virus vaccine: reactogenicity and immunogenicity in mother and infant. J Infect Dis 1979;140:141--6.
- Munoz FM, Greisinger AJ, Wehmanen OA, et al. Safety of influenza vaccination during pregnancy. Am J Obstet Gynecol 2005;192:1098--106.
- Englund JA, Mbawuike IN, Hammill H, et al. Maternal immunization with influenza or tetanus toxoid vaccine for passive antibody protection in young infants. J Infect Dis 1993;168:647--56.
- Puck JM, Glezen WP, Frank AL, et al. Protection of infants from infection with influenza A virus by transplacentally acquired antibody. J Infect Dis 1980;142:844--9.
- Reuman PD, Ayoub EM, Small PA. Effect of passive maternal antibody on influenza illness in children: a prospective study of influenza A in mother-infant pairs. Pediatr Infect Dis J 1987;6:398--403.
- Black SB, Shinefield HR, France EK, et al. Effectiveness of influenza vaccine during pregnancy in preventing hospitalizations and outpatient visits for respiratory illness in pregnant women and their infants. Am J Perinatol 2004;21:333--9.
- France EK, Smith-Ray R, McClure D, et al. Impact of maternal influenza vaccination during pregnancy on the incidence of acute respiratory illness visits among infants. Arch Pediatr Adolesc Med 2006;160:1277--83.
- Zaman K, Roy E, Arifeen SE, et al. Effectiveness of maternal influenza immunization in mothers and infants. N Engl J Med 2008;359:1555--64.
- McElhaney JE. The unmet need in the elderly: designing new influenza vaccines for older adults. Vaccine 2005;23(Suppl 1):S10--25.
- Goodwin K, Viboud C, Simonsen L. Antibody response to influenza vaccination in the elderly: a quantitative review. Vaccine 2006;24:1159--69.
- Skowronski DM, Tweed SA, De Serres G. Rapid decline of influenza vaccine-induced antibody in the elderly: is it real, or is it relevant? J Infect Dis 2008;197:490--502.
- Berry BB, Ehlert DA, Battiola RJ, et al. Influenza vaccination is safe and immunogenic when administered to hospitalized patients. Vaccine 2001;19:3493--8.
- Govaert TM, Thijs CT, Masurel N, et al. The efficacy of influenza vaccination in elderly individuals. A randomized double-blind placebo-controlled trial. JAMA 1994;272:1661--5.
- Thijs C, Beyer WE, Govaert PM, et al. Mortality benefits of influenza vaccination in elderly people. Lancet Infect Dis 2008;8:460--1; author reply 463--5.
- Monto AS, Hornbuckle K, Ohmit SE. Influenza vaccine effectiveness among elderly nursing home residents: a cohort study. Am J Epidemiol 2001;154:155--60.
- Ohmit SE, Arden NH, Monto AS. Effectiveness of inactivated influenza vaccine among nursing home residents during an influenza type A (H3N2) epidemic. J Am Geriatr Soc 1999;47:165--71.
- Coles FB, Balzano GJ, Morse DL. An outbreak of influenza A (H3N2) in a well immunized nursing home population. J Am Geriatr Soc 1992;40:589--92.
- Libow LS, Neufeld RR, Olson E, et al. Sequential outbreak of influenza A and B in a nursing home: efficacy of vaccine and amantadine. J Am Geriatr Soc 1996;44:1153--7.
- Arden N, Patriarcha P, Kendal A. Experiences in the use and efficacy of inactivated influenza vaccine in nursing homes. In: Kendal A, Patriarca P, eds. Options for the control of influenza. New York, NY: Alan R. Liss, Inc.; 1986.
- Jefferson T, Rivetti D, Rivetti A, et al. Efficacy and effectiveness of influenza vaccines in elderly people: a systematic review. Lancet 2005;366:1165--74.
- Patriarca PA, Weber JA, Parker RA, et al. Efficacy of influenza vaccine in nursing homes. Reduction in illness and complications during an influenza A (H3N2) epidemic. JAMA 1985;253:1136--9.
- Nichol KL, Wuorenma J, von Sternberg T. Benefits of influenza vaccination for low-, intermediate-, and high-risk senior citizens. Arch Intern Med 1998;158:1769--76.
- Mullooly JP, Bennett MD, Hornbrook MC, et al. Influenza vaccination programs for elderly persons: cost-effectiveness in a health maintenance organization. Ann Intern Med 1994;121:947--52.
- Nichol KL, Nordin JD, Nelson DB, et al. Effectiveness of influenza vaccine in the community-dwelling elderly. N Engl J Med 2007;357:1373--81.
- Gross PA, Hermogenes AW, Sacks HS, et al. The efficacy of influenza vaccine in elderly persons. A meta-analysis and review of the literature. Ann Intern Med 1995;123:518--27.
- Hak E, Nordin J, Wei F, et al. Influence of high-risk medical conditions on the effectiveness of influenza vaccination among elderly members of 3 large managed-care organizations. Clin Infect Dis 2002;35:370--7.
- Nordin J, Mullooly J, Poblete S, et al. Influenza vaccine effectiveness in preventing hospitalizations and deaths in persons 65 years or older in Minnesota, New York, and Oregon: data from 3 health plans. J Infect Dis 2001;184:665--70.
- Patriarca PA, Weber JA, Parker RA, et al. Risk factors for outbreaks of influenza in nursing homes. A case-control study. Am J Epidemiol 1986;124:114--9.
- Jackson LA, Nelson JC, Benson P, et al. Functional status is a confounder of the association of influenza vaccine and risk of all cause mortality in seniors. Int J Epidemiol 2006;35:345--52.
- Simonsen L, Viboud C, Taylor RJ. Effectiveness of influenza vaccination. N Engl J Med 2007;357:2729--30;author reply 2730--1.
- Nelson JC, Jackson ML, Jackson LA. Effectiveness of influenza vaccination. N Engl J Med 2007;357:2728--9; author reply 2730--1.
- Poland GA, Borrud A, Jacobson RM, et al. Determination of deltoid fat pad thickness. Implications for needle length in adult immunization. JAMA 1997;277:1709--11.
- CDC. General recommendations on immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 2006;55(No. RR-15).
- France EK, Glanz JM, Xu S, et al. Safety of the trivalent inactivated influenza vaccine among children: a population-based study. Arch Pediatr Adolesc Med 2004;158:1031--6.
- Hambidge SJ, Glanz JM, France EK, et al. Safety of trivalent inactivated influenza vaccine in children 6 to 23 months old. JAMA 2006;296:1990--7.
- Scheifele DW, Bjornson G, Johnston J. Evaluation of adverse events after influenza vaccination in hospital personnel. CMAJ 1990;142:127--30.
- Barry DW, Mayner RE, Hochstein HD, et al. Comparative trial of influenza vaccines. II. Adverse reactions in children and adults. Am J Epidemiol 1976;104:47--59.
- McMahon AW, Iskander JK, Haber P, et al. Inactivated influenza vaccine (IIV) in children <2 years of age: examination of selected adverse events reported to the Vaccine Adverse Event Reporting System (VAERS) after thimerosal-free or thimerosal-containing vaccine. Vaccine 2008;26:427--9.
- Rosenberg M, Sparks R, McMahon A, et al. Serious adverse events rarely reported after trivalent inactivated influenza vaccine (TIV) in children 6--23 months of age. Vaccine 2009.
- Govaert TM, Dinant GJ, Aretz K, et al. Adverse reactions to influenza vaccine in elderly people: randomised double blind placebo controlled trial. BMJ 1993;307:988--90.
- Margolis KL, Nichol KL, Poland GA, et al. Frequency of adverse reactions to influenza vaccine in the elderly. A randomized, placebo-controlled trial. JAMA 1990;264:1139--41.
- Nichol KL, Margolis KL, Lind A, et al. Side effects associated with influenza vaccination in healthy working adults. A randomized, placebo-controlled trial. Arch Intern Med 1996;156:1546--50.
- Vellozzi C, Burwen DR, Dobardzic A, et al. Safety of trivalent inactivated influenza vaccines in adults: background for pandemic influenza vaccine safety monitoring. Vaccine 2009;27:2114--20.
- Heinonen OP, Shapiro S, Monson RR, et al. Immunization during pregnancy against poliomyelitis and influenza in relation to childhood malignancy. Int J Epidemiol 1973;2:229--35.
- Pool V, Iskander J. Safety of influenza vaccination during pregnancy. Am J Obstet Gynecol 2006;194:1200;author reply 1201.
- Deinard AS, Ogburn P Jr. A/NJ/8/76 influenza vaccination program: effects on maternal health and pregnancy outcome. Am J Obstet Gynecol 1981;140:240--5.
- Mak TK, Mangtani P, Leese J, et al. Influenza vaccination in pregnancy: current evidence and selected national policies. Lancet Infect Dis 2008;8:44--52.
- Centers TALAAR. The safety of inactivated influenza vaccine in adults and children with asthma. N Engl J Med 2001;345:1529--36.
- Kmiecik T, Arnoux S, Kobryn A, Gorski P. Influenza vaccination in adults with asthma: safety of an inactivated trivalent influenza vaccine. J Asthma 2007;44:817--22.196. Groothuis JR, Levin MJ, Rabalais GP, et al. Immunization of high-risk infants younger than 18 months of age with split-product influenza vaccine. Pediatrics 1991;87:823--8.
- Ho DD. HIV-1 viraemia and influenza. Lancet 1992;339:1549.
- O'Brien WA, Grovit-Ferbas K, Namazi A, et al. Human immunodeficiency virus-type 1 replication can be increased in peripheral blood of seropositive patients after influenza vaccination. Blood 1995;86:1082--9.
- Glesby MJ, Hoover DR, Farzadegan H, et al. The effect of influenza vaccination on human immunodeficiency virus type 1 load: a randomized, double-blind, placebo-controlled study. J Infect Dis 1996;174:1332--6.
- Fowke KR, D'Amico R, Chernoff DN, et al. Immunologic and virologic evaluation after influenza vaccination of HIV-1--infected patients. AIDS 1997;11:1013--21.
- Fuller JD, Craven DE, Steger KA, et al. Influenza vaccination of human immunodeficiency virus (HIV)-infected adults: impact on plasma levels of HIV type 1 RNA and determinants of antibody response. Clin Infect Dis 1999;28:541--7.
- Amendola A, Boschini A, Colzani D, et al. Influenza vaccination of HIV-1--positive and HIV-1--negative former intravenous drug users. J Med Virol 2001;65:644--8.
- Sullivan PS, Hanson DL, Dworkin MS, et al. Effect of influenza vaccination on disease progression among HIV-infected persons. AIDS 2000;14:2781--5.
- Gunthard HF, Wong JK, Spina CA, et al. Effect of influenza vaccination on viral replication and immune response in persons infected with human immunodeficiency virus receiving potent antiretroviral therapy. J Infect Dis 2000;181:522--31.
- Wood RA, Berger M, Dreskin SC, et al. An algorithm for treatment of patients with hypersensitivity reactions after vaccines. Pediatrics 2008;122:e771--7.
- Ruggeberg JU, Gold MS, Bayas JM, et al. Anaphylaxis: case definition and guidelines for data collection, analysis, and presentation of immunization safety data. Vaccine 2007;25:5675--84.
- GlaxoSmithKline Biologicals. FLULAVAL [Prescribing information]. Research Triangle Park, NC: GlaxoSmithKline; 2008.
- CSL Biotherapies Inc. AFLURIA [Prescribing information]. King of Prussia, PA: CSL Biotherapies Inc.; 2008.
- Grabenstein JD. Clinical management of hypersensitivities to vaccine components. Hosp Pharm 1997;32:77--87.
- Bohlke K, Davis RL, Marcy SM, et al. Risk of anaphylaxis after vaccination of children and adolescents. Pediatrics 2003;112:815--20.
- Tey D, Heine RG. Egg allergy in childhood: an update. Curr Opin Allergy Clin Immunol 2009;9:244--50.
- James JM, Zeiger RS, Lester MR, et al. Safe administration of influenza vaccine to patients with egg allergy. J Pediatr 1998;133:624--8.
- Murphy KR, Strunk RC. Safe administration of influenza vaccine in asthmatic children hypersensitive to egg proteins. J Pediatr 1985;106:931--3.
- Zeiger RS. Current issues with influenza vaccination in egg allergy. J Allergy Clin Immunol 2002;110:834--40.
- Zheng W, Dreskin SC. Thimerosal in influenza vaccine: an immediate hypersensitivity reaction. Ann Allergy Asthma Immunol 2007;99:574--5.
- Aberer W. Vaccination despite thimerosal sensitivity. Contact Dermatitis 1991;24:6--10.
- Kirkland LR. Ocular sensitivity to thimerosal: a problem with hepatitis B vaccine? South Med J 1990;83:497--9.
- GlaxoSmithKline Biologicals. FLUARIX [Prescribing information]. Research Triangle Park, NC: GlaxoSmithKline; 2008.
- Sanofi Pasteur Inc. Fluzone [Prescribing information]. Swiftwater, PA: Sanofi Pasteur Inc.; 2008.
- Novartis. FLUVIRIN [Prescribing information]. Emeryville, CA: Novartis; 2008.
- Ohmit SE, Victor JC, Teich ER, et al. Prevention of symptomatic seasonal influenza in 2005--2006 by inactivated and live attenuated vaccines. J Infect Dis 2008;198:312--7.
- Ohmit SE, Victor JC, Rotthoff JR, et al. Prevention of antigenically drifted influenza by inactivated and live attenuated vaccines. N Engl J Med 2006;355:2513--22.
- National Advisory Committee on Immunization. An Advisory Committee Statement (ACS). Supplementary statement on influenza vaccination: continued use of Fluviral influenza vaccine in the 2000--2001 season. Can Commun Dis Rep 2001;27:1--3.
- Boulianne N, De Serres G, Duval B, et al. Clinical manifestations and incidence of oculo-respiratory syndrome following influenza vaccination---Quebec, 2000. Can Commun Dis Rep 2001;27:85--90.
- Spila-Alegiani S, Salmaso S, Rota MC, et al. Reactogenicity in the elderly of nine commercial influenza vaccines: results from the Italian SVEVA study. Study for the evaluation of adverse events of influenza vaccination. Vaccine 1999;17:1898--904.
- Anonymous. Oculo-respiratory syndrome following influenza vaccination: review of post-marketing surveillance through four influenza seasons in Canada. Can Commun Dis Rep 2005;31:217--25.
- Khromova A PV, Chen R. Oculo-respiratory syndrome following influenza vaccine---United States, 1990--2002: new or previously unrecognized? [Presentation].1st international conference on therapeutic risk management and 19th international conference on pharmacoepidemiology. Philadelphia, PA, USA; August 21--23, 2003.
- Skowronski DM, De Serres G, Hebert J, et al. Skin testing to evaluate oculo-respiratory syndrome (ORS) associated with influenza vaccination during the 2000--2001 season. Vaccine 2002;20:2713--9.
- Scheifele DW, Duval B, Russell ML, et al. Ocular and respiratory symptoms attributable to inactivated split influenza vaccine: evidence from a controlled trial involving adults. Clin Infect Dis 2003;36:850--7.
- Skowronski DM, Strauss B, Kendall P, et al. Low risk of recurrence of oculorespiratory syndrome following influenza revaccination. CMAJ 2002;167:853--8.
- De Serres G, Skowronski DM, Guay M, et al. Recurrence risk of oculorespiratory syndrome after influenza vaccination: randomized controlled trial of previously affected persons. Arch Intern Med 2004;164:2266--72.
- Ropper AH. The Guillain-Barre syndrome. N Engl J Med 1992;326:1130--6.
- Guarino M, Casmiro M, D'Alessandro R. Campylobacter jejuni infection and Guillain-Barre syndrome: a case-control study. Emilia-Romagna Study Group on Clinical and Epidemiological problems in neurology. Neuroepidemiology 1998;17:296--302.
- Jacobs BC, Rothbarth PH, van der Meche FG, et al. The spectrum of antecedent infections in Guillain-Barre syndrome: a case-control study. Neurology 1998;51:1110--5.
- Sheikh KA, Nachamkin I, Ho TW, et al. Campylobacter jejuni lipopolysaccharides in Guillain-Barre syndrome: molecular mimicry and host susceptibility. Neurology 1998;51:371--8.
- Sivadon-Tardy V, Orlikowski D, Porcher R, et al. Guillain-Barre syndrome and influenza virus infection. Clin Infect Dis 2009;48:48--56.
- Haber P, DeStefano F, Angulo FJ, et al. Guillain-Barre syndrome following influenza vaccination. JAMA 2004;292:2478--81.
- Lasky T, Terracciano GJ, Magder L, et al. The Guillain-Barre syndrome and the 1992--1993 and 1993--1994 influenza vaccines. N Engl J Med 1998;339:1797--802.
- Schonberger LB, Bregman DJ, Sullivan-Bolyai JZ, et al. Guillain-Barre syndrome following vaccination in the National Influenza Immunization Program, United States, 1976--1977. Am J Epidemiol 1979;110:105--23.
- Hurwitz ES, Schonberger LB, Nelson DB, et al. Guillain-Barre syndrome and the 1978--1979 influenza vaccine. N Engl J Med 1981;304:1557--61.
- Kaplan JE, Katona P, Hurwitz ES, et al. Guillain-Barre syndrome in the United States, 1979--1980 and 1980--1981. Lack of an association with influenza vaccination. JAMA 1982;248:698--700.
- Chen R, Kent J, Rhodes P, et al. Investigations of a possible association between influenza vaccination and Guillain-Barre syndrome in the United States, 1990--1991 [Abstract 040]. Post Marketing Surveillance 1992;6:5--6.
- Juurlink DN, Stukel TA, Kwong J, et al. Guillain-Barre syndrome after influenza vaccination in adults: a population-based study. Arch Intern Med 2006;166:2217--21.
- Tam CC, O'Brien SJ, Petersen I, et al. Guillain-Barre syndrome and preceding infection with campylobacter, influenza and Epstein-Barr virus in the general practice research database. PLoS ONE 2007;2:e344.
- Hughes RA, Charlton J, Latinovic R, et al. No association between immunization and Guillain-Barre syndrome in the United Kingdom, 1992 to 2000. Arch Intern Med 2006;166:1301--4.
- Stowe J, Andrews N, Wise L, et al. Investigation of the temporal association of Guillain-Barre syndrome with influenza vaccine and influenzalike illness using the United Kingdom General Practice Research Database. Am J Epidemiol 2009;169:382--8.
- Pritchard J, Mukherjee R, Hughes RA. Risk of relapse of Guillain-Barre syndrome or chronic inflammatory demyelinating polyradiculoneuropathy following immunisation. J Neurol Neurosurg Psychiatry 2002;73:348--9.
- CDC. Recommendations regarding the use of vaccines that contain thimerosal as a preservative. MMWR 1999;48:996--8.
- CDC. Summary of the joint statement on thimerosal in vaccines. MMWR 2000;49:622, 631.
- McCormick M, Bayer R, Berg A, et al. Report of the Institute of Medicine. Immunization safety review: vaccines and autism. Washington, DC: Institute of Medicine; 2004.
- Pichichero ME, Cernichiari E, Lopreiato J, et al. Mercury concentrations and metabolism in infants receiving vaccines containing thiomersal: a descriptive study. Lancet 2002;360:1737--41.
- Verstraeten T, Davis RL, DeStefano F, et al. Safety of thimerosal-containing vaccines: a two-phased study of computerized health maintenance organization databases. Pediatrics 2003;112:1039--48.
- Tozzi AE, Bisiacchi P, Tarantino V, et al. Neuropsychological performance 10 years after immunization in infancy with thimerosal-containing vaccines. Pediatrics 2009;123:475--82.
- Schechter R, Grether JK. Continuing increases in autism reported to California's developmental services system: mercury in retrograde. Arch Gen Psychiatry 2008;65:19--24.
- Pichichero ME, Gentile A, Giglio N, et al. Mercury levels in newborns and infants after receipt of thimerosal-containing vaccines. Pediatrics 2008;121:e208--14.
- Thompson WW, Price C, Goodson B, et al. Early thimerosal exposure and neuropsychological outcomes at 7 to 10 years. N Engl J Med 2007;357:1281--92.
- Parker SK, Schwartz B, Todd J, Pickering LK. Thimerosal-containing vaccines and autistic spectrum disorder: a critical review of published original data. Pediatrics 2004;114:793--804.
- Croen LA, Matevia M, Yoshida CK, et al. Maternal Rh D status, anti-D immune globulin exposure during pregnancy, and risk of autism spectrum disorders. Am J Obstet Gynecol 2008;199:234 e1--6.
- Stratton K, Gable A, McCormick M., eds. Report of the Institute of Medicine. Immunization safety review: thimerosal-containing vaccines and neurodevelopmental disorders. Washington, DC: Institute of Medicine; 2001.
- Gostin LO. Medical countermeasures for pandemic influenza: ethics and the law. JAMA 2006;295:554--6.
- Medimmune Vaccines, Inc. FluMist [Package insert]. Gaithersburg, MD: Medimmune Vaccines, Inc.; 2007.
- Vesikari T, Karvonen A, Korhonen T, et al. A randomized, double-blind study of the safety, transmissibility and phenotypic and genotypic stability of cold-adapted influenza virus vaccine. Pediatr Infect Dis J 2006;25:590--5.
- Block SL, Yogev R, Hayden FG, et al. Shedding and immunogenicity of live attenuated influenza vaccine virus in subjects 5--49 years of age. Vaccine 2008;26:4940--6.
- Talbot TR, Crocker DD, Peters J, et al. Duration of virus shedding after trivalent intranasal live attenuated influenza vaccination in adults. Infect Control Hosp Epidemiol 2005;26:494--500.
- Ali T, Scott N, Kallas W, et al. Detection of influenza antigen with rapid antibody-based tests after intranasal influenza vaccination (FluMist). Clin Infect Dis 2004;38:760--2.
- King JC Jr, Treanor J, Fast PE, et al. Comparison of the safety, vaccine virus shedding, and immunogenicity of influenza virus vaccine, trivalent, types A and B, live cold-adapted, administered to human immunodeficiency virus (HIV)-infected and non-HIV-infected adults. J Infect Dis 2000;181:725--8.
- King JC Jr, Fast PE, Zangwill KM, et al. Safety, vaccine virus shedding and immunogenicity of trivalent, cold-adapted, live attenuated influenza vaccine administered to human immunodeficiency virus-infected and noninfected children. Pediatr Infect Dis J 2001;20:1124--31.
- Cha TA, Kao K, Zhao J, et al. Genotypic stability of cold-adapted influenza virus vaccine in an efficacy clinical trial. J Clin Microbiol 2000;38:839--45.
- Buonagurio DA, O'Neill RE, Shutyak L, et al. Genetic and phenotypic stability of cold-adapted influenza viruses in a trivalent vaccine administered to children in a day care setting. Virology 2006;347:296--306.
- King JC Jr, Lagos R, Bernstein DI, et al. Safety and immunogenicity of low and high doses of trivalent live cold-adapted influenza vaccine administered intranasally as drops or spray to healthy children. J Infect Dis 1998;177:1394--7.
- Lee MS, Mahmood K, Adhikary L, et al. Measuring antibody responses to a live attenuated influenza vaccine in children. Pediatr Infect Dis J 2004;23:852--6.
- Zangwill KM, Droge J, Mendelman P, et al. Prospective, randomized, placebo-controlled evaluation of the safety and immunogenicity of three lots of intranasal trivalent influenza vaccine among young children. Pediatr Infect Dis J 2001;20:740--6.
- Nolan T, Lee MS, Cordova JM, et al. Safety and immunogenicity of a live-attenuated influenza vaccine blended and filled at two manufacturing facilities. Vaccine 2003;21:1224--31.
- Bernstein DI, Yan L, Treanor J, et al. Effect of yearly vaccinations with live, attenuated, cold-adapted, trivalent, intranasal influenza vaccines on antibody responses in children. Pediatr Infect Dis J 2003;22:28--34.
- Belshe RB, Gruber WC, Mendelman PM, et al. Correlates of immune protection induced by live, attenuated, cold-adapted, trivalent, intranasal influenza virus vaccine. J Infect Dis 2000;181:1133--7.
- Boyce TG, Gruber WC, Coleman-Dockery SD, et al. Mucosal immune response to trivalent live attenuated intranasal influenza vaccine in children. Vaccine 1999;18:82--8.
- Belshe RB, Mendelman PM, Treanor J, et al. The efficacy of live attenuated, cold-adapted, trivalent, intranasal influenzavirus vaccine in children. N Engl J Med 1998;338:1405--12.
- Belshe RB, Gruber WC, Mendelman PM, et al. Efficacy of vaccination with live attenuated, cold-adapted, trivalent, intranasal influenza virus vaccine against a variant (A/Sydney) not contained in the vaccine. J Pediatr 2000;136:168--75.
- Belshe RB, Gruber WC. Prevention of otitis media in children with live attenuated influenza vaccine given intranasally. Pediatr Infect Dis J 2000;19:S66--71.
- Vesikari T, Fleming DM, Aristegui JF, et al. Safety, efficacy, and effectiveness of cold-adapted influenza vaccine-trivalent against community-acquired, culture-confirmed influenza in young children attending day care. Pediatrics 2006;118:2298--312.
- Tam JS, Capeding MR, Lum LC, et al. Efficacy and safety of a live attenuated, cold-adapted influenza vaccine, trivalent against culture-confirmed influenza in young children in Asia. Pediatr Infect Dis J 2007;26:619--28.
- Gaglani MJ, Piedra PA, Herschler GB, et al. Direct and total effectiveness of the intranasal, live-attenuated, trivalent cold-adapted influenza virus vaccine against the 2000--2001 influenza A(H1N1) and B epidemic in healthy children. Arch Pediatr Adolesc Med 2004;158:65--73.
- Nichol KL, Mendelman PM, Mallon KP, et al. Effectiveness of live, attenuated intranasal influenza virus vaccine in healthy, working adults: a randomized controlled trial. JAMA 1999;282:137--44.
- Redding G, Walker RE, Hessel C, et al. Safety and tolerability of cold-adapted influenza virus vaccine in children and adolescents with asthma. Pediatr Infect Dis J 2002;21:44--8.
- Piedra PA, Yan L, Kotloff K, et al. Safety of the trivalent, cold-adapted influenza vaccine in preschool-aged children. Pediatrics 2002;110:662--72.
- Bergen R, Black S, Shinefield H, et al. Safety of cold-adapted live attenuated influenza vaccine in a large cohort of children and adolescents. Pediatr Infect Dis J 2004;23:138--44.
- Belshe RB, Ambrose CSYi T. Safety and efficacy of live attenuated influenza vaccine in children 2--7 years of age. Vaccine 2008;26(Suppl 4):D10--6.
- Belshe RB, Edwards KM, Vesikari T, et al. Live attenuated versus inactivated influenza vaccine in infants and young children. N Engl J Med 2007;356:685--96.
- Piedra PA, Gaglani MJ, Riggs M, et al. Live attenuated influenza vaccine, trivalent, is safe in healthy children 18 months to 4 years, 5 to 9 years, and 10 to 18 years of age in a community-based, nonrandomized, open-label trial. Pediatrics 2005;116:e397--407.
- Gaglani MJ, Piedra PA, Riggs M, et al. Safety of the intranasal, trivalent, live attenuated influenza vaccine (LAIV) in children with intermittent wheezing in an open-label field trial. Pediatr Infect Dis J 2008;27:444--52.
- MedImmune, FluMist [Prescribing information]. Gaithersburg, MD: Medimmune Vaccines, Inc.; 2007.
- Izurieta HS, Haber P, Wise RP, et al. Adverse events reported following live, cold-adapted, intranasal influenza vaccine. JAMA 2005;294:2720--5.
- Jackson LA, Holmes SJ, Mendelman PM, et al. Safety of a trivalent live attenuated intranasal influenza vaccine, FluMist, administered in addition to parenteral trivalent inactivated influenza vaccine to seniors with chronic medical conditions. Vaccine 1999;17:1905--9.
- Treanor JJ, Kotloff K, Betts RF, et al. Evaluation of trivalent, live, cold-adapted (CAIV-T) and inactivated (TIV) influenza vaccines in prevention of virus infection and illness following challenge of adults with wild-type influenza A (H1N1), A (H3N2), and B viruses. Vaccine 1999;18:899--906.
- Piedra PA, Gaglani MJ, Kozinetz CA, et al. Trivalent live attenuated intranasal influenza vaccine administered during the 2003--2004 influenza type A (H3N2) outbreak provided immediate, direct, and indirect protection in children. Pediatrics 2007;120:e553--64.
- Wang Z, Tobler S, Roayaei J, et al. Live attenuated or inactivated influenza vaccines and medical encounters for respiratory illnesses among US military personnel. JAMA 2009;301:945--53
- Fleming DM, Crovari P, Wahn U, et al. Comparison of the efficacy and safety of live attenuated cold-adapted influenza vaccine, trivalent, with trivalent inactivated influenza virus vaccine in children and adolescents with asthma. Pediatr Infect Dis J 2006;25:860--9.
- Ashkenazi S, Vertruyen A, Aristegui J, et al. Superior relative efficacy of live attenuated influenza vaccine compared with inactivated influenza vaccine in young children with recurrent respiratory tract infections. Pediatr Infect Dis J 2006;25:870--9.
- Wilde JA, McMillan JA, Serwint J, et al. Effectiveness of influenza vaccine in health care professionals: a randomized trial. JAMA 1999;281:908--13.
- Elder AG, O'Donnell B, McCruden EA, et al. Incidence and recall of influenza in a cohort of Glasgow healthcare workers during the 1993--4 epidemic: results of serum testing and questionnaire. BMJ 1996;313:1241--2.
- Lester RT, McGeer A, Tomlinson G, et al. Use of, effectiveness of, and attitudes regarding influenza vaccine among house staff. Infect Control Hosp Epidemiol 2003;24:839--44.
- Cunney RJ, Bialachowski A, Thornley D, et al. An outbreak of influenza A in a neonatal intensive care unit. Infect Control Hosp Epidemiol 2000;21:449--54.
- Salgado CD, Giannetta ET, Hayden FG, et al. Preventing nosocomial influenza by improving the vaccine acceptance rate of clinicians. Infect Control Hosp Epidemiol 2004;25:923--8.
- Sato M, Saito R, Tanabe N, et al. Antibody response to influenza vaccination in nursing home residents and healthcare workers during four successive seasons in Niigata, Japan. Infect Control Hosp Epidemiol 2005;26:859--66.
- Carman WF, Elder AG, Wallace LA, et al. Effects of influenza vaccination of health-care workers on mortality of elderly people in long-term care: a randomised controlled trial. Lancet 2000;355:93--7.
- Potter J, Stott DJ, Roberts MA, et al. Influenza vaccination of health care workers in long-term-care hospitals reduces the mortality of elderly patients. J Infect Dis 1997;175:1--6.
- Hayward AC, Harling R, Wetten S, et al. Effectiveness of an influenza vaccine programme for care home staff to prevent death, morbidity, and health service use among residents: cluster randomised controlled trial. BMJ 2006;333:1241.
- Thomas RE, Jefferson TO, Demicheli V, et al. Influenza vaccination for health-care workers who work with elderly people in institutions: a systematic review. Lancet Infect Dis 2006;6:273--9.
- Hurwitz ES, Haber M, Chang A, et al. Effectiveness of influenza vaccination of day care children in reducing influenza-related morbidity among household contacts. JAMA 2000;284:1677--82.
- Esposito S, Marchisio P, Cavagna R, et al. Effectiveness of influenza vaccination of children with recurrent respiratory tract infections in reducing respiratory-related morbidity within the households. Vaccine 2003;21:3162--8.
- Piedra PA, Gaglani MJ, Kozinetz CA, et al. Herd immunity in adults against influenza-related illnesses with use of the trivalent-live attenuated influenza vaccine (CAIV-T) in children. Vaccine 2005;23:1540--8.
- King JC Jr, Stoddard JJ, Gaglani MJ, et al. Effectiveness of school-based influenza vaccination. N Engl J Med 2006;355:2523--32.
- Davis MM, King JC Jr, Moag L, et al. Countywide school-based influenza immunization: direct and indirect impact on student absenteeism. Pediatrics 2008;122:e260--5.
- Monto AS, Davenport FM, Napier JA, et al. Modification of an outbreak of influenza in Tecumseh, Michigan by vaccination of schoolchildren. J Infect Dis 1970;122:16--25.
- Ghendon YZ, Kaira AN, Elshina GA. The effect of mass influenza immunization in children on the morbidity of the unvaccinated elderly. Epidemiol Infect 2006;134:71--8.
- Kwong JC, Stukel TA, Lim J, et al.. The effect of universal influenza immunization on mortality and health care use. PLoS Medicine 2008;5:e211.
- CDC. Interim within-season estimate of the effectiveness of trivalent inactivated influenza vaccine--Marshfield, Wisconsin, 2007--08 influenza season. MMWR 2008;57:393--8.
- CDC. Serum cross-reactive antibody response to a novel influenza A (H1N1) virus after vaccination with seasonal influenza vaccine. MMWR 2009;58:521--4.
- Molinari NA, Ortega-Sanchez IR, Messonnier ML, et al. The annual impact of seasonal influenza in the US: measuring disease burden and costs. Vaccine 2007;25:5086--96.
- Riddiough MA, Sisk JE, Bell JC. Influenza vaccination. JAMA 1983;249:3189--95.
- Maciosek MV, Solberg LI, Coffield AB, et al. Influenza vaccination health impact and cost effectiveness among adults aged 50 to 64 and 65 and older. Am J Prev Med 2006;31:72--9.
- Nichol KL. Cost-benefit analysis of a strategy to vaccinate healthy working adults against influenza. Arch Intern Med 2001;161:749--59.
- Nichol KL, Mallon KP, Mendelman PM. Cost benefit of influenza vaccination in healthy, working adults: an economic analysis based on the results of a clinical trial of trivalent live attenuated influenza virus vaccine. Vaccine 2003;21:2207--17.
- Meltzer MI, Neuzil KM, Griffin MR, et al. An economic analysis of annual influenza vaccination of children. Vaccine 2005;23:1004--14.
- Keren R, Zaoutis TE, Saddlemire S, et al. Direct medical cost of influenza-related hospitalizations in children. Pediatrics 2006;118:e1321--7.
- Prosser LA, Bridges CB, Uyeki TM, et al. Health benefits, risks, and cost-effectiveness of influenza vaccination of children. Emerg Infect Dis 2006;12:1548--58.
- Cohen GM, Nettleman MD. Economic impact of influenza vaccination in preschool children. Pediatrics 2000;106:973--6.
- White T, Lavoie S, Nettleman MD. Potential cost savings attributable to influenza vaccination of school-aged children. Pediatrics 1999;103:e73.
- Luce BR, Zangwill KM, Palmer CS, et al. Cost-effectiveness analysis of an intranasal influenza vaccine for the prevention of influenza in healthy children. Pediatrics 2001;108:E24.
- Dayan GH, Nguyen VH, Debbag R, et al. Cost-effectiveness of influenza vaccination in high-risk children in Argentina. Vaccine 2001;19:4204--13.
- Prosser LA, O'Brien MA, Molinari NA, et al. Non-traditional settings for influenza vaccination of adults: costs and cost effectiveness. Pharmacoeconomics 2008;26:163--78.
- Coleman MS, Fontanesi J, Meltzer MI, et al. Estimating medical practice expenses from administering adult influenza vaccinations. Vaccine 2005;23:915--23.
- US Department of Health and Human Services. Healthy people 2000: national health promotion and disease prevention objectives---full report, with commentary. Washington, DC: US Department of Health and Human Services, Public Health Service: Washington, DC; 1991
- US Department of Health and Human Services. Healthy People 2010, With understanding and improving health and objectives for improving health (2 vols.) Washington, DC: US Department of Health and Human Services; 2000.
- CDC. Improving influenza, pneumococcal polysaccharide, and hepatitis B vaccination coverage among adults aged <65 years at high risk: a report on recommendations of the Task Force on Community Preventive Services. MMWR 2005;54(No. RR-5).
- Ndiaye SM, Hopkins DP, Shefer AM, et al. Interventions to improve influenza, pneumococcal polysaccharide, and hepatitis B vaccination coverage among high-risk adults: a systematic review. Am J Prev Med 2005;28:248--79.
- Lu P, Bridges CB, Euler GL, et al. Influenza vaccination of recommended adult populations, U.S., 1989--2005. Vaccine 2008;26:1786--93.
- CDC. Early release of selected estimates based on data from the January--June 2008 National Health Interview Survey. Hyattsville, MD: US Department of Health and Human Services, CDC, National Center for Health Statistics; 2008.
- CDC. Early release of selected estimates based on data from the January--September 2008 National Health Interview Survey. Hyattsville, MD: US Department of Health and Human Services, CDC, National Center for Health Statistics; 2009.
- Zerr DM, Englund JA, Robertson AS, et al. Hospital-based influenza vaccination of children: an opportunity to prevent subsequent hospitalization. Pediatrics 2008;121:345--8.
- Bratzler DW, Houck PM, Jiang H, et al. Failure to vaccinate Medicare inpatients: a missed opportunity. Arch Intern Med 2002;162:2349--56.
- Verani JR, Irigoyen M, Chen S, et al. Influenza vaccine coverage and missed opportunities among inner-city children aged 6 to 23 months: 2000--2005. Pediatrics 2007;119:e580--6.
- Fedson DS, Houck P, Bratzler D. Hospital-based influenza and pneumococcal vaccination: Sutton's Law applied to prevention. Infect Control Hosp Epidemiol 2000;21:692--9.
- Brewer NT, Hallman WK. Subjective and objective risk as predictors of influenza vaccination during the vaccine shortage of 2004--2005. Clin Infect Dis 2006;43:1379--86.
- CDC. Early release of selected estimates based on data from the January--September 2007 National Health Interview Survey. Hyattsville, MD: US Department of Health and Human Services, CDC, National Center for Health Statistics; 2008.
- Hebert PL, Frick KD, Kane RL, et al. The causes of racial and ethnic differences in influenza vaccination rates among elderly Medicare beneficiaries. Health Serv Res 2005;40:517--37.
- Winston CA, Wortley PM, Lees KA. Factors associated with vaccination of medicare beneficiaries in five U.S. communities: results from the racial and ethnic adult disparities in immunization initiative survey, 2003. J Am Geriatr Soc 2006;54:303--10.
- Fiscella K, Dressler R, Meldrum S, et al. Impact of influenza vaccination disparities on elderly mortality in the United States. Prev Med 2007;45:83--7.
- CDC. Influenza vaccination coverage among children with asthma--United States, 2004--05 influenza season. MMWR 2007;56:193--6.
- Marshall BC, Henshaw C, Evans DA, et al. Influenza vaccination coverage level at a cystic fibrosis center. Pediatrics 2002;109:E80--0.
- CDC. Childhood influenza vaccination coverage--United States, 2004--05 influenza season. MMWR 2006;55:1062--5.
- Jackson LA, Neuzil KM, Baggs J, et al. Compliance with the recommendations for 2 doses of trivalent inactivated influenza vaccine in children less than 9 years of age receiving influenza vaccine for the first time: a Vaccine Safety Datalink study. Pediatrics 2006;118:2032--7.
- CDC. Rapid assessment of influenza vaccination coverage among HMO members---northern California influenza seasons, 2001--02 through 2004--05. MMWR 2005;54:676--8.
- CDC. Influenza vaccination coverage among children aged 6--23 months---United States, 2006--07 influenza season. MMWR 2008;57:1039--43.
- Nowalk MP, Zimmerman RK, Lin CJ, et al. Parental perspectives on influenza immunization of children aged 6 to 23 months. Am J Prev Med 2005;29:210--4.
- Gnanasekaran SK, Finkelstein JA, Hohman K, et al. Parental perspectives on influenza vaccination among children with asthma. Public Health Rep 2006;121:181--8.
- Gaglani M, Riggs M, Kamenicky C, et al. A computerized reminder strategy is effective for annual influenza immunization of children with asthma or reactive airway disease. Pediatr Infect Dis J 2001;20:1155--60.
- National Foundation for Infectious Diseases. Call to action: influenza immunization among health-care workers, 2003. Bethesda, MD: National Foundation for Infectious Diseases; 2003. Available at http://www.nfid.org/pdf/publications/fluhealthcarecta08.pdf.
- Poland GA, Tosh P, Jacobson RM. Requiring influenza vaccination for health care workers: seven truths we must accept. Vaccine 2005;23:2251--5.
- CDC. Influenza vaccination of health-care personnel: recommendations of the Healthcare Infection Control Practices Advisory Committee (HICPAC) and the Advisory Committee on Immunization Practices (ACIP). MMWR 2006;55(No. RR-2).
- Walker Frances AJ, Singleton JA, Lu P, et al. Influenza vaccination of healthcare workers in the United States, 1989--2002. Infect Control and Hosp Epidemiol 2006;27:257--265.
- Ofstead CL, Tucker SJ, Beebe TJ, et al. Influenza vaccination among registered nurses: information receipt, knowledge, and decision-making at an institution with a multifaceted educational program. Infect Control Hosp Epidemiol 2008;29:99--106.
- Yeager DP, Toy EC, Baker B 3rd. Influenza vaccination in pregnancy. Am J Perinatol 1999;16:283--6.
- Gonik BM, Jones TM, Contreras DM, et al. The obstetrician-gynecologist's role in vaccine-preventable diseases and immunization. Obstetrics & Gynecology 2000;96:81--84.
- CDC. National Influenza Vaccination Week---November 26--December 2, 2007. MMWR 2007;56:1216--7.
- Zimmerman RK, Raymund M, Janosky JE, et al. Sensitivity and specificity of patient self-report of influenza and pneumococcal polysaccharide vaccinations among elderly outpatients in diverse patient care strata. Vaccine 2003;21:1486--91.
- MacDonald R, Baken L, Nelson A, et al. Validation of self-report of influenza and pneumococcal vaccination status in elderly outpatients. Am J Prev Med 1999;17:173--7.
- Food and Drug Administration. Influenza virus vaccine 2009--2010 season. Washington, DC: Food and Drug Administration; 2009. Available at http://www.fda.gov/cber/flu/flu2009.htm.
- Dagan R, Hall CB. Influenza A virus infection imitating bacterial sepsis in early infancy. Pediatr Infect Dis 1984;3:218--21.
- Anonymous. Prevention of influenza: recommendations for influenza immunization of children, 2007--2008. Pediatrics 2008;121:e1016--31.
- Talbot TR, Bradley SE, Cosgrove SE, et al. Influenza vaccination of healthcare workers and vaccine allocation for healthcare workers during vaccine shortages. Infect Control Hosp Epidemiol 2005;26:882--90.
- Polgreen PM, Chen Y, Beekmann S, et al. Elements of influenza vaccination programs that predict higher vaccination rates: results of an emerging infections network survey. Clin Infect Dis 2008;46:14--9.
- Polgreen PM, Septimus EJ, Parry MF, et al. Relationship of influenza vaccination declination statements and influenza vaccination rates for healthcare workers in 22 US hospitals. Infect Control Hosp Epidemiol 2008;29:675--7.
- CDC. Interventions to increase influenza vaccination of health-care workers---California and Minnesota. MMWR 2005;54:196--9.
- Joint Commission on Accreditation of Healthcare Organizations. New infection control requirement for offering influenza vaccination to staff and licensed independent practitioners. Jt Comm Perspect 2006;26:10--1.
- Infectious Diseases Society of America. Pandemic and seasonal influenza: principles for U.S. action. Arlington, VA: Infectious Diseases Society of America; 2007.
- Stewart AC, Cox M, Rosenbaum S, The epidemiology of U.S. immunization law: immunization requirements for staff and residents of long-term care facilities under state laws/regulations. Washington, DC: George Washington University; 2005.
- Lindley MC, Horlick GA, Shefer AM, et al. Assessing state immunization requirements for healthcare workers and patients. Am J Prev Med 2007;32:459--65.
- CDC. State immunization laws for healthcare workers and patients. Atlanta, GA: US Department of Health and Human Services, CDC; 2009. Available at http://www2a.cdc.gov/nip/stateVaccApp/StateVaccsApp/default.asp.
- CDC. Guidelines for environmental infection control in health-care facilities. Recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee (HICPAC). MMWR 2003;52(No. RR-10).
- CDC. Recommended adult immunization schedule-United States, 2009. MMWR 2009;57:Q1--4.
- Miller JM, Tam TW, Maloney S, et al. Cruise ships: high-risk passengers and the global spread of new influenza viruses. Clin Infect Dis 2000;31:433--8.
- Uyeki TM, Zane SB, Bodnar UR, et al. Large summertime influenza A outbreak among tourists in Alaska and the Yukon Territory. Clin Infect Dis 2003;36:1095--102.
- Mutsch M, Tavernini M, Marx A, et al. Influenza virus infection in travelers to tropical and subtropical countries. Clin Infect Dis 2005;40:1282--7.
- Nichol KL, D'Heilly S, Ehlinger E. Colds and influenza-like illnesses in university students: impact on health, academic and work performance, and health care use. Clin Infect Dis 2005;40:1263--70.
- Awofeso N, Fennell M, Waliuzzaman Z, et al. Influenza outbreak in a correctional facility. Aust N Z J Public Health 2001;25:443--6.
- CDC. Expansion of use of live attenuated influenza vaccine (FluMist(r)) to children aged 2--4 years and other FluMist changes for the 2007--08 influenza season. MMWR 2007;56:1217--9.
- Nolan T, Bernstein DI, Block SL, et al. Safety and immunogenicity of concurrent administration of live attenuated influenza vaccine with measles-mumps-rubella and varicella vaccines to infants 12 to 15 months of age. Pediatrics 2008;121:508--16.
- Kerzner B, Murray AV, Cheng E, et al. Safety and immunogenicity profile of the concomitant administration of ZOSTAVAX and inactivated influenza vaccine in adults aged 50 and older. J Am Geriatr Soc 2007;55:1499--507.
- Gross PA, Russo C, Dran S, et al. Time to earliest peak serum antibody response to influenza vaccine in the elderly. Clin Diagn Lab Immunol 1997;4:491--2.
- Brokstad KA, Cox RJ, Olofsson J, et al. Parenteral influenza vaccination induces a rapid systemic and local immune response. J Infect Dis 1995;171:198--203.
- Lawson F, Baker V, Au D, et al. Standing orders for influenza vaccination increased vaccination rates in inpatient settings compared with community rates. J Gerontol A Biol Sci Med Sci 2000;55:M522--6.
- Centers for Medicare and Medicaid Services. Medicare and Medicaid programs; conditions of participation: immunization standards for hospitals, long-term care facilities, and home health agencies. Final rule with comment period. Fed Regist 2002;67:61808--14.
- Centers for Medicare and Medicaid Services. Emergency update to the 2007 Medicare Physician Fee Schedule Database (MPFSDB). Washington, DC: US Department of Health and Human Services, Centers for Medicare and Medicaid Services; 2009. Available at http://www.cms.hhs.gov/MLNMattersArticles/downloads/MM5459.pdf.
- Centers for Medicare and Medicaid Services. 2006--2007 influenza (flu) season resources for health care professionals. Washington, DC: US Department of Health and Human Services, Centers for Medicare and Medicaid Services; 2009. Available at http://www.cms.hhs.gov/MLNMattersArticles/downloads/SE0667.pdf.
- CDC. Use of standing orders programs to increase adult vaccination rates. MMWR 2000;49(No. RR-1):15--26.
- Rue-Cover A, Iskander J, Lyn S, et al. Death and serious illness following influenza vaccination: a multidisciplinary investigation. Pharmacoepidemiol Drug Saf 2009;18:504--11.
- Stefanacci RG. Creating artificial barriers to vaccinations. J Am Med Dir Assoc 2005;6:357--8.
- CMS. Medicare and Medicaid programs; condition of participation: immunization standard for long term care facilities. Final rule. Fed Regist 2005;70:58833--52.
- Simonsen L, Reichert TA, Viboud C, et al. Impact of influenza vaccination on seasonal mortality in the US elderly population. Arch Intern Med 2005;165:265--72.
- Nichol KL, Nordin J, Mullooly J. Influence of clinical outcome and outcome period definitions on estimates of absolute clinical and economic benefits of influenza vaccination in community dwelling elderly persons. Vaccine 2006;24:1562--8.
- Nachamkin I, Shadomy SV, Moran AP, et al. Anti-ganglioside antibody induction by swine (A/NJ/1976/H1N1) and other influenza vaccines: insights into vaccine-associated Guillain-Barre syndrome. J Infect Dis 2008;198:226--33.
- Weycker D, Edelsberg J, Halloran ME, et al. Population-wide benefits of routine vaccination of children against influenza. Vaccine 2005;23:1284--93.
- Longini IM Jr, Halloran ME. Strategy for distribution of influenza vaccine to high-risk groups and children. Am J Epidemiol 2005;161:303--6.
- Jordan R, Connock M, Albon E, et al. Universal vaccination of children against influenza: are there indirect benefits to the community? A systematic review of the evidence. Vaccine 2006;24:1047--62.
- Schwartz B, Hinman A, Abramson J, et al. Universal influenza vaccination in the United States: are we ready? Report of a meeting. J Infect Dis 2006;194 Suppl 2:S147--54.
- Abramson JS, Neuzil KM, Tamblyn SE. Annual universal influenza vaccination: ready or not? Clin Infect Dis 2006;42:132--5.
- Glezen WP. Herd protection against influenza. J Clin Virol 2006;37:237--43.
- Helms CM, Guerra FA, Klein JO, et al. Strengthening the nation's influenza vaccination system: a National Vaccine Advisory Committee assessment. Am J Prev Med 2005;29:221--6.
- Council of State and Territorial Epidemiologists. Interim position statement 07-ID-01: national reporting for initial detections of novel influenza A viruses. Atlanta, GA: Council of State and Territorial Epidemiologists; 2007. Available at http://www.cste.org/PS/2007ps/ID/07-ID-01.pdf.
- World Health Organization. Update: WHO-confirmed human cases of avian influenza A (H5N1) infection, November 2003--May 2008. Wkly Epidemiol Rec 2008;83:415--20.
- Kandun IN, Wibisono H, Sedyaningsih ER, et al. Three Indonesian clusters of H5N1 virus infection in 2005. N Engl J Med 2006;355:2186--94.
- Oner AF, Bay A, Arslan S, et al. Avian influenza A (H5N1) infection in eastern Turkey in 2006. N Engl J Med 2006;355:2179--85.
- Areechokchai D, Jiraphongsa C, Laosiritaworn Y, et al. Investigation of avian influenza (H5N1) outbreak in humans---Thailand, 2004. MMWR 2006;55 Suppl 1:3--6.
- Dinh PN, Long HT, Tien NT, et al. Risk factors for human infection with avian influenza A H5N1, Vietnam, 2004. Emerg Infect Dis 2006;12:1841--7.
- Gilsdorf A, Boxall N, Gasimov V, et al. Two clusters of human infection with influenza A/H5N1 virus in the Republic of Azerbaijan, February-March 2006. Euro Surveill 2006;11:122--6.
- WHO. Update: WHO-confirmed human cases of avian influenza A(H5N1) infection, 25 November 2003--24 November 2006. Wkly Epidemiol Rec 2007;82:41--8.
- Kandun IN, Tresnaningsih E, Purba WH, et al. Factors associated with case fatality of human H5N1 virus infections in Indonesia: a case series. Lancet 2008;372:744--9.
- Yu H, Gao Z, Feng Z, et al. Clinical characteristics of 26 human cases of highly pathogenic avian influenza A (H5N1) virus infection in China. PLoS ONE 2008;3:e2985.
- Wang H, Feng Z, Shu Y, et al. Probable limited person-to-person transmission of highly pathogenic avian influenza A (H5N1) virus in China. Lancet 2008;371:1427--34.
- Abdel-Ghafar AN, Chotpitayasunondh T, Gao Z, et al. Update on avian influenza A (H5N1) virus infection in humans. N Engl J Med 2008;358:261--73.
- Monto AS. The threat of an avian influenza pandemic. N Engl J Med 2005;352:323--5.
- Maines TR, Chen LM, Matsuoka Y, et al. Lack of transmission of H5N1 avian-human reassortant influenza viruses in a ferret model. Proc Natl Acad Sci U S A 2006;103:12121--6.
- CDC. Updated interim guidance for laboratory testing of persons with suspected infection with highly pathogenic avian influenza A (H5N1) virus in the United States. Atlanta, GA: US Department of Health and Human Services, CDC; 2009. Available at http://www.cdc.gov/flu/avian/professional/guidance-labtesting.htm.
- CDC. Guidance for follow-up of contacts of persons with suspected infection with highly pathogenic avian influenza A (H5N1) virus in the United States. Atlanta, GA: US Department of Health and Human Services, CDC; 2009. Available at http://www.cdc.gov/flu/avian/professional/guidance-followup.htm.
- Nguyen-Van-Tam JS, Nair P, Acheson P, et al. Outbreak of low pathogenicity H7N3 avian influenza in UK, including associated case of human conjunctivitis. Euro Surveill 2006;11:E060504 2.
- Kurtz J, Manvell RJ, Banks J. Avian influenza virus isolated from a woman with conjunctivitis. Lancet 1996;348:901--2.
- Peiris M, Yuen KY, Leung CW, et al. Human infection with influenza H9N2. Lancet 1999;354:916--7.
- CDC. Update: influenza activity---United States and worldwide, 2003--04 season, and composition of the 2004--05 influenza vaccine. MMWR 2004;53:547--52.
- Uyeki TM, Chong YH, Katz JM, et al. Lack of evidence for human-to-human transmission of avian influenza A (H9N2) viruses in Hong Kong, China 1999. Emerg Infect Dis 2002;8:154--9.
- Yuanji G. Influenza activity in China: 1998--1999. Vaccine 2002;20 Suppl 2:S28--35.
- Editorial Team, Eurosurveillance Editorial Office. Avian influenza A/(H7N2) outbreak in the United Kingdom. Euro Surveill 2007;12:E070531 2.
- Fouchier RA, Schneeberger PM, Rozendaal FW, et al. Avian influenza A virus (H7N7) associated with human conjunctivitis and a fatal case of acute respiratory distress syndrome. Proc Natl Acad Sci U S A 2004;101:1356--61.
- Koopmans M, Wilbrink B, Conyn M, et al. Transmission of H7N7 avian influenza A virus to human beings during a large outbreak in commercial poultry farms in the Netherlands. Lancet 2004;363:587--93.
- Tweed SA, Skowronski DM, David ST, et al. Human illness from avian influenza H7N3, British Columbia. Emerg Infect Dis 2004;10:2196--9.
- Olsen CW. The emergence of novel swine influenza viruses in North America. Virus Res 2002;85:199--210.
- Ma W, Vincent AL, Gramer MR, et al. Identification of H2N3 influenza A viruses from swine in the United States. Proc Natl Acad Sci U S A 2007;104:20949--54.
- Myers KP, Olsen CW, Setterquist SF, et al. Are swine workers in the United States at increased risk of infection with zoonotic influenza virus? Clin Infect Dis 2006;42:14--20.
- Dowdle WR, Hattwick MA. Swine influenza virus infections in humans. J Infect Dis 1977;136 Suppl:S386--9.
- Myers KP, Olsen CW, Gray GC. Cases of swine influenza in humans: a review of the literature. Clin Infect Dis 2007;44:1084--8.
- Newman AP, Reisdorf E, Beinemann J, et al. Human case of swine influenza A (H1N1) triple reassortant virus infection, Wisconsin. Emerg Infect Dis 2008;14:1470--2.
- Dacso CC, Couch RB, Six HR, et al. Sporadic occurrence of zoonotic swine influenza virus infections. J Clin Microbiol 1984;20:833--5.
- Gray GC, McCarthy T, Capuano AW, et al. Swine workers and swine influenza virus infections. Emerg Infect Dis 2007;13:1871--8.
- CDC. Update: influenza activity---United States and worldwide, May 20--September 15, 2007. MMWR 2007;56:1001--4.
- Shinde V, Bridges CB, Uyeki TM, et al. Triple-reassortant swine influenza A (H1) in humans in the United States, 2005--2009. N Engl J Med 2009.
- Olsen CW, Brammer L, Easterday BC, et al. Serologic evidence of H1 swine Influenza virus infection in swine farm residents and employees. Emerg Infect Dis 2002;8:814--9.
- CDC. Outbreak of swine-origin influenza A (H1N1) virus infection---Mexico, March--April 2009. MMWR 2009;58:467--70.
- CDC. Interim guidance for protection of persons involved in U.S. avian influenza outbreak disease control and eradication activities. Atlanta, GA: US Department of Health and Human Services, CDC; 2006. Available at http://www.cdc.gov/flu/avian/professional/protect-guid.htm.
- Occupational Safety and Health Administration. OSHA guidance update on protecting employees from avian flu (avian influenza) viruses. Washington, DC: US Department of Labor, Occupational Safety and Health Administration; 2006. Available at http://www.osha.gov/OshDoc/data_AvianFlu/avian_flu_guidance_english.pdf.
- Dharan NJ, Gubareva LV, Meyer JJ, et al. Infections with oseltamivir-resistant influenza A(H1N1) virus in the United States. JAMA 2009;301:1034--41.
- Lackenby A, Thompson CIDemocratis J. The potential impact of neuraminidase inhibitor resistant influenza. Curr Opin Infect Dis 2008;21:626--38.
- Meijer A, Lackenby A, Hungnes O, et al. Oseltamivir-resistant influenza virus A (H1N1), Europe, 2007--08 Season. Emerg Infect Dis 2009;15:552--60.
- World Health Organization. Viruses resistant to oseltamivir (Tamiflu) identified. Geneva, Switzerland: World Health Organization; 2009. Available at: http://www.who.int/csr/disease/swineflu/notes/h1n1_antiviral_resistance_20090708/en/index.html.
- CDC. Interim guidance on antiviral recommendations for patients with novel influenza A (H1N1) virus infection and their close contacts. Atlanta, GA: US Department of Health and Human Services, CDC; 2009. Available at http://www.cdc.gov/h1n1flu/recommendations.htm.
- Harper SA, Bradley JS, Englund JA, et al. Seasonal influenza in adults and children-diagnosis, treatment, chemoprophylaxis, and institutional outbreak management: clinical practice guidelines of the Infectious Diseases Society of America. Clin Infect Dis 2009;48:1003--32.