 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Investigation of Avian Influenza (H5N1) Outbreak in Humans --- Thailand, 2004Darin Areechokchai,1 C.
Jiraphongsa,1 Y.
Laosiritaworn,1 W.
Hanshaoworakul,1 M. O'Reilly2
Corresponding author: Darin Areechokchai, Bureau of Epidemiology, Ministry of Public Health, Nonthaburi 11000, Thailand. Telephone: 662-5901734; Fax: 662-5918581; E-mail: doggyrin@health3.moph.go.th. Disclosure of relationship: The contributors of this report have disclosed that they have no financial interest, relationship, affiliation, or other association with any organization that might represent a conflict of interest. In addition, this report does not contain any discussion of unlabeled use of commercial products or products for investigational use. AbstractIntroduction: Beginning in late 2003, a substantial outbreak of influenza A (H5N1) virus spread among poultry in Thailand. On January 23, 2004, the Ministry of Public Health (MPH) detected the first confirmed human case of H5N1 infection in humans. Methods: During February--November 2004, the MPH's Bureau of Epidemiology and provincial health offices worked together to investigate the H5N1 outbreak in humans. Two studies were conducted: a descriptive study to describe clinical manifestations and epidemiologic characteristic of the cases and a matched case-control study to determine risk factors for persons who might subsequently become ill with H5N1. Results: A total of 16 patients with confirmed H5N1 were identified for the case-control study. Fever and respiratory symptoms predominated. Leucopenia and thrombocytopenia were present respectively in nine (100%) and four (44%) persons aged <15 years. Direct touching of unexpectedly dead poultry was the most significant risk factor (odds ratio = 29.0; 95% confidence interval = 2.7--308.2). Overall mortality was 75%; mortality for persons aged <15 years was 90%, compared with 57% for persons aged >15 years. Conclusion: Avian influenza was more severe in children, who should avoid handling dead poultry during epizootics. Early avian influenza in children resembled the more common dengue fever, but presence of cough and absence of hemoconcentration distinguished avian influenza, which often progressed rapidly to acute respiratory distress syndrome, requiring intensive care. IntroductionIn 1997, the first outbreak of avian influenza A (H5N1) in humans was reported in Hong Kong, and exposure to infected poultry was identified as the probable route of transmission (1--3). Since late 2003, this highly pathogenic virus has caused disease outbreaks in poultry in at least eight East Asian countries, and fatalities among humans have been attributed to the same virus in Cambodia, China, Indonesia, Thailand, and Vietnam (4,5). During December 2003--October 2004, a total of 17 cases of respiratory disease caused by H5N1 viruses were reported in Thailand; these cases coincided in time and place with the epizootic disease in poultry. Millions of persons in Thailand raise backyard chickens near their houses, which has increased concern about the potential for a substantial H5N1 outbreak in humans. MethodsDuring February--November 2004, the Bureau of Epidemiology (BoE) of the Ministry of Public Health (MPH) and provincial health offices worked together to investigate the H5N1 outbreak in humans. Two studies were conducted: a descriptive study to describe clinical manifestations and epidemiologic characteristic of the cases and a matched case-control study to determine risk factors for persons who might subsequently become ill with H5N1. Descriptive StudyCase Definition Under Thailand's nationwide avian influenza surveillance system, which BoE initiated in December 2003, any patient admitted to a health-care facility with pneumonia or influenza-like illness who has a history of exposure to poultry is reported to BoE. For this study, a case was defined as H5N1 illness occurring in a person who had received a diagnosis of pneumonia or influenza-like illness and who had either a positive viral culture for H5N1 virus or confirmation of H5 strain by real-time reverse transcription-polymerase chain reaction (RT-PCR) (6,7). Laboratory Testing Each patient was evaluated for H5N1 infection status at the time of hospital admission. Nasopharyngeal aspirates or nasopharyngeal swabs were collected and submitted to the Thai National Institute of Health or to Siriraj Hospital at Mahidol University for laboratory testing. Data Collection and Analysis A standard form was used to collect information concerning clinical manifestations as recorded on medical records. Clinical manifestations and clinical progressions were analyzed using Epi Info 2002 version 2 (CDC, Atlanta, Georgia) and Excel (Microsoft, Redmond, Washington). Analytic StudyStudy Design and Sampling A matched case-control study was conducted during the outbreak using the same case definition as the descriptive study. Population registers in the primary health-care unit of each village were used as the control sampling frame. Controls were selected matching village and age ±1 year to each patient and then randomly selecting four controls for each patient. For one patient who was thought to have been infected through human-to-human transmission, four controls were selected from among health-care workers in the hospital to which the patient was admitted. A control was defined as any person who had no fever (i.e., temperature >100.4°F [>38.0°C]) <7 days before or after the onset of illness of the matched case-patient. With respect to exposure, unexpectedly dead poultry was defined as death of >10% of all poultry in a farm or house within 1 day or >40% within 3 days. Data Collection and Analysis Interviews were conducted by physicians in BoE's Field Epidemiology Training Program. For persons who had died or were aged <12 years, adult proxies were interviewed. A standard questionnaire was used to collect information regarding demographic characteristics, underlying diseases, exposure to other persons with suspected avian influenza, and activities related to poultry in the week before onset of illness in the matched case-patient. In the univariate matched analyses, the matched odds ratio (OR) and 95% confidence interval (CI) were calculated for each exposure using Epi Info 2002 version 2. The Mantel-Haenszel summary chi square test was used for the matched data. ResultsThe outbreak in humans occurred in two episodes: January--March and August--October 2004 (Figure 1). Of 17 cases reported from the avian influenza surveillance system, one was excluded because of incomplete laboratory results; 16 patients with confirmed H5N1 infection were included in the study. Patients lived in 11 provinces, primarily in central Thailand. Nine were male and seven female. Patients varied by age (range: 2--58 years; median: 13 years). Of the 16 patients included in the study, nine (56%) were aged <15 years. Overall mortality was 75%; mortality for persons aged <15 years was 90%, compared with 57% for persons aged >15 years (OR = 6.4; CI = 0.4--204.2). The incubation period varied (range: 2--8 days; median: 3 days). All patients had fever. Respiratory symptoms predominated, including cough (12 patients [75%]), sputum (12 [75%]), dyspnea (11 [69%]), and rhinorrhea (seven [44%]); four (25%) patients had gastrointestinal symptoms (Figure 2). All nine patients aged <15 years had leucopenia (white blood cell count: <5,000 cells/mm3; normal range: 5,000--10,000 cells/mm3), and four (44%) also had thrombocytopenia (platelet count: <100,000 cells/mm3; normal range: 100,000--400,000 cells/mm3). The median hematocrit in persons aged <15 years was 39% (range: 30%--41%). Clinical progressions demonstrated rapid progressive pneumonia and acute respiratory distress syndrome (ARDS) in all persons who died (Table 1). In the matched case-control study, case-patients and controls were similar with respect to the majority of variables (Table 2). From the univariate matched analyses, direct touching of unexpectedly dead poultry yielded the highest odds ratio (OR = 29.0; 95% CI = 2.7--308.2). Having poultry die in or near the house or performing activities related to poultry was significantly associated with H5N1 disease (Table 3). No statistically significant difference was identified among patients reporting contact with persons with a suspected case of human avian influenza (Table 3). DiscussionThe H5N1 outbreak in humans in Thailand during January--March and August--October 2004 affected primarily children. Overall mortality was high (75%). During the same period, mortality among persons with human cases in Vietnam was also high (80%) (8). These data suggest that the disease has become more severe than that reported in Hong Kong in 1997, in which mortality was only 33% (2). However, availability of early aggressive treatment in Hong Kong in 1997 or mutation of the virus might explain this difference. During December 16, 2004--December 8, 2005, global mortality among persons with human cases was 29% (5). Basic laboratory profiles in children indicated leucopenia and thrombocytopenia. These laboratory results in children led attending physicians initially to suspect dengue fever in the differential diagnosis. However, certain differences helped clinicians differentiate between H5N1 infection and dengue fever. In H5N1 cases, respiratory symptoms typically predominate, and case-patients did not exhibit hemoconcentration (7). The rapid progressive pneumonia and ARDS required physicians in the epizootic areas to be aware of the possibility of H5N1 infection before patients' illness progressed to severe pneumonia. In the matched analyses, H5N1 disease was associated with recent exposure to sick or unexpectedly dead poultry, particularly direct touching of unexpectedly dead poultry. Only one patient was reported to have been exposed in a live poultry market. In contrast, the 1997 Hong Kong outbreak was related to exposure to live poultry market (2,9). A family cluster was recognized in Thailand in September 2004, probably as a result of person-to-person transmission during unprotected exposure to a critically ill index patient (10). Vietnam also reported probable human-to-human transmission in February 2004 (11). Moreover, in Hong Kong in 1997, a study demonstrated H5N1 infection among health-care workers exposed to a patient with H5N1 infection (12). The risk for person-to-person transmission of H5N1 virus might increase, either through viral mutation or reassortment. This emphasizes the need for strong surveillance, early detection, and intensive measures to protect unexpected person-to-person transmission. The findings in this report are subject to at least five limitations. First, because a substantial number of patients had died, case interviews were conducted more frequently by proxy than were control interviews. Second, recall bias might have occurred as a result of the public's high level of alarm. Third, exposure to poultry was part of the surveillance requirement, and results might have been biased toward these exposures. Fourth, because dengue serology was not documented for patients, combined dengue and H5N1 infections cannot be ruled out. Finally, matching one case patient with health-care workers rather than community controls might have introduced bias. During and after performance of this study, multiple recommendations were provided to health-care workers. In areas that have unexpectedly dead poultry, clinicians should have a high index of suspicion for avian influenza in patients with acute respiratory illness and quickly distinguish between H5N1 and other viral infections (e.g., dengue fever) that have similar laboratory profiles in children. Persons (especially children) should avoid direct contact with unexpectedly sick or dead poultry and not store products from unexpectedly dead poultry in the home. When contact is unavoidable, persons should use personal protective equipment and wash their hands frequently. Long-term public health surveillance and control measures are needed to monitor for person-to-person transmission and the emergence of a potentially pandemic H5N1 influenza virus. Acknowledgments Assistance in field investigations, advice, and technical support was provided by the Bureau of Epidemiology, Kumnuan Ungchusak, MD, Director; provincial health offices for Kanchanaburi, Kamphaeng Phet, Khon Kaen, Nakhon Ratchasima, Petchabun, Lop Buri, Sukhothai, Suphan Buri, Chaiyaphum, Pathum Thani and Phrachin Buri provinces; staff members and fellows of the Thai Field Epidemiology Training Program; the Thai National Institute of Health; the Thai Ministry of Public Health--CDC Collaboration; and Timothy M. Uyeki, MD, National Center for Infectious Diseases, CDC. References
Table 1  Return to top. Figure 1 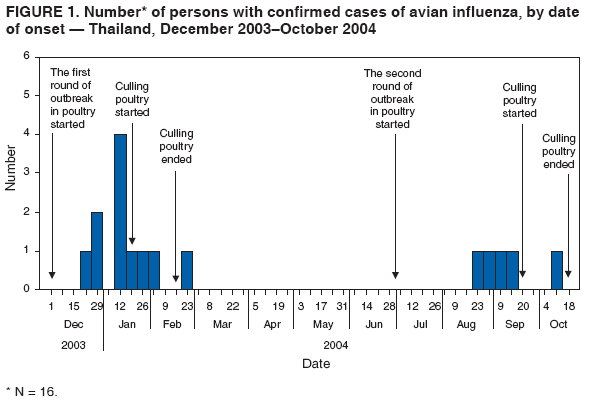 Return to top. Table 2 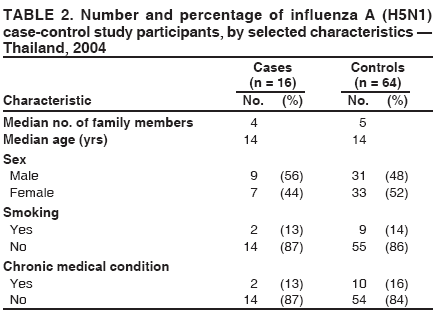 Return to top. Figure 2 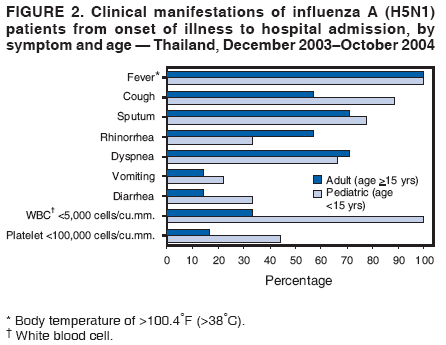 Return to top. Table 3 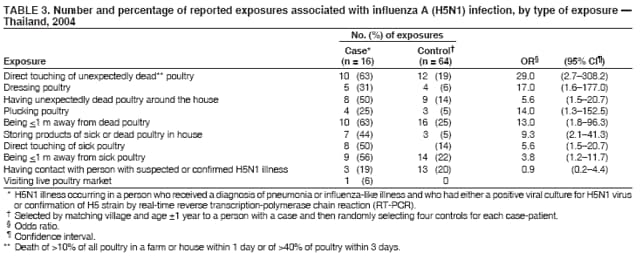 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 4/6/2006 |
|||||||||
|