 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Prevention and Control of InfluenzaRecommendations of the Advisory Committee on Immunization Practices (ACIP) Prepared by The material is this report originated in the National Center for Infectious Diseases, James M. Hughes, M.D., Director, and the Division of Viral and Rickettsial Diseases, James LeDuc, Ph.D., Director; and the National Immunization Program, Stephen Cochi, M.D., Acting Director, and Epidemiology and Surveillance Division, Melinda Wharton, M.D., Director. Summary This report updates the 2003 recommendations by the Advisory Committee on Immunization Practices (ACIP) on the use of influenza vaccine and antiviral agents (CDC. Prevention and control of influenza: recommendations of the Advisory Committee on Immunization Practices [ACIP]. MMWR 2003;52[No. RR-8]:1--34). The 2004 recommendations include new or updated information regarding 1) influenza vaccine for children aged 6--23 months; 2) vaccination of health-care workers with live, attenuated influenza vaccine (LAIV); 3) personnel who may administer LAIV; 4) the 2004--05 trivalent inactivated vaccine virus strains: A/Fujian/411/2002 (H3N2)-like, A/New Caledonia/20/99 (H1N1)-like, and B/Shanghai/361/2002-like antigens (for the A/Fujian/411/2002 (H3N2)-like antigen, manufacturers may use the antigenically equivalent A/Wyoming/3/2003 [H3N2] virus, and for the B/Shanghai/361/2002-like antigen, manufacturers may use the antigenically equivalent B/Jilin/20/2003 virus or B/Jiangsu/10/2003 virus); and 5) the assessment of vaccine supply and timing of influenza vaccination. A link to this report and other information regarding influenza can be accessed at http://www.cdc.gov/flu. IntroductionEpidemics of influenza typically occur during the winter months in temperate regions and have been responsible for an average of approximately 36,000 deaths/year in the United States during 1990--1999 (1). Influenza viruses also can cause pandemics, during which rates of illness and death from influenza-related complications can increase worldwide. Influenza viruses cause disease among all age groups (2--4). Rates of infection are highest among children, but rates of serious illness and death are highest among persons aged >65 years and persons of any age who have medical conditions that place them at increased risk for complications from influenza (2,5--7). Influenza vaccination is the primary method for preventing influenza and its severe complications. In this report from the Advisory Committee on Immunization Practices (ACIP), the primary target groups recommended for annual vaccination are 1) persons at increased risk for influenza-related complications (e.g., those aged >65 years, children aged 6--23 months, pregnant women, and persons of any age with certain chronic medical conditions); 2) persons aged 50--64 years because this group has an elevated prevalence of certain chronic medical conditions; and 3) persons who live with or care for persons at high risk (e.g., health-care workers and household contacts who have frequent contact with persons at high risk and who can transmit influenza to those persons at high risk). Vaccination is associated with reductions in influenza-related respiratory illness and physician visits among all age groups, hospitalization and death among persons at high risk, otitis media among children, and work absenteeism among adults (8--18). Although influenza vaccination levels increased substantially during the 1990s, further improvements in vaccine coverage levels are needed, chiefly among persons aged <65 years who are at increased risk for influenza-related complications among all racial and ethnic groups, among blacks and Hispanics aged >65 years, among children aged 6--23 months, and among health-care workers. ACIP recommends using strategies to improve vaccination levels, including using reminder/recall systems and standing orders programs (19,20). Although influenza vaccination remains the cornerstone for the control and treatment of influenza, information on antiviral medications is also presented because these agents are an adjunct to vaccine. Primary Changes and Updates in the RecommendationsThe 2004 recommendations include four principal changes or updates:
Influenza and Its BurdenBiology of Influenza Influenza A and B are the two types of influenza viruses that cause epidemic human disease (21). Influenza A viruses are further categorized into subtypes on the basis of two surface antigens: hemagglutinin (H) and neuraminidase (N). Influenza B viruses are not categorized into subtypes. Since 1977, influenza A (H1N1) viruses, influenza A (H3N2) viruses, and influenza B viruses have been in global circulation. In 2001, influenza A (H1N2) viruses that probably emerged after genetic reassortment between human A (H3N2) and A (H1N1) viruses began circulating widely. Both influenza A and B viruses are further separated into groups on the basis of antigenic characteristics. New influenza virus variants result from frequent antigenic change (i.e., antigenic drift) resulting from point mutations that occur during viral replication. Influenza B viruses undergo antigenic drift less rapidly than influenza A viruses. A person's immunity to the surface antigens, including hemagglutinin, reduces the likelihood of infection and severity of disease if infection occurs (22). Antibody against one influenza virus type or subtype confers limited or no protection against another. Furthermore, antibody to one antigenic variant of influenza virus might not protect against a new antigenic variant of the same type or subtype (23). Frequent development of antigenic variants through antigenic drift is the virologic basis for seasonal epidemics and the reason for the usual incorporation of one or more new strains in each year's influenza vaccine. Clinical Signs and Symptoms of Influenza Influenza viruses are spread from person to person primarily through the coughing and sneezing of infected persons (21). The incubation period for influenza is 1--4 days, with an average of 2 days (24). Adults typically are infectious from the day before symptoms begin through approximately 5 days after illness onset. Children can be infectious for >10 days, and young children can shed virus for <6 days before their illness onset. Severely immunocompromised persons can shed virus for weeks or months (25--28). Uncomplicated influenza illness is characterized by the abrupt onset of constitutional and respiratory signs and symptoms (e.g., fever, myalgia, headache, malaise, nonproductive cough, sore throat, and rhinitis) (29). Among children, otitis media, nausea, and vomiting are also commonly reported with influenza illness (30--32). Respiratory illness caused by influenza is difficult to distinguish from illness caused by other respiratory pathogens on the basis of symptoms alone (see Role of Laboratory Diagnosis). Reported sensitivities and specificities of clinical definitions for influenza-like illness in studies primarily among adults that include fever and cough have ranged from 63% to 78% and 55% to 71%, respectively, compared with viral culture (33,34). Sensitivity and predictive value of clinical definitions can vary, depending on the degree of co-circulation of other respiratory pathogens and the level of influenza activity (35). A study among older nonhospitalized patients determined that symptoms of fever, cough, and acute onset had a positive predictive value of 30% for influenza (36), whereas a study of hospitalized older patients with chronic cardiopulmonary disease determined that a combination of fever, cough, and illness of <7 days was 78% sensitive and 73% specific for influenza (37). However, a study among vaccinated older persons with chronic lung disease reported that cough was not predictive of influenza infection, although having a fever or feverishness was 68% sensitive and 54% specific for influenza infection (38). Influenza illness typically resolves after a limited number of days for the majority of persons, although cough and malaise can persist for >2 weeks. Among certain persons, influenza can exacerbate underlying medical conditions (e.g., pulmonary or cardiac disease), lead to secondary bacterial pneumonia or primary influenza viral pneumonia, or occur as part of a coinfection with other viral or bacterial pathogens (39). Young children with influenza infection can have initial symptoms mimicking bacterial sepsis with high fevers (40,41), and <20% of children hospitalized with influenza can have febrile seizures (31,42). Influenza infection has also been associated with encephalopathy, transverse myelitis, Reye syndrome, myositis, myocarditis, and pericarditis. (31,39,43,44). Hospitalizations and Deaths from Influenza The risks for complications, hospitalizations, and deaths from influenza are higher among persons aged >65 years, young children, and persons of any age with certain underlying health conditions (see Persons at Increased Risk for Complications) than among healthy older children and younger adults (1,6,8,45--50). Estimated rates of influenza-associated hospitalizations have varied substantially by age group in studies conducted during different influenza epidemics (Table 1). Among children aged 0--4 years, hospitalization rates have ranged from approximately 500/100,000 children for those with high-risk medical conditions to 100/100,000 children for those without high-risk medical conditions (51--54). Within the 0--4 year age group, hospitalization rates are highest among children aged 0--1 years and are comparable to rates reported among persons >65 years (53,54) (Table 1). During influenza epidemics from 1969--70 through 1994--95, the estimated overall number of influenza-associated hospitalizations in the United States ranged from approximately 16,000 to 220,000/epidemic. An average of approximately 114,000 influenza-related excess hospitalizations occurred per year, with 57% of all hospitalizations occurring among persons aged <65 years. Since the 1968 influenza A (H3N2) virus pandemic, the greatest numbers of influenza-associated hospitalizations have occurred during epidemics caused by type A (H3N2) viruses, with an estimated average of 142,000 influenza-associated hospitalizations per year (55). Influenza-related deaths can result from pneumonia as well as from exacerbations of cardiopulmonary conditions and other chronic diseases. Older adults account for >90% of deaths attributed to pneumonia and influenza (1,50). In a recent study of influenza epidemics, approximately 19,000 influenza-associated pulmonary and circulatory deaths per influenza season occurred during 1976--1990, compared with approximately 36,000 deaths during 1990--1999 (1). Estimated rates of influenza-associated pulmonary and circulatory deaths/100,000 persons were 0.4--0.6 among persons aged 0--49 years, 7.5 among persons aged 50--64 years, and 98.3 among persons aged >65 years. In the United States, the number of influenza-associated deaths might be increasing in part because the number of older persons is increasing (56). In addition, influenza seasons in which influenza A (H3N2) viruses predominate are associated with higher mortality (57); influenza A (H3N2) viruses predominated in 90% of influenza seasons during 1990--1999, compared with 57% of seasons during 1976--1990 (1). Deaths from influenza are uncommon among children with and without high-risk conditions, but do occur (58,59). A study that modeled influenza-related deaths estimated that an average of 92 deaths occurred among children aged <5 years annually during the 1990's compared with 35,274 deaths among adults aged >50 years (1). Preliminary reports of laboratory- confirmed pediatric deaths during the 2003--04 influenza season indicated that among these 143 influenza-related deaths (as of April 10, 2004), 58 (41%) were aged <2 years and, of those aged 2--17 years, 65 (45%) did not have an underlying medical condition traditionally considered to place a person at risk for influenza-related complications (unpublished data, CDC National Center for Infectious Diseases, 2004). Further information is needed regarding the risk of severe influenza-complications and optimal strategies for minimizing severe disease and death among children. Options for Controlling InfluenzaIn the United States, the primary option for reducing the effect of influenza is immunoprophylaxis with vaccine. Inactivated (i.e., killed virus) influenza vaccine and live, attenuated influenza vaccine are available for use in the United States (see Recommendations for Using Inactivated and Live, Attenuated Influenza Vaccine). Vaccinating persons at high risk for complications and their contacts each year before seasonal increases in influenza virus circulation is the most effective means of reducing the effect of influenza. Vaccination coverage can be increased by administering vaccine to persons during hospitalizations or routine health-care visits before the influenza season, making special visits to physicians' offices or clinics unnecessary. When vaccine and epidemic strains are well-matched, achieving increased vaccination rates among persons living in closed settings (e.g., nursing homes and other chronic-care facilities) and among staff can reduce the risk for outbreaks by inducing herd immunity (13). Vaccination of health-care workers and other persons in close contact with persons at increased risk for severe influenza illness can also reduce transmission of influenza and subsequent influenza-related complications. Antiviral drugs used for chemoprophylaxis or treatment of influenza are a key adjunct to vaccine (see Recommendations for Using Antiviral Agents for Influenza). However, antiviral medications are not a substitute for vaccination. Influenza Vaccine Composition Both the inactivated and live, attenuated vaccines prepared for the 2004--05 season will include A/Fujian/411/2002 (H3N2)-like, A/New Caledonia/20/99 (H1N1)-like, and B/Shanghai/361/2002-like antigens. For the A/Fujian/411/2002 (H3N2)-like antigen, manufacturers may use the antigenically equivalent A/Wyoming/3/2003 (H3N2) virus, and for the B/Shanghai/361/2002-like antigen, manufacturers may use the antigenically equivalent B/Jilin/20/2003 virus or B/Jiangsu/10/2003 virus. These viruses will be used because of their growth properties and because they are representative of influenza viruses likely to circulate in the United States during the 2004--05 influenza season. Because circulating influenza A (H1N2) viruses are a reassortant of influenza A (H1N1) and (H3N2) viruses, antibody directed against influenza A (H1N1) and influenza (H3N2) vaccine strains will provide protection against circulating influenza A (H1N2) viruses. Influenza viruses for both the inactivated and live attenuated influenza vaccines are initially grown in embryonated hens' eggs. Thus, both vaccines might contain limited amounts of residual egg protein. For the inactivated vaccine, the vaccine viruses are made noninfectious (i.e., inactivated or killed) (60). Subvirion and purified surface antigen preparations of the inactivated vaccine are available. Manufacturing processes differ by manufacturer. Manufacturers might use different compounds to inactivate influenza viruses and add antibiotics to prevent bacterial contamination. Package inserts should be consulted for additional information. Thimerosal Thimerosal, a mercury-containing compound, has been used as a preservative in vaccines since the 1930s and is used in multidose vials of inactivated influenza vaccine to reduce the likelihood of bacterial contamination. Although no scientific evidence indicates that thimerosal in vaccines leads to serious adverse events in vaccine recipients, in 1999, the U.S. Public Health Service and other organizations recommended that efforts be made to eliminate or reduce the thimerosal content in vaccines to decrease total mercury exposure, chiefly among infants (61--63). Since mid-2001, vaccines routinely recommended for infants in the United States have been manufactured either without or with only trace amounts of thimerosal to provide a substantial reduction in the total mercury exposure from vaccines for children (64). Vaccines containing trace amounts of thimerosal have <1 mcg mercury/dose. In 1999, 15 of 28 vaccine products for which CDC had contracts did not contain thimerosal as a preservative. In 2004, 27 of 29 products under CDC contract do not contain thimerosal as a preservative. Influenza Vaccines and Thimerosal. LAIV does not contain thimerosal. Thimerosal preservative-containing inactivated influenza vaccines, distributed in multidose containers in the United States, contain 25 mcg of mercury/0.5-mL dose (61,62). Inactivated influenza virus vaccines distributed in the United States as preservative-free vaccines in single-dose syringes contain only trace amounts of thimerosal as a residual from early manufacturing steps. Inactivated influenza vaccine that does not contain thimerosal as a preservative has <1 mcg mercury/0.5-mL dose or <0.5 mcg mercury/0.25-mL dose. This information is included in the package insert provided with each type of inactivated influenza virus vaccine. Beginning in 2004, influenza vaccine is part of the routine childhood immunization schedule. For the 2004--05 influenza season, 6--8 million single-dose syringes of inactivated influenza virus vaccine without thimerosal as a preservative probably will be available. This represents a substantial increase in the available amount of inactivated influenza vaccine without thimerosal as a preservative, compared with approximately 3.2 million doses that were available during the 2003--04 influenza season. Inactivated influenza vaccine without thimerosal as a preservative is available from two manufacturers. Chiron produces Fluvirin™, which is approved by the Food and Drug Administration (FDA) for persons aged >4 years. Fluvirin is marketed as a formulation with thimerosal as a preservative in multidose vials and as a formulation without thimerosal as a preservative in 0.5-mL unit dose syringes. Aventis Pasteur produces FluZone®, which is FDA-approved for persons aged >6 months. FluZone containing thimerosal as a preservative is available in multidose vials. Preservative-free FluZone packaged as 0.25-mL unit dose syringes is available for use among persons aged 6--35 months. The total amount of inactivated influenza vaccine available without thimerosal as a preservative will be increased as manufacturing capabilities are expanded. The risks of severe illness from influenza infection are elevated among both young children and pregnant women, and both groups benefit from vaccination by preventing illness and death from influenza. In contrast, no scientifically conclusive evidence exists of harm from exposure to thimerosal preservative-containing vaccine, whereas evidence is accumulating of lack of any harm resulting from exposure to such vaccines (61,65). Therefore, the benefits of influenza vaccination outweigh the theoretical risk, if any, for thimerosal exposure through vaccination. Nonetheless, certain persons remain concerned regarding exposure to thimerosal. The U.S. vaccine supply for infants and pregnant women is in a period of transition during which thimerosal in vaccines intended for these groups is being reduced by manufacturers as a feasible means of reducing an infant's total exposure to mercury because other environmental sources of exposure are more difficult or impossible to eliminate. Reductions in thimerosal in other vaccines have been achieved already and have resulted in substantially lowered cumulative exposure to thimerosal from vaccination among infants and children. For all of these reasons, persons recommended to receive inactivated influenza vaccine may receive either vaccine preparation, depending on availability. Supplies of inactivated influenza vaccines without thimerosal as a preservative will be increased for the 2004--05 influenza season compared with the 2003--04 season, and they will be included in CDC contracts to meet anticipated public demand in 2004. Efficacy and Effectiveness of Inactivated Influenza Vaccine The effectiveness of inactivated influenza vaccine depends primarily on the age and immunocompetence of the vaccine recipient and the degree of similarity between the viruses in the vaccine and those in circulation. The majority of vaccinated children and young adults develop high postvaccination hemagglutination inhibition antibody titers (66--68). These antibody titers are protective against illness caused by strains similar to those in the vaccine (67--70). Adults Aged <65 Years. When the vaccine and circulating viruses are antigenically similar, influenza vaccine prevents influenza illness among approximately 70%--90% of healthy adults aged <65 years (9,12,71,72). Vaccination of healthy adults also has resulted in decreased work absenteeism and decreased use of health-care resources, including use of antibiotics, when the vaccine and circulating viruses are well-matched (9--12,72,73). Children. Children aged as young as 6 months can develop protective levels of antibody after influenza vaccination (66,67,74--77), although the antibody response among children at high risk for influenza-related complications might be lower than among healthy children (78,79). In a randomized study among children aged 1--15 years, inactivated influenza vaccine was 77%--91% effective against influenza respiratory illness and was 44%--49%, 74%--76%, and 70%--81% effective against influenza seroconversion among children aged 1--5, 6--10, and 11--15 years, respectively (68). One study (80) reported a vaccine efficacy of 56% against influenza illness among healthy children aged 3--9 years, and another study (81) determined vaccine efficacy of 22%--54% and 60%--78% among children with asthma aged 2--6 years and 7--14 years, respectively. A 2-year randomized study of children aged 6--24 months determined that >89% of children seroconverted to all three vaccine strains during both years (82). During year 1, among 411 children, vaccine efficacy was 66% (95% confidence interval [CI] = 34% and 82%) against culture-confirmed influenza (attack rates: 5.5% and 15.9% among vaccine and placebo groups, respectively). During year 2, among 375 children, vaccine efficacy was --7% (95% CI = --247% and 67%; attack rates: 3.6% and 3.3% among vaccine and placebo groups, respectively; the second year exhibited lower attack rates overall and was considered a mild season). However, no overall reduction in otitis media was reported (82). Other studies report that trivalent inactivated influenza vaccine decreases the incidence of influenza-associated otitis media among young children by approximately 30% (16,17). Adults Aged >65 Years. Older persons and persons with certain chronic diseases might develop lower postvaccination antibody titers than healthy young adults and thus can remain susceptible to influenza-related upper respiratory tract infection (83--85). A randomized trial among noninstitutionalized persons aged >60 years reported a vaccine efficacy of 58% against influenza respiratory illness, but indicated that efficacy might be lower among those aged >70 years (86). The vaccine can also be effective in preventing secondary complications and reducing the risk for influenza-related hospitalization and death among adults >65 years with and without high-risk medical conditions (e.g., heart disease and diabetes) (13--15,18,87). Among elderly persons not living in nursing homes or similar chronic-care facilities, influenza vaccine is 30%--70% effective in preventing hospitalization for pneumonia and influenza (15,88). Among older persons who do reside in nursing homes, influenza vaccine is most effective in preventing severe illness, secondary complications, and deaths. Among this population, the vaccine can be 50%--60% effective in preventing hospitalization or pneumonia and 80% effective in preventing death, although the effectiveness in preventing influenza illness often ranges from 30% to 40% (89--91). Efficacy and Effectiveness of LAIV Healthy Children. A randomized, double-blind, placebo-controlled trial among 1,602 healthy children initially aged 15--71 months assessed the efficacy of trivalent LAIV against culture-confirmed influenza during two seasons (92,93). This trial included subsets of 238 healthy children (163 vaccinees and 75 placebo recipients) aged 60--71 months who received 2 doses and 74 children (54 vaccinees and 20 placebo recipients) aged 60--71 months who received a single dose during season one, and a subset of 544 children (375 vaccinees and 169 placebo recipients) aged 60--84 months during season two. Children who continued from season one to season two remained in the same study group. In season one, when vaccine and circulating virus strains were well-matched, efficacy was 93% for all participants, regardless of age, among persons receiving 2 doses of LAIV. Efficacy was 87% in the 60--71-month subset for those who received 2 doses, and was 91% in the subset for those who received 1 or 2 doses. In season two, when the A (H3N2) component was not well-matched between vaccine and circulating virus strains, efficacy was 86% overall and 87% among those aged 60--84 months. The vaccine was 92% efficacious in preventing culture-confirmed influenza during the two-season study. Other results included a 27% reduction in febrile otitis media and a 28% reduction in otitis media with concomitant antibiotic use. Receipt of LAIV also resulted in decreased fever and otitis media among vaccine recipients who experienced influenza. Healthy Adults. A randomized, double-blind, placebo-controlled trial among 4,561 healthy working adults aged 18--64 years assessed multiple endpoints, including reductions in illness, absenteeism, health-care visits, and medication use during peak and total influenza outbreak periods (94). The study was conducted during the 1997--98 influenza season, when the vaccine and circulating A (H3N2) strains were not well-matched. The study did not include testing of viruses by a laboratory. During peak outbreak periods, no difference was identified between LAIV and placebo recipients experiencing any febrile episodes. However, vaccination was associated with reductions in severe febrile illnesses of 19% and febrile upper respiratory tract illnesses of 24%. Vaccination also was associated with fewer days of illness, fewer days of work lost, fewer days with health-care provider visits, and reduced use of prescription antibiotics and over-the-counter medications. Among the subset of 3,637 healthy adults aged 18--49 years, LAIV recipients (n = 2,411) had 26% fewer febrile upper-respiratory illness episodes; 27% fewer lost work days as a result of febrile upper respiratory illness; and 18%--37% fewer days of health-care provider visits caused by febrile illness, compared with placebo recipients (n = 1,226). Days of antibiotic use were reduced by 41%--45% in this age subset. Another randomized, double-blind, placebo-controlled challenge study among 92 healthy adults (LAIV, n = 29; placebo, n = 31; inactivated influenza vaccine, n = 32) aged 18--41 years assessed the efficacy of both LAIV and inactivated vaccine (95). The overall efficacy of LAIV and inactivated influenza vaccine in preventing laboratory-documented influenza from all three influenza strains combined was 85% and 71%, respectively, on the basis of experimental challenge by viruses to which study participants were susceptible before vaccination. The difference between the two vaccines was not statistically significant. Cost-Effectiveness of Influenza Vaccine Influenza vaccination can reduce both health-care costs and productivity losses associated with influenza illness. Economic studies of influenza vaccination of persons aged >65 years conducted in the United States have reported overall societal cost savings and substantial reductions in hospitalization and death (15,88,96). Studies of adults aged <65 years have reported that vaccination can reduce both direct medical costs and indirect costs from work absenteeism (8,10--12,72,97). Reductions of 34%--44% in physician visits, 32%--45% in lost workdays (10,12), and 25% in antibiotic use for influenza-associated illnesses have been reported (12). One cost-effectiveness analysis estimated a cost of approximately $60--$4,000/illness averted among healthy persons aged 18--64 years, depending on the cost of vaccination, the influenza attack rate, and vaccine effectiveness against influenza-like illness (72). Another cost-benefit economic model estimated an average annual savings of $13.66/person vaccinated (98). In the second study, 78% of all costs prevented were costs from lost work productivity, whereas the first study did not include productivity losses from influenza illness. Economic studies specifically evaluating the cost-effectiveness of vaccinating persons aged 50--64 years are not available, and the number of studies that examine the economics of routinely vaccinating children with inactivated or live, attenuated vaccine are limited (8,99--102). However, in a study of inactivated vaccine that included all age groups, cost utility improved with increasing age and among those with chronic medical conditions (8). Among persons aged >65 years, vaccination resulted in a net savings per quality-adjusted life year (QALY) gained and resulted in costs of $23--$256/QALY among younger age groups. Additional studies of the relative cost-effectiveness and cost utility of influenza vaccination among children and among adults aged <65 years are needed and should be designed to account for year-to-year variations in influenza attack rates, illness severity, and vaccine efficacy when evaluating the long-term costs and benefits of annual vaccination. Vaccination Coverage Levels Among persons aged >65 years, influenza vaccination levels increased from 33% in 1989 (103) to 66% in 1999 (104), surpassing the Healthy People 2000 objective of 60% (105). Vaccine coverage reached the highest levels recorded (68%) during the 1999--00 influenza season, using the percentage of adults reporting influenza vaccination during the past 12 months who participated in the National Health Interview Survey (NHIS) during the first and second quarters of each calendar year as a proxy measure of influenza vaccine coverage for the previous influenza season (104). Possible reasons for the increase in influenza vaccination levels among persons aged >65 years through the 1999--00 influenza season include 1) greater acceptance of preventive medical services by practitioners; 2) increased delivery and administration of vaccine by health-care providers and sources other than physicians; 3) new information regarding influenza vaccine effectiveness, cost-effectiveness, and safety; and 4) initiation of Medicare reimbursement for influenza vaccination in 1993 (8,14,15,89,90,106,107). Vaccine coverage increased more rapidly through the mid-1990s than during subsequent seasons (average annual percentage increase of 4% from 1988--89 to 1996--97 versus 1% from 1996--97 to 1999--00). Estimated national adult vaccine coverage for the 2001--02 season (Table 2), the most recent for which complete data are available, was 66% for adults aged >65 years and 34% for adults aged 50--64 years (104; unpublished data, CDC National Immunization Program, 2004). The estimated vaccination coverage among adults with high-risk conditions aged 18--49 years and 50--64 years was 23% and 44%, respectively, substantially lower than the Healthy People 2000 and 2010 objective of 60% (104,105,108). Continued annual monitoring is needed to determine the effects of vaccine supply delays, changes in influenza vaccination recommendations and target groups for vaccination, and other factors related to vaccination coverage among adults and children. The Healthy People 2010 objective is to achieve vaccination coverage for 90% of persons aged >65 years (108). Reducing racial and ethnic health disparities, including disparities in vaccination coverage, is an overarching national goal (108). Although estimated influenza vaccination coverage for the 1999--00 season reached the highest levels recorded among older black, Hispanic, and white populations, vaccination levels among blacks and Hispanics continue to lag behind those among whites (104,109). Estimated influenza vaccination levels for 2001 among persons aged >65 years were 66% among non-Hispanic whites, 48% among non-Hispanic blacks, and 54% among Hispanics (109,110). Additional strategies are needed to achieve the Healthy People 2010 objectives among all racial and ethnic groups. In 1997 and 1998, vaccination coverage estimates among nursing home residents were 64%--82% and 83%, respectively (111,112). The Healthy People 2010 goal is to achieve influenza vaccination of 90% among nursing home residents, an increase from the Healthy People 2000 goal of 80% (105,108). Reported vaccination levels are low among children at increased risk for influenza complications. One study conducted among patients in health maintenance organizations reported influenza vaccination percentages ranging from 9% to 10% among children with asthma (113). A 25% vaccination level was reported among children with severe to moderate asthma who attended an allergy and immunology clinic (114). However, a study conducted in a pediatric clinic demonstrated an increase in the vaccination percentage of children with asthma or reactive airways disease from 5% to 32% after implementing a reminder/recall system (115). One study reported 79% vaccination coverage among children attending a cystic fibrosis treatment center (116). Increasing vaccination coverage among persons who have high-risk conditions and are aged <65 years, including children at high risk, is the highest priority for expanding influenza vaccine use. Annual vaccination is recommended for health-care workers. Nonetheless, NHIS reported vaccination coverage of only 34% and 38% among health-care workers in the 1997 and 2002 surveys, respectively (117,118; unpublished data, CDC National Immunization Program, 2004) (Table 2). Vaccination of health-care workers has been associated with reduced work absenteeism (9) and fewer deaths among nursing home patients (119,120). Limited information is available regarding use of influenza vaccine among pregnant women. Among women aged 18--44 years without diabetes responding to the 2001 Behavioral Risk Factor Surveillance System, those reporting they were pregnant were less likely to report influenza vaccination during the past 12 months (13.7%) than those not pregnant (16.8%) (121). Only 12% of pregnant women reported vaccination according to 2002 NHIS data, excluding pregnant women who reported diabetes, heart disease, lung disease, and other selected high-risk conditions (unpublished data, CDC National Immunization Program, 2004) (Table 2). Although not directly measuring influenza vaccination among women who were past the first trimester of pregnancy during influenza season, these data indicate low compliance with the ACIP recommendations for pregnant women. In a study of influenza vaccine acceptance by pregnant women, 71% who were offered the vaccine chose to be vaccinated (122). However, a 1999 survey of obstetricians and gynecologists determined that only 39% administered influenza vaccine to obstetric patients, although 86% agreed that pregnant women's risk for influenza-related morbidity and mortality increases during the last two trimesters (123). Recent data indicate that self-report of influenza vaccination among adults, compared with extraction from the medical record, is both sensitive and specific. Patient self-reports should be accepted as evidence of influenza vaccination in clinical practice (124). However, information on the validity of parents' reports of pediatric influenza vaccination is not yet available. Recommendations for Using Inactivated and Live, Attenuated Influenza VaccinesBoth the inactivated influenza vaccine and LAIV can be used to reduce the risk of influenza. LAIV is only approved for use among healthy persons aged 5--49 years. Inactivated influenza vaccine is approved for persons aged >6 months, including those with high-risk conditions (see following sections on inactivated influenza vaccine and live, attenuated influenza vaccine). Target Groups for VaccinationPersons at Increased Risk for Complications Vaccination with inactivated influenza vaccine is recommended for the following persons who are at increased risk for complications from influenza:
In 2000, approximately 73 million persons in the United States were included in one or more of these target groups, including 35 million persons aged >65 years, 12 million adults aged 50--64 years, 18 million adults aged 18--49 years, and 8 million children aged 6 months--17 years with one or more medical conditions that are associated with an increased risk for influenza-related complications (125). Persons Aged 50--64 Years Vaccination is recommended for persons aged 50--64 years because this group has an increased prevalence of persons with high-risk conditions. In 2000, approximately 42 million persons in the United States were aged 50--64 years, of whom 12 million (29%) had one or more high-risk medical conditions (125). Influenza vaccine has been recommended for this entire age group to increase the low vaccination rates among persons in this age group with high-risk conditions (see preceding section). Age-based strategies are more successful in increasing vaccine coverage than patient-selection strategies based on medical conditions. Persons aged 50--64 years without high-risk conditions also receive benefit from vaccination in the form of decreased rates of influenza illness, decreased work absenteeism, and decreased need for medical visits and medication, including antibiotics (9--12). Further, 50 years is an age when other preventive services begin and when routine assessment of vaccination and other preventive services has been recommended (126,127). Persons Who Can Transmit Influenza to Those at High Risk Persons who are clinically or subclinically infected can transmit influenza virus to persons at high risk for complications from influenza. Decreasing transmission of influenza from caregivers and household contacts to persons at high risk might reduce influenza-related deaths among persons at high risk. Evidence from two studies indicates that vaccination of health-care personnel is associated with decreased deaths among nursing home patients (119,120). Health-care workers should be vaccinated against influenza annually. Facilities that employ heath-care workers are strongly encouraged to provide vaccine to workers by using approaches that maximize immunization rates. This will protect health-care workers, their patients, and communities, and will improve prevention, patient safety, and reduce disease burden. Health-care workers' influenza immunization rates should be regularly measured and reported. Although rates of health-care worker vaccination are typically <40%, with moderate effort, organized campaigns can attain higher rates of vaccination among this population (118). The following groups should be vaccinated:
In addition, because children aged 0--23 months are at increased risk for influenza-related hospitalization (52--54), vaccination is recommended for their household contacts and out-of-home caregivers, particularly for contacts of children aged 0--5 months, because influenza vaccines have not been approved by FDA for use among children aged <6 months (see Healthy Young Children). Healthy persons aged 5--49 years in these groups who are not contacts of severely immunosuppressed persons (see Live, Attenuated Influenza Vaccine Recommendations) can receive either LAIV or inactivated influenza vaccine. All other persons in this group should receive inactivated influenza vaccine. Additional Information Regarding Vaccination of Specific PopulationsPregnant Women Influenza-associated excess deaths among pregnant women were documented during the pandemics of 1918--19 and 1957--58 (128--131). Case reports and limited studies also indicate that pregnancy can increase the risk for serious medical complications of influenza (132--136). An increased risk might result from 1) increases in heart rate, stroke volume, and oxygen consumption; 2) decreases in lung capacity; and 3) changes in immunologic function during pregnancy. A study of the effect of influenza during 17 interpandemic influenza seasons demonstrated that the relative risk for hospitalization for selected cardiorespiratory conditions among pregnant women enrolled in Medicaid increased from 1.4 during weeks 14--20 of gestation to 4.7 during weeks 37--42, in comparison with women who were 1--6 months postpartum (137). Women in their third trimester of pregnancy were hospitalized at a rate (i.e., 250/100,000 pregnant women) comparable with that of nonpregnant women who had high-risk medical conditions. Researchers estimate that an average of 1--2 hospitalizations can be prevented for every 1,000 pregnant women vaccinated. Because of the increased risk for influenza-related complications, women who will be pregnant during the influenza season should be vaccinated. Vaccination can occur in any trimester. One study of influenza vaccination of >2,000 pregnant women demonstrated no adverse fetal effects associated with influenza vaccine (138). Healthy Young Children Studies indicate that rates of hospitalization are higher among young children than older children when influenza viruses are in circulation (51--53,139,140). The increased rates of hospitalization are comparable with rates for other groups considered at high risk for influenza-related complications. However, the interpretation of these findings has been confounded by co-circulation of respiratory syncytial viruses, which are a cause of serious respiratory viral illness among children and which frequently circulate during the same time as influenza viruses (141--143). Two recent studies have attempted to separate the effects of respiratory syncytial viruses and influenza viruses on rates of hospitalization among children who do not have high-risk conditions (52,53). Both studies reported that otherwise healthy children aged <2 years, and possibly children aged 2--4 years, are at increased risk for influenza-related hospitalization compared with older healthy children (Table 1). Among the Tennessee Medicaid population during 1973--1993, healthy children aged 6 months--<3 years had rates of influenza-associated hospitalization comparable with or higher than rates among children aged 3--14 years with high-risk conditions (Table 1) (52,54). Another Tennessee study reported a hospitalization rate per year of 3--4/1,000 healthy children aged <2 years for laboratory-confirmed influenza (32). Because children aged 6--23 months are at substantially increased risk for influenza-related hospitalizations, ACIP recommends vaccination of all children in this age group (144). ACIP continues to recommend influenza vaccination of persons aged >6 months who have high-risk medical conditions. The current inactivated influenza vaccine is not approved by FDA for use among children aged <6 months, the pediatric group at greatest risk for influenza-related complications (52). Vaccinating their household contacts and out-of-home caregivers might decrease the probability of influenza infection among these children. Beginning in March 2003, the group of children eligible for influenza vaccine coverage under the Vaccines for Children (VFC) program was expanded to include all VFC-eligible children aged 6--23 months and VFC-eligible children aged 2--18 years who are household contacts of children aged 0--23 months (145). Persons Infected with HIV Limited information is available regarding the frequency and severity of influenza illness or the benefits of influenza vaccination among persons with HIV infection (146,147). However, a retrospective study of young and middle-aged women enrolled in Tennessee's Medicaid program determined that the attributable risk for cardiopulmonary hospitalizations among women with HIV infection was higher during influenza seasons than during the peri-influenza periods. The risk for hospitalization was higher for HIV-infected women than for women with other well-recognized high-risk conditions, including chronic heart and lung diseases (148). Another study estimated that the risk for influenza-related death was 9.4--14.6/10,000 persons with acquired immunodeficiency syndrome (AIDS) compared with 0.09--0.10/10,000 among all persons aged 25--54 years and 6.4--7.0/10,000 among persons aged >65 years (149). Other reports indicate that influenza symptoms might be prolonged and the risk for complications from influenza increased for certain HIV-infected persons (150--152). Influenza vaccination has been demonstrated to produce substantial antibody titers against influenza among vaccinated HIV-infected persons who have minimal AIDS-related symptoms and high CD4+ T-lymphocyte cell counts (153--156). A limited, randomized, placebo-controlled trial determined that influenza vaccine was highly effective in preventing symptomatic, laboratory-confirmed influenza infection among HIV-infected persons with a mean of 400 CD4+ T-lymphocyte cells/mm3; a limited number of persons with CD4+ T-lymphocyte cell counts of <200 were included in that study (147). A nonrandomized study among HIV-infected persons determined that influenza vaccination was most effective among persons with >100 CD4+ cells and among those with <30,000 viral copies of HIV type-1/mL (152). Among persons who have advanced HIV disease and low CD4+ T-lymphocyte cell counts, influenza vaccine might not induce protective antibody titers (155,156); a second dose of vaccine does not improve the immune response in these persons (156,157). One study determined that HIV RNA (ribonucleic acid) levels increased transiently in one HIV-infected person after influenza infection (158). Studies have demonstrated a transient (i.e., 2--4 week) increase in replication of HIV-1 in the plasma or peripheral blood mononuclear cells of HIV-infected persons after vaccine administration (155,159). Other studies using similar laboratory techniques have not documented a substantial increase in the replication of HIV (160--163). Deterioration of CD4+ T-lymphocyte cell counts or progression of HIV disease have not been demonstrated among HIV-infected persons after influenza vaccination compared with unvaccinated persons (156,164). Limited information is available concerning the effect of antiretroviral therapy on increases in HIV RNA levels after either natural influenza infection or influenza vaccination (146,165). Because influenza can result in serious illness, and because influenza vaccination can result in the production of protective antibody titers, vaccination will benefit HIV-infected persons, including HIV-infected pregnant women. Breastfeeding Mothers Influenza vaccine does not affect the safety of mothers who are breastfeeding or their infants. Breastfeeding does not adversely affect the immune response and is not a contraindication for vaccination. Travelers The risk for exposure to influenza during travel depends on the time of year and destination. In the tropics, influenza can occur throughout the year. In the temperate regions of the Southern Hemisphere, the majority of influenza activity occurs during April--September. In temperate climate zones of the Northern and Southern Hemispheres, travelers also can be exposed to influenza during the summer, especially when traveling as part of large organized tourist groups (e.g., on cruise ships) that include persons from areas of the world where influenza viruses are circulating (166,167). Persons at high risk for complications of influenza who were not vaccinated with influenza vaccine during the preceding fall or winter should consider receiving influenza vaccine before travel if they plan to
No information is available regarding the benefits of revaccinating persons before summer travel who were already vaccinated in the preceding fall. Persons at high risk who receive the previous season's vaccine before travel should be revaccinated with the current vaccine the following fall or winter. Persons aged >50 years and others at high risk should consult with their physicians before embarking on travel during the summer to discuss the symptoms and risks for influenza and the advisability of carrying antiviral medications for either prophylaxis or treatment of influenza. General Population In addition to the groups for which annual influenza vaccination is recommended, physicians should administer influenza vaccine to any person who wishes to reduce the likelihood of becoming ill with influenza (the vaccine can be administered to children >6 months), depending on vaccine availability (see Influenza Vaccine Supply). Persons who provide essential community services should be considered for vaccination to minimize disruption of essential activities during influenza outbreaks. Students or other persons in institutional settings (e.g., those who reside in dormitories) should be encouraged to receive vaccine to minimize the disruption of routine activities during epidemics. Comparison of LAIV with Inactivated Influenza VaccineBoth inactivated influenza vaccine and LAIV are available to reduce the risk of influenza infection and illness. However, the vaccines also differ in key ways (Table 3). Major Similarities LAIV and inactivated influenza vaccine contain strains of influenza viruses that are antigenically equivalent to the annually recommended strains: one influenza A (H3N2) virus, one A (H1N1) virus, and one B virus. Each year, one or more virus strains might be changed on the basis of global surveillance for influenza viruses and the emergence and spread of new strains. Viruses for both vaccines are grown in eggs. Both vaccines are administered annually to provide optimal protection against influenza infection (Table 3). Major Differences Inactivated influenza vaccine contains killed viruses, whereas LAIV contains attenuated viruses still capable of replication. LAIV is administered intranasally by sprayer, whereas inactivated influenza vaccine is administered intramuscularly by injection. LAIV is more expensive than inactivated influenza vaccine. LAIV is approved for use only among healthy persons aged 5--49 years; inactivated influenza vaccine is approved for use among persons aged >6 months, including those who are healthy and those with chronic medical conditions (Table 3). Inactivated Influenza Vaccine RecommendationsPersons Who Should Not Be Vaccinated with Inactivated Influenza Vaccine Inactivated influenza vaccine should not be administered to persons known to have anaphylactic hypersensitivity to eggs or to other components of the influenza vaccine without first consulting a physician (see Side Effects and Adverse Reactions). Prophylactic use of antiviral agents is an option for preventing influenza among such persons. However, persons who have a history of anaphylactic hypersensitivity to vaccine components but who are also at high risk for complications from influenza can benefit from vaccine after appropriate allergy evaluation and desensitization. Information regarding vaccine components is located in package inserts from each manufacturer. Persons with acute febrile illness usually should not be vaccinated until their symptoms have abated. However, minor illnesses with or without fever do not contraindicate use of influenza vaccine, particularly among children with mild upper respiratory tract infection or allergic rhinitis. Dosage Dosage recommendations vary according to age group (Table 4). Among previously unvaccinated children aged <9 years, 2 doses administered >1 month apart are recommended for satisfactory antibody responses. If possible, the second dose should be administered before December. If a child aged <9 years receiving vaccine for the first time does not receive a second dose of vaccine within the same season, only 1 dose of vaccine should be administered the following season. Two doses are not required at that time. Among adults, studies have indicated limited or no improvement in antibody response when a second dose is administered during the same season (168--170). Even when the current influenza vaccine contains one or more antigens administered in previous years, annual vaccination with the current vaccine is necessary because immunity declines during the year after vaccination (171,172). Vaccine prepared for a previous influenza season should not be administered to provide protection for the current season. Route The intramuscular route is recommended for influenza vaccine. Adults and older children should be vaccinated in the deltoid muscle. A needle length >1 inch can be considered for these age groups because needles <1 inch might be of insufficient length to penetrate muscle tissue in certain adults and older children (173). Infants and young children should be vaccinated in the anterolateral aspect of the thigh (64). ACIP recommends a needle length of 7/8--1 inch for children aged <12 months for intramuscular vaccination into the anterolateral thigh. When injecting into the deltoid muscle among children with adequate deltoid muscle mass, a needle length of 7/8--1.25 inches is recommended (64). Side Effects and Adverse Reactions When educating patients regarding potential side effects, clinicians should emphasize that 1) inactivated influenza vaccine contains noninfectious killed viruses and cannot cause influenza; and 2) coincidental respiratory disease unrelated to influenza vaccination can occur after vaccination. Local Reactions In placebo-controlled studies among adults, the most frequent side effect of vaccination is soreness at the vaccination site (affecting 10%--64% of patients) that lasts <2 days (12,174--176). These local reactions typically are mild and rarely interfere with the person's ability to conduct usual daily activities. One blinded, randomized, cross-over study among 1,952 adults and children with asthma, demonstrated that only body aches were reported more frequently after inactivated influenza vaccine (25.1%) than placebo-injection (20.8%) (177). One study (79) reported 20%--28% of children with asthma aged 9 months--18 years with local pain and swelling and another study (77) reported 23% of children aged 6 months--4 years with chronic heart or lung disease had local reactions. A different study (76) reported no difference in local reactions among 53 children aged 6 months--6 years with high-risk medical conditions or among 305 healthy children aged 3--12 years in a placebo-controlled trial of inactivated influenza vaccine. In a study of 12 children aged 5--32 months, no substantial local or systemic reactions were noted (178). Systemic Reactions Fever, malaise, myalgia, and other systemic symptoms can occur after vaccination with inactivated vaccine and most often affect persons who have had no prior exposure to the influenza virus antigens in the vaccine (e.g., young children) (179,180). These reactions begin 6--12 hours after vaccination and can persist for 1--2 days. Recent placebo-controlled trials demonstrate that among older persons and healthy young adults, administration of split-virus influenza vaccine is not associated with higher rates of systemic symptoms (e.g., fever, malaise, myalgia, and headache) when compared with placebo injections (12,174--176). Less information from published studies is available for children, compared with adults. However, in a randomized cross-over study among both children and adults with asthma, no increase in asthma exacerbations was reported for either age group (177). An analysis of 215,600 children aged <18 years and 8,476 children aged 6--23 months enrolled in one of five health maintenance organizations reported no increase in biologically plausible medically attended events during the 2 weeks after inactivated influenza vaccination, compared with control periods 3--4 weeks before and after vaccination (181). In a study of 791 healthy children (68), postvaccination fever was noted among 11.5% of children aged 1--5 years, 4.6% among children aged 6--10 years, and 5.1% among children aged 11--15 years. Among children with high-risk medical conditions, one study of 52 children aged 6 months--4 years reported fever among 27% and irritability and insomnia among 25% (77); and a study among 33 children aged 6--18 months reported that one child had irritability and one had a fever and seizure after vaccination (182). No placebo comparison was made in these studies. However, in pediatric trials of A/New Jersey/76 swine influenza vaccine, no difference was reported between placebo and split-virus vaccine groups in febrile reactions after injection, although the vaccine was associated with mild local tenderness or erythema (76). Limited data regarding potential adverse events after influenza vaccination are available from the Vaccine Adverse Event Reporting System (VAERS). During January 1, 1991--January 23, 2003, VAERS received 1,072 reports of adverse events among children aged <18 years, including 174 reports of adverse events among children aged 6--23 months. The number of influenza vaccine doses received by children during this time period is unknown. The most frequently reported events among children were fever, injection-site reactions, and rash (unpublished data, CDC, 2003). Because of the limitations of spontaneous reporting systems, determining causality for specific types of adverse events, with the exception of injection-site reactions, is usually not possible by using VAERS data alone. Health-care professionals should promptly report all clinically significant adverse events after influenza vaccination of children to VAERS, even if the health-care professional is not certain that the vaccine caused the event. The Institute of Medicine has specifically recommended reporting of potential neurologic complications (e.g., demyelinating disorders such as Guillain-Barré [GBS] syndrome), although no evidence exists of a causal relationship between influenza vaccine and neurologic disorders in children. Immediate --- presumably allergic --- reactions (e.g., hives, angioedema, allergic asthma, and systemic anaphylaxis) rarely occur after influenza vaccination (183). These reactions probably result from hypersensitivity to certain vaccine components; the majority of reactions probably are caused by residual egg protein. Although current influenza vaccines contain only a limited quantity of egg protein, this protein can induce immediate hypersensitivity reactions among persons who have severe egg allergy. Persons who have had hives or swelling of the lips or tongue, or who have experienced acute respiratory distress or collapse after eating eggs should consult a physician for appropriate evaluation to help determine if vaccine should be administered. Persons who have documented immunoglobulin E (IgE)-mediated hypersensitivity to eggs, including those who have had occupational asthma or other allergic responses to egg protein, might also be at increased risk for allergic reactions to influenza vaccine, and consultation with a physician should be considered. Protocols have been published for safely administering influenza vaccine to persons with egg allergies (184--186). Hypersensitivity reactions to any vaccine component can occur. Although exposure to vaccines containing thimerosal can lead to induction of hypersensitivity, the majority of patients do not have reactions to thimerosal when it is administered as a component of vaccines, even when patch or intradermal tests for thimerosal indicate hypersensitivity (187,188). When reported, hypersensitivity to thimerosal usually has consisted of local, delayed hypersensitivity reactions (187). Guillain-Barré Syndrome The 1976 swine influenza vaccine was associated with an increased frequency of GBS (189,190). Among persons who received the swine influenza vaccine in 1976, the rate of GBS was <10 cases/1 million persons vaccinated. The risk for influenza vaccine-associated GBS is higher among persons aged >25 years than persons <25 years (189). Evidence for a causal relation of GBS with subsequent vaccines prepared from other influenza viruses is unclear. Obtaining strong epidemiologic evidence for a possible limited increase in risk is difficult for such a rare condition as GBS, which has an annual incidence of 10--20 cases/1 million adults (191). More definitive data probably will require using other methodologies (e.g., laboratory studies of the pathophysiology of GBS). During three of four influenza seasons studied during 1977--1991, the overall relative risk estimates for GBS after influenza vaccination were slightly elevated but were not statistically significant in any of these studies (192--194). However, in a study of the 1992--93 and 1993--94 seasons, the overall relative risk for GBS was 1.7 (95% CI = 1.0--2.8; p = 0.04) during the 6 weeks after vaccination, representing approximately 1 additional case of GBS/1 million persons vaccinated. The combined number of GBS cases peaked 2 weeks after vaccination (195). Thus, investigations to date indicate no substantial increase in GBS associated with influenza vaccines (other than the swine influenza vaccine in 1976), and that, if influenza vaccine does pose a risk, it is probably slightly more than one additional case/1 million persons vaccinated. Cases of GBS after influenza infection have been reported, but no epidemiologic studies have documented such an association (196,197). Substantial evidence exists that multiple infectious illnesses, most notably Campylobacter jejuni, as well as upper respiratory tract infections are associated with GBS (191,198--200). Even if GBS were a true side effect of vaccination in the years after 1976, the estimated risk for GBS of approximately 1 additional case/1 million persons vaccinated is substantially less than the risk for severe influenza, which can be prevented by vaccination among all age groups, especially persons aged >65 years and those who have medical indications for influenza vaccination (Table 1) (see Hospitalizations and Deaths from Influenza). The potential benefits of influenza vaccination in preventing serious illness, hospitalization, and death substantially outweigh the possible risks for experiencing vaccine-associated GBS. The average case fatality ratio for GBS is 6% and increases with age (191,201). No evidence indicates that the case fatality ratio for GBS differs among vaccinated persons and those not vaccinated. The incidence of GBS among the general population is low, but persons with a history of GBS have a substantially greater likelihood of subsequently experiencing GBS than persons without such a history (192,202). Thus, the likelihood of coincidentally experiencing GBS after influenza vaccination is expected to be greater among persons with a history of GBS than among persons with no history of this syndrome. Whether influenza vaccination specifically might increase the risk for recurrence of GBS is unknown; therefore, avoiding vaccinating persons who are not at high risk for severe influenza complications and who are known to have experienced GBS within 6 weeks after a previous influenza vaccination is prudent. As an alternative, physicians might consider using influenza antiviral chemoprophylaxis for these persons. Although data are limited, for the majority of persons who have a history of GBS and who are at high risk for severe complications from influenza, the established benefits of influenza vaccination justify yearly vaccination. Live, Attenuated Influenza Vaccine RecommendationsBackground Description and Action Mechanisms. LAIVs have been in development since the 1960s in the United States, where they have been evaluated as mono-, bi-, and trivalent formulations (203--207). The LAIV licensed for use in the United States beginning in 2003 is produced by MedImmune, Inc. (Gaithersburg, Maryland; http://www.medimmune.com) and marketed under the name FluMist™. It is a live, trivalent, intranasally administered vaccine that is
In animal studies, LAIV viruses replicate in the mucosa of the nasopharynx, inducing protective immunity against viruses included in the vaccine, but replicate inefficiently in the lower airways or lungs. The first step in developing an LAIV was the derivation of two stably attenuated master donor viruses (MDV), one for type A and one for type B influenza viruses. The two MDVs each acquired the cold-adapted, temperature-sensitive, attenuated phenotypes through serial passage in viral culture conducted at progressively lower temperatures. The vaccine viruses in LAIV are reassortant viruses containing genes from these MDVs that confer attenuation, temperature sensitivity, and cold adaptation and genes from the recommended contemporary wild-type influenza viruses, encoding the surface antigens hemagglutinin (HA) and neuraminidase (NA). Thus, MDVs provide the stably attenuated vehicles for presenting influenza HA and NA antigens, to which the protective antibody response is directed, to the immune system. The reassortant vaccine viruses are grown in embryonated hens' eggs. After the vaccine is formulated and inserted into individual sprayers for nasal administration, the vaccine must be stored at --15ºC or colder. The immunogenicity of the approved LAIV has been assessed in multiple studies (96,208--213), which included approximately 100 children aged 5--17 years, and approximately 300 adults aged 18--49 years. LAIV virus strains replicate primarily in nasopharyngeal epithelial cells. The protective mechanisms induced by vaccination with LAIV are not completely understood but appear to involve both serum and nasal secretory antibodies. No single laboratory measurement closely correlates with protective immunity induced by LAIV. Shedding and Transmission of Vaccine Viruses. Available data indicate that both children and adults vaccinated with LAIV can shed vaccine viruses for >2 days after vaccination, although in lower titers than typically occur with shedding of wild-type influenza viruses. Shedding should not be equated with person-to-person transmission of vaccine viruses, although, in rare instances, shed vaccine viruses can be transmitted from vaccinees to nonvaccinated persons. One unpublished study in a child care center setting assessed transmissibility of vaccine viruses from 98 vaccinated to 99 unvaccinated subjects, all aged 8--36 months. Eighty percent of vaccine recipients shed one or more virus strains, with a mean of 7.6 days' duration (214). One vaccine type influenza type B isolate was recovered from a placebo recipient and was confirmed to be vaccine-type virus. The type B isolate retained the cold-adapted, temperature-sensitive, attenuated phenotype, and it possessed the same genetic sequence as a virus shed from a vaccine recipient in the same children's play group. The placebo recipient from whom the influenza type B vaccine virus was isolated did not exhibit symptoms that were different from those experienced by vaccine recipients. The estimated probability of acquiring vaccine virus after close contact with a single LAIV recipient in this child care population was 0.58%--2.4%. One study assessing shedding of vaccine viruses in 20 healthy vaccinated adults aged 18--49 years demonstrated that the majority of shedding occurred within the first 3 days after vaccination, although one subject was noted to shed virus on day 7 after vaccine receipt. No subject shed vaccine viruses >10 days after vaccination. Duration or type of symptoms associated with receipt of LAIV did not correlate with duration of shedding of vaccine viruses. Person-to-person transmission of vaccine viruses was not assessed in this study (215). Stability of Vaccine Viruses. In clinical trials, viruses shed by vaccine recipients have been phenotypically stable. In one study, nasal and throat swab specimens were collected from 17 study participants for 2 weeks after vaccine receipt (216). Virus isolates were analyzed by multiple genetic techniques. All isolates retained the LAIV genotype after replication in the human host, and all retained the cold-adapted and temperature-sensitive phenotypes. Using Live, Attenuated Influenza Vaccine LAIV is an option for vaccination of healthy persons aged 5--49 years, including persons in close contact with groups at high risk and those wanting to avoid influenza. Possible advantages of LAIV include its potential to induce a broad mucosal and systemic immune response, its ease of administration, and the acceptability of an intranasal rather than intramuscular route of administration. Persons Who Should Not Be Vaccinated with LAIV The following populations should not be vaccinated with LAIV:
Close Contacts of Persons at High Risk for Complications from Influenza Close contacts of persons at high risk for complications from influenza should receive influenza vaccine to reduce transmission of wild-type influenza viruses to persons at high risk. Use of inactivated influenza vaccine is preferred for vaccinating household members, health-care workers, and others who have close contact with severely immunosuppressed persons (e.g., patients with hematopoietic stem cell transplants) during those periods in which the immunosuppressed person requires care in a protective environment. The rationale for not using LAIV among health-care workers caring for such patients is the theoretical risk that a live, attenuated vaccine virus could be transmitted to the severely immunosuppressed person and cause disease. No preference exists for inactivated influenza vaccine use by health-care workers or other persons who have close contact with persons with lesser degrees of immunosuppression (e.g., persons with diabetes, persons with asthma taking corticosteroids, or persons infected with human immunodeficiency virus), and no preference exists for inactivated influenza vaccine use by health-care workers or other healthy persons aged 5--49 years in close contact with all other groups at high risk. If a health-care worker receives LAIV, that worker should refrain from contact with severely immunosuppressed patients as described previously for 7 days after vaccine receipt. Hospital visitors who have received LAIV should refrain from contact with severely immunosuppressed persons for 7 days after vaccination; however, such persons need not be excluded from visitation of patients who are not severely immunosuppressed. Personnel Who May Administer LAIV Low-level introduction of vaccine viruses into the environment is likely unavoidable when administering LAIV. The risk of acquiring vaccine viruses from the environment is unknown but likely to be limited. Severely immunosuppressed persons should not administer LAIV. However, other persons at high risk for influenza complications may administer LAIV. These include persons with underlying medical conditions placing them at high risk or who are likely to be at risk, including pregnant women, persons with asthma, and persons aged >50 years. LAIV Dosage and Administration LAIV is intended for intranasal administration only and should not be administered by the intramuscular, intradermal, or intravenous route. LAIV must be stored at --15ºC or colder. LAIV should not be stored in a frost-free freezer (because the temperature might cycle above --15ºC), unless a manufacturer-supplied freezer box is used. LAIV must be thawed before administration. This can be accomplished by holding an individual sprayer in the palm of the hand until thawed, with subsequent immediate administration. Alternatively, the vaccine can be thawed in a refrigerator and stored at 2ºC--8ºC for <24 hours before use. Vaccine should not be refrozen after thawing. LAIV is supplied in a prefilled single-use sprayer containing 0.5 mL of vaccine. Approximately 0.25 mL (i.e., half of the total sprayer contents) is sprayed into the first nostril while the recipient is in the upright position. An attached dose-divider clip is removed from the sprayer to administer the second half of the dose into the other nostril. If the vaccine recipient sneezes after administration, the dose should not be repeated. LAIV should be administered annually according to the following schedule:
LAIV and Use of Influenza Antiviral Medications The effect on safety and efficacy of LAIV coadministration with influenza antiviral medications has not been studied. However, because influenza antivirals reduce replication of influenza viruses, LAIV should not be administered until 48 hours after cessation of influenza antiviral therapy, and influenza antiviral medications should not be administered for 2 weeks after receipt of LAIV. LAIV Storage LAIV must be stored at --15ºC or colder. LAIV should not be stored in a frost-free freezer because the temperature might cycle above --15ºC, unless a manufacturer-supplied freezer box or other strategy is used. LAIV can be thawed in a refrigerator and stored at 2ºC--8ºC for <24 hours before use. It should not be refrozen after thawing. Additional information is available at Wyeth Product Quality (1-800-411-0086) or at http://www.FluMist.com. Side Effects and Adverse Reactions Twenty prelicensure clinical trials assessed the safety of the approved LAIV. In these combined studies, approximately 28,000 doses of the vaccine were administered to >20,000 subjects. A subset of these trials were randomized, placebo-controlled studies in which >4,000 healthy children aged 5--17 years and >2,000 healthy adults aged 18--49 years were vaccinated. The incidence of adverse events possibly complicating influenza (e.g., pneumonia, bronchitis, bronchiolitis, or central nervous system events) was not statistically different among LAIV and placebo recipients aged 5--49 years. Children. Signs and symptoms reported more often among vaccine recipients than placebo recipients included runny nose or nasal congestion (20%--75%), headache (2%--46%), fever (0%--26%), and vomiting (3%--13%), abdominal pain (2%), and myalgias (0%--21%) (208,211,213,217--219). These symptoms were associated more often with the first dose and were self-limited. In a subset of healthy children aged 60--71 months from one clinical trial (92,93), certain signs and symptoms were reported more often among LAIV recipients after the first dose (n = 214) than placebo recipients (n = 95) (e.g., runny nose, 48.1% versus 44.2%; headache, 17.8% versus 11.6%; vomiting, 4.7% versus 3.2%; myalgias, 6.1% versus 4.2%), but these differences were not statistically significant. Unpublished data from a study including subjects aged 1--17 years indicated an increase in asthma or reactive airways disease in the subset aged 12--59 months. Because of this, LAIV is not approved for use among children aged <60 months. Adults. Among adults, runny nose or nasal congestion (28%--78%), headache (16%--44%), and sore throat (15%--27%) have been reported more often among vaccine recipients than placebo recipients (94,220,221). In one clinical trial (94), among a subset of healthy adults aged 18--49 years, signs and symptoms reported more frequently among LAIV recipients (n = 2,548) than placebo recipients (n = 1,290) within 7 days after each dose included cough (13.9% versus 10.8%); runny nose (44.5% versus 27.1%); sore throat (27.8% versus 17.1%); chills (8.6% versus 6.0%); and tiredness/weakness (25.7% versus 21.6%). Safety Among Groups at High Risk from Influenza-Related Morbidity. Until additional data are acquired, persons at high risk for experiencing complications from influenza infection (e.g., immunocompromised patients; patients with asthma, cystic fibrosis, or chronic obstructive pulmonary disease; or persons aged >65 years) should not be vaccinated with LAIV. Protection from influenza among these groups should be accomplished by using inactivated influenza vaccine. Serious Adverse Events. Serious adverse events among healthy children aged 5--17 years or healthy adults aged 18--49 years occurred at a rate of <1%. Surveillance should continue for adverse events that might not have been detected in previous studies. Health-care professionals should promptly report all clinically significant adverse events after LAIV administration to VAERS, as recommended for inactivated influenza vaccine. Recommended Vaccines for Different Age GroupsWhen vaccinating children aged 6 months--3 years, health-care providers should use inactivated influenza vaccine that has been approved by FDA for this age group. Inactivated influenza vaccine from Aventis Pasteur, Inc., (FluZone split-virus) is approved for use among persons aged >6 months. Inactivated influenza vaccine from Chiron (Fluvirin) is labeled in the United States for use only among persons aged >4 years because data to demonstrate efficacy among younger persons have not been provided to FDA. Live, attenuated influenza vaccine from MedImmune (FluMist) is approved for use by healthy persons aged 5--49 years (Table 5). Timing of Annual Influenza VaccinationThe annual supply of inactivated influenza vaccine and the timing of its distribution cannot be guaranteed in any year. Information regarding the supply of 2004--05 vaccine might not be available until late summer or early fall 2004. To allow vaccine providers to plan for the upcoming vaccination season, taking into account the yearly possibility of vaccine delays or shortages and the need to ensure vaccination of persons at high risk and their contacts, ACIP recommends that vaccine campaigns conducted in October focus their efforts primarily on persons at increased risk for influenza complications and their contacts, including health-care workers. Campaigns conducted in November and later should continue to vaccinate persons at high risk and their contacts, but also vaccinate other persons who wish to decrease their risk for influenza infection. Vaccination efforts for all groups should continue into December and beyond. CDC and other public health agencies will assess the vaccine supply on a continuing basis throughout the manufacturing period and will make recommendations in the summer preceding the 2004--05 influenza season regarding the need for tiered timing of vaccination of different risk groups. Vaccination in October and November The optimal time to vaccinate is usually during October--November. ACIP recommends that vaccine providers focus their vaccination efforts in October and earlier primarily on persons aged >50 years, persons aged <50 years at increased risk for influenza-related complications (including children aged 6--23 months), household contacts of persons at high risk (including out-of-home caregivers and household contacts of children aged 0--23 months), and health-care workers. Vaccination of children aged <9 years who are receiving vaccine for the first time should also begin in October or earlier because those persons need a booster dose 1 month after the initial dose. Efforts to vaccinate other persons who wish to decrease their risk for influenza infection should begin in November; however, if such persons request vaccination in October, vaccination should not be deferred. Materials to assist providers in prioritizing early vaccine are available at http://www.cdc.gov/flu/professionals/vaccination/index.htm (see also Travelers in this report). Timing of Organized Vaccination Campaigns Persons planning substantial organized vaccination campaigns should consider scheduling these events after mid-October because the availability of vaccine in any location cannot be ensured consistently in early fall. Scheduling campaigns after mid-October will minimize the need for cancellations because vaccine is unavailable. Campaigns conducted before November should focus efforts on vaccination of persons aged >50 years, persons aged <50 years at increased risk for influenza-related complications (including children aged 6--23 months), health-care workers, and household contacts of persons at high-risk (including children aged 0--23 months) to the extent feasible. Vaccination in December and Later After November, many persons who should or want to receive influenza vaccine remain unvaccinated. In addition, substantial amounts of vaccine have remained unused during three of the past four influenza seasons. To improve vaccine coverage, influenza vaccine should continue to be offered in December and throughout the influenza season as long as vaccine supplies are available, even after influenza activity has been documented in the community. In the United States, seasonal influenza activity can begin to increase as early as October or November, but influenza activity has not reached peak levels in the majority of recent seasons until late December--early March (Table 6). Therefore, although the timing of influenza activity can vary by region, vaccine administered after November is likely to be beneficial in the majority of influenza seasons. Adults develop peak antibody protection against influenza infection 2 weeks after vaccination (222,223). Vaccination Before October To avoid missed opportunities for vaccination of persons at high risk for serious complications, such persons should be offered vaccine beginning in September during routine health-care visits or during hospitalizations, if vaccine is available. In facilities housing older persons (e.g., nursing homes), vaccination before October typically should be avoided because antibody levels in such persons can begin to decline within a limited time after vaccination (224). In addition, children aged <9 years who have not been previously vaccinated and who need 2 doses before the start of the influenza season can receive their first dose in September or earlier. Strategies for Implementing Vaccination Recommendations in Health-Care SettingsSuccessful vaccination programs combine publicity and education for health-care workers and other potential vaccine recipients, a plan for identifying persons at high risk, use of reminder/recall systems, and efforts to remove administrative and financial barriers that prevent persons from receiving the vaccine, including use of standing orders programs (19,225). Using standing orders programs is recommended for long-term--care facilities (e.g., nursing homes and skilled nursing facilities), hospitals, and home health agencies to ensure the administration of recommended vaccinations for adults (226). Standing orders programs for both influenza and pneumococcal vaccination should be conducted under the supervision of a licensed practitioner according to a physician-approved facility or agency policy by health-care personnel trained to screen patients for contraindications to vaccination, administer vaccine, and monitor for adverse events. The Centers for Medicare and Medicaid Services (CMS) has removed the physician signature requirement for the administration of influenza and pneumococcal vaccines to Medicare and Medicaid patients in hospitals, long-term--care facilities, and home health agencies (226). To the extent allowed by local and state law, these facilities and agencies may implement standing orders for influenza and pneumococcal vaccination of Medicare- and Medicaid-eligible patients. Other settings (e.g., outpatient facilities, managed care organizations, assisted living facilities, correctional facilities, pharmacies, and adult workplaces) are encouraged to introduce standing orders programs as well (20). Persons for whom influenza vaccine is recommended can be identified and vaccinated in the settings described in the following sections. Outpatient Facilities Providing Ongoing Care Staff in facilities providing ongoing medical care (e.g., physicians' offices, public health clinics, employee health clinics, hemodialysis centers, hospital specialty-care clinics, and outpatient rehabilitation programs) should identify and label the medical records of patients who should receive vaccination. Vaccine should be offered during visits beginning in September and throughout the influenza season. The offer of vaccination and its receipt or refusal should be documented in the medical record. Patients for whom vaccination is recommended and who do not have regularly scheduled visits during the fall should be reminded by mail, telephone, or other means of the need for vaccination. Outpatient Facilities Providing Episodic or Acute Care Beginning each September, acute health-care facilities (e.g., emergency rooms and walk-in clinics) should offer vaccinations to persons for whom vaccination is recommended or provide written information regarding why, where, and how to obtain the vaccine. This written information should be available in languages appropriate for the populations served by the facility. Nursing Homes and Other Residential Long-Term--Care Facilities During October and November each year, vaccination should be routinely provided to all residents of chronic-care facilities with the concurrence of attending physicians. Consent for vaccination should be obtained from the resident or a family member at the time of admission to the facility or anytime afterwards. All residents should be vaccinated at one time, preceding the influenza season. Residents admitted through March after completion of the facility's vaccination program should be vaccinated at the time of admission. Acute-Care Hospitals Persons of all ages (including children) with high-risk conditions and persons aged >50 years who are hospitalized at any time during September--March should be offered and strongly encouraged to receive influenza vaccine before they are discharged. In one study, 39%--46% of adult patients hospitalized during the winter with influenza-related diagnoses had been hospitalized during the preceding autumn (227). Thus, the hospital serves as a setting in which persons at increased risk for subsequent hospitalization can be identified and vaccinated. However, vaccination of persons at high risk during or after their hospitalizations is often not done. In a study of hospitalized Medicare patients, only 31.6% were vaccinated before admission, 1.9% during admission, and 10.6% after admission (228). Using standing orders in hospitals increases vaccination rates among hospitalized persons (229). Visiting Nurses and Others Providing Home Care to Persons at High Risk Beginning in September, nursing-care plans should identify patients for whom vaccination is recommended, and vaccine should be administered in the home, if necessary. Caregivers and other persons in the household (including children) should be referred for vaccination. Other Facilities Providing Services to Persons Aged >50 Years Beginning in October, such facilities as assisted living housing, retirement communities, and recreation centers should offer unvaccinated residents and attendees vaccination on-site before the influenza season. Staff education should emphasize the need for influenza vaccine. Health-Care Personnel Beginning in October each year, health-care facilities should offer influenza vaccinations to all personnel, including night and weekend staff. Particular emphasis should be placed on providing vaccinations to persons who care for members of groups at high risk. Efforts should be made to educate health-care personnel regarding the benefits of vaccination and the potential health consequences of influenza illness for themselves and their patients. All health-care personnel should be provided convenient access to influenza vaccine at the work site, free of charge, as part of employee health programs (118). Influenza Vaccine SupplyDuring the 2002--03 season, approximately 95 million doses of influenza vaccine were produced, but 12 million doses went unused and had to be destroyed. During the 2003--04 season, approximately 87 million doses of vaccine were produced. During that season, shortages of vaccine were noted in multiple regions of the United States after an unprecedented demand for vaccine lasted longer into the season than usual, caused in part by increased media attention to influenza. On the basis of early projections, manufacturers anticipate production of 90--100 million doses of vaccine for the 2004--05 season. Influenza vaccine delivery delays or vaccine shortages remain possible in part because of the inherent critical time constraints in manufacturing the vaccine given the annual updating of the influenza vaccine strains. Steps being taken to address possible future delays or vaccine shortages include identification and implementation of ways to expand the influenza vaccine supply and improvement of targeted delivery of vaccine to groups at high risk when delays or shortages are expected. Future DirectionsACIP plans to review new vaccination strategies for improving prevention and control of influenza, including the possibility of expanding recommendations for use of influenza vaccines. In addition, strategies for regularly monitoring vaccine effectiveness will be reviewed. Recommendations for Using Antiviral Agents for InfluenzaAntiviral drugs for influenza are an adjunct to influenza vaccine for controlling and preventing influenza. However, these agents are not a substitute for vaccination. Four licensed influenza antiviral agents are available in the United States: amantadine, rimantadine, zanamivir, and oseltamivir. Amantadine and rimantadine are chemically related antiviral drugs known as adamantanes with activity against influenza A viruses but not influenza B viruses. Amantadine was approved in 1966 for chemoprophylaxis of influenza A (H2N2) infection and was later approved in 1976 for treatment and chemoprophylaxis of influenza type A virus infections among adults and children aged >1 year. Rimantadine was approved in 1993 for treatment and chemoprophylaxis of influenza A infection among adults and prophylaxis among children. Although rimantadine is approved only for chemoprophylaxis of influenza A infection among children, certain specialists in the management of influenza consider it appropriate for treatment of influenza A among children (230). Zanamivir and oseltamivir are chemically related antiviral drugs known as neuraminidase inhibitors that have activity against both influenza A and B viruses. Both zanamivir and oseltamivir were approved in 1999 for treating uncomplicated influenza infections. Zanamivir is approved for treating persons aged >7 years, and oseltamivir is approved for treatment for persons aged >1 year. In 2000, oseltamivir was approved for chemoprophylaxis of influenza among persons aged >13 years. The four drugs differ in pharmacokinetics, side effects, routes of administration, approved age groups, dosages, and costs. An overview of the indications, use, administration, and known primary side effects of these medications is presented in the following sections. Information contained in this report might not represent FDA approval or approved labeling for the antiviral agents described. Package inserts should be consulted for additional information. Role of Laboratory DiagnosisAppropriate treatment of patients with respiratory illness depends on accurate and timely diagnosis. Early diagnosis of influenza can reduce the inappropriate use of antibiotics and provide the option of using antiviral therapy. However, because certain bacterial infections can produce symptoms similar to influenza, bacterial infections should be considered and appropriately treated, if suspected. In addition, bacterial infections can occur as a complication of influenza. Influenza surveillance information and diagnostic testing can aid clinical judgment and help guide treatment decisions. The accuracy of clinical diagnosis of influenza on the basis of symptoms alone is limited because symptoms from illness caused by other pathogens can overlap considerably with influenza (29,33,34). Influenza surveillance by state and local health departments and CDC can provide information regarding the presence of influenza viruses in the community. Surveillance can also identify the predominant circulating types, subtypes, and strains of influenza. Diagnostic tests available for influenza include viral culture, serology, rapid antigen testing, polymerase chain reaction (PCR) and immunofluorescence (24). Sensitivity and specificity of any test for influenza might vary by the laboratory that performs the test, the type of test used, and the type of specimen tested. Among respiratory specimens for viral isolation or rapid detection, nasopharyngeal specimens are typically more effective than throat swab specimens (231). As with any diagnostic test, results should be evaluated in the context of other clinical information available to health-care providers. Commercial rapid diagnostic tests are available that can be used by laboratories in outpatient settings to detect influenza viruses within 30 minutes (24,232). These rapid tests differ in the types of influenza viruses they can detect and whether they can distinguish between influenza types. Different tests can detect 1) only influenza A viruses; 2) both influenza A and B viruses, but not distinguish between the two types; or 3) both influenza A and B and distinguish between the two. The types of specimens acceptable for use (i.e., throat swab, nasal wash, or nasal swab) also vary by test. The specificity and, in particular, the sensitivity of rapid tests are lower than for viral culture and vary by test (233,234). Because of the lower sensitivity of the rapid tests, physicians should consider confirming negative tests with viral culture or other means. Further, when interpreting results of a rapid influenza test, physicians should consider the positive and negative predictive values of the test in the context of the level of influenza activity in their community. Package inserts and the laboratory performing the test should be consulted for more details regarding use of rapid diagnostic tests. Additional information concerning diagnostic testing is located at http://www.cdc. gov/flu/professionals/labdiagnosis.htm. Despite the availability of rapid diagnostic tests, collecting clinical specimens for viral culture is critical, because only culture isolates can provide specific information regarding circulating influenza subtypes and strains. This information is needed to compare current circulating influenza strains with vaccine strains, to guide decisions regarding influenza treatment and chemoprophylaxis, and to formulate vaccine for the coming year. Virus isolates also are needed to monitor the emergence of antiviral resistance and the emergence of novel influenza A subtypes that might pose a pandemic threat. Indications for UseTreatment When administered within 2 days of illness onset to otherwise healthy adults, amantadine and rimantadine can reduce the duration of uncomplicated influenza A illness, and zanamivir and oseltamivir can reduce the duration of uncomplicated influenza A and B illness by approximately 1 day, compared with placebo (72,235--249). More clinical data are available concerning the efficacy of zanamivir and oseltamivir for treatment of influenza A infection than for treatment of influenza B infection (250--266). However, in vitro data and studies of treatment among mice and ferrets (267--274), in addition to clinical studies, have documented that zanamivir and oseltamivir have activity against influenza B viruses (241,245--247,275,276). Data are limited regarding the effectiveness of the four antiviral agents in preventing serious influenza-related complications (e.g., bacterial or viral pneumonia or exacerbation of chronic diseases). Evidence for the effectiveness of these four antiviral drugs is principally based on studies of patients with uncomplicated influenza (277). Data are limited and inconclusive concerning the effectiveness of amantadine, rimantadine, zanamivir, and oseltamivir for treatment of influenza among persons at high risk for serious complications of influenza (27,235,237,238,240,241,248,250--254). One study assessing oseltamivir treatment primarily among adults reported a reduction in complications necessitating antibiotic therapy compared with placebo (255). Fewer studies of the efficacy of influenza antivirals have been conducted among pediatric populations (235,238,244,245,251, 256,257). One study of oseltamivir treatment documented a decreased incidence of otitis media among children (245). Inadequate data exist regarding the safety and efficacy of any of the influenza antiviral drugs for use among children aged <1 year (234). To reduce the emergence of antiviral drug-resistant viruses, amantadine or rimantadine therapy for persons with influenza A illness should be discontinued as soon as clinically warranted, typically after 3--5 days of treatment or within 24--48 hours after the disappearance of signs and symptoms. The recommended duration of treatment with either zanamivir or oseltamivir is 5 days. Chemoprophylaxis Chemoprophylactic drugs are not a substitute for vaccination, although they are critical adjuncts in preventing and controlling influenza. Both amantadine and rimantadine are indicated for chemoprophylaxis of influenza A infection, but not influenza B. Both drugs are approximately 70%--90% effective in preventing illness from influenza A infection (72,235,251). When used as prophylaxis, these antiviral agents can prevent illness while permitting subclinical infection and development of protective antibody against circulating influenza viruses. Therefore, certain persons who take these drugs will develop protective immune responses to circulating influenza viruses. Amantadine and rimantadine do not interfere with the antibody response to the vaccine (235). Both drugs have been studied extensively among nursing home populations as a component of influenza outbreak-control programs, which can limit the spread of influenza within chronic care institutions (235,250,258--260). Among the neuraminidase inhibitor antivirals, zanamivir and oseltamivir, only oseltamivir has been approved for prophylaxis, but community studies of healthy adults indicate that both drugs are similarly effective in preventing febrile, laboratory-confirmed influenza illness (efficacy: zanamivir, 84%; oseltamivir, 82%) (261,262,278). Both antiviral agents have also been reported to prevent influenza illness among persons administered chemoprophylaxis after a household member was diagnosed with influenza (263,275,278). Experience with prophylactic use of these agents in institutional settings or among patients with chronic medical conditions is limited in comparison with the adamantanes (247,253, 254,264--266). One 6-week study of oseltamivir prophylaxis among nursing home residents reported a 92% reduction in influenza illness (247,279). Use of zanamivir has not been reported to impair the immunologic response to influenza vaccine (246,280). Data are not available regarding the efficacy of any of the four antiviral agents in preventing influenza among severely immunocompromised persons. When determining the timing and duration for administering influenza antiviral medications for prophylaxis, factors related to cost, compliance, and potential side effects should be considered. To be maximally effective as prophylaxis, the drug must be taken each day for the duration of influenza activity in the community. However, to be most cost-effective, one study of amantadine or rimantadine prophylaxis reported that the drugs should be taken only during the period of peak influenza activity in a community (281). Persons at High Risk Who Are Vaccinated After Influenza Activity Has Begun. Persons at high risk for complications of influenza still can be vaccinated after an outbreak of influenza has begun in a community. However, development of antibodies in adults after vaccination takes approximately 2 weeks (222,223). When influenza vaccine is administered while influenza viruses are circulating, chemoprophylaxis should be considered for persons at high risk during the time from vaccination until immunity has developed. Children aged <9 years who receive influenza vaccine for the first time can require 6 weeks of prophylaxis (i.e., prophylaxis for 4 weeks after the first dose of vaccine and an additional 2 weeks of prophylaxis after the second dose). Persons Who Provide Care to Those at High Risk. To reduce the spread of virus to persons at high risk during community or institutional outbreaks, chemoprophylaxis during peak influenza activity can be considered for unvaccinated persons who have frequent contact with persons at high risk. Persons with frequent contact include employees of hospitals, clinics, and chronic-care facilities, household members, visiting nurses, and volunteer workers. If an outbreak is caused by a variant strain of influenza that might not be controlled by the vaccine, chemoprophylaxis should be considered for all such persons, regardless of their vaccination status. Persons Who Have Immune Deficiencies. Chemoprophylaxis can be considered for persons at high risk who are expected to have an inadequate antibody response to influenza vaccine. This category includes persons infected with HIV, chiefly those with advanced HIV disease. No published data are available concerning possible efficacy of chemoprophylaxis among persons with HIV infection or interactions with other drugs used to manage HIV infection. Such patients should be monitored closely if chemoprophylaxis is administered. Other Persons. Chemoprophylaxis throughout the influenza season or during peak influenza activity might be appropriate for persons at high risk who should not be vaccinated. Chemoprophylaxis can also be offered to persons who wish to avoid influenza illness. Health-care providers and patients should make this decision on an individual basis. Control of Influenza Outbreaks in Institutions Using antiviral drugs for treatment and prophylaxis of influenza is a key component of influenza outbreak control in institutions. In addition to antiviral medications, other outbreak-control measures include instituting droplet precautions and establishing cohorts of patients with confirmed or suspected influenza, re-offering influenza vaccinations to unvaccinated staff and patients, restricting staff movement between wards or buildings, and restricting contact between ill staff or visitors and patients (282--284) (for additional information regarding outbreak control in specific settings, see Additional Information Regarding Influenza Infection Control Among Specific Populations). The majority of published reports concerning use of antiviral agents to control influenza outbreaks in institutions are based on studies of influenza A outbreaks among nursing home populations where amantadine or rimantadine were used (235,250,258--260,281). Less information is available concerning use of neuraminidase inhibitors in influenza A or B institutional outbreaks (253,254,266,279,285). When confirmed or suspected outbreaks of influenza occur in institutions that house persons at high risk, chemoprophylaxis should be started as early as possible to reduce the spread of the virus. In these situations, having preapproved orders from physicians or plans to obtain orders for antiviral medications on short notice can substantially expedite administration of antiviral medications. When outbreaks occur in institutions, chemoprophylaxis should be administered to all residents, regardless of whether they received influenza vaccinations during the previous fall, and should continue for a minimum of 2 weeks. If surveillance indicates that new cases continue to occur, chemoprophylaxis should be continued until approximately 1 week after the end of the outbreak. The dosage for each resident should be determined individually. Chemoprophylaxis also can be offered to unvaccinated staff who provide care to persons at high risk. Prophylaxis should be considered for all employees, regardless of their vaccination status, if the outbreak is caused by a variant strain of influenza that is not well-matched by the vaccine. In addition to nursing homes, chemoprophylaxis also can be considered for controlling influenza outbreaks in other closed or semiclosed settings (e.g., dormitories or other settings where persons live in close proximity). For example, chemoprophylaxis with rimantadine has been used successfully to control an influenza A outbreak aboard a large cruise ship (167). To limit the potential transmission of drug-resistant virus during outbreaks in institutions, whether in chronic or acute-care settings or other closed settings, measures should be taken to reduce contact as much as possible between persons taking antiviral drugs for treatment and other persons, including those taking chemoprophylaxis (see Antiviral Drug-Resistant Strains of Influenza). DosageDosage recommendations vary by age group and medical conditions (Table 7). Children Amantadine. Use of amantadine among children aged <1 year has not been adequately evaluated. The FDA-approved dosage for children aged 1--9 years for treatment and prophylaxis is 4.4--8.8 mg/kg body weight/day, not to exceed 150 mg/day. Although further studies are needed to determine the optimal dosage for children aged 1--9 years, physicians should consider prescribing only 5 mg/kg body weight/day (not to exceed 150 mg/day) to reduce the risk for toxicity. The approved dosage for children aged >10 years is 200 mg/day (100 mg twice a day); however, for children weighing <40 kg, prescribing 5 mg/kg body weight/day, regardless of age, is advisable (252). Rimantadine. Rimantadine is approved for prophylaxis among children aged >1 year and for treatment and prophylaxis among adults. Although rimantadine is approved only for prophylaxis of infection among children, certain specialists in the management of influenza consider it appropriate for treatment among children (230). Use of rimantadine among children aged <1 year has not been adequately evaluated. Rimantadine should be administered in 1 or 2 divided doses at a dosage of 5 mg/kg body weight/day, not to exceed 150 mg/day for children aged 1--9 years. The approved dosage for children aged >10 years is 200 mg/day (100 mg twice a day); however, for children weighing <40 kg, prescribing 5 mg/kg body weight/day, regardless of age, is recommended (286). Zanamivir. Zanamivir is approved for treatment among children aged >7 years. The recommended dosage of zanamivir for treatment of influenza is two inhalations (one 5-mg blister per inhalation for a total dose of 10 mg) twice daily (approximately 12 hours apart) (246). Oseltamivir. Oseltamivir is approved for treatment among persons aged >1 year and for chemoprophylaxis among persons aged >13 years. Recommended treatment dosages for children vary by the weight of the child: the dosage recommendation for children who weigh <15 kg is 30 mg twice a day; for children weighing >15--23 kg, the dosage is 45 mg twice a day; for those weighing >23--40 kg, the dosage is 60 mg twice a day; and for children weighing >40 kg, the dosage is 75 mg twice a day. The treatment dosage for persons aged >13 years is 75 mg twice daily. For children aged >13 years, the recommended dose for prophylaxis is 75 mg once a day (247). Persons Aged >65 Years Amantadine. The daily dosage of amantadine for persons aged >65 years should not exceed 100 mg for prophylaxis or treatment, because renal function declines with increasing age. For certain older persons, the dose should be further reduced. Rimantadine. Among older persons, the incidence and severity of central nervous system (CNS) side effects are substantially lower among those taking rimantadine at a dosage of 100 mg/day than among those taking amantadine at dosages adjusted for estimated renal clearance (287). However, chronically ill older persons have had a higher incidence of CNS and gastrointestinal symptoms and serum concentrations 2--4 times higher than among healthy, younger persons when rimantadine has been administered at a dosage of 200 mg/day (235). For prophylaxis among persons aged >65 years, the recommended dosage is 100 mg/day. For treatment of older persons in the community, a reduction in dosage to 100 mg/day should be considered if they experience side effects when taking a dosage of 200 mg/day. For treatment of older nursing home residents, the dosage of rimantadine should be reduced to 100 mg/day (286). Zanamivir and Oseltamivir. No reduction in dosage is recommended on the basis of age alone. Persons with Impaired Renal Function Amantadine. A reduction in dosage is recommended for patients with creatinine clearance <50 mL/min/1.73m2. Guidelines for amantadine dosage on the basis of creatinine clearance are located in the package insert. Because recommended dosages on the basis of creatinine clearance might provide only an approximation of the optimal dose for a given patient, such persons should be observed carefully for adverse reactions. If necessary, further reduction in the dose or discontinuation of the drug might be indicated because of side effects. Hemodialysis contributes minimally to amantadine clearance (288,289). Rimantadine. A reduction in dosage to 100 mg/day is recommended for persons with creatinine clearance <10 mL/min. Because of the potential for accumulation of rimantadine and its metabolites, patients with any degree of renal insufficiency, including older persons, should be monitored for adverse effects, and either the dosage should be reduced or the drug should be discontinued, if necessary. Hemodialysis contributes minimally to drug clearance (290). Zanamivir. Limited data are available regarding the safety and efficacy of zanamivir for patients with impaired renal function. Among patients with renal failure who were administered a single intravenous dose of zanamivir, decreases in renal clearance, increases in half-life, and increased systemic exposure to zanamivir were observed (246,291). However, a limited number of healthy volunteers who were administered high doses of intravenous zanamivir tolerated systemic levels of zanamivir that were substantially higher than those resulting from administration of zanamivir by oral inhalation at the recommended dose (292,293). On the basis of these considerations, the manufacturer recommends no dose adjustment for inhaled zanamivir for a 5-day course of treatment for patients with either mild to moderate or severe impairment in renal function (246). Oseltamivir. Serum concentrations of oseltamivir carboxylate (GS4071), the active metabolite of oseltamivir, increase with declining renal function (247,294). For patients with creatinine clearance of 10--30 mL/min (247), a reduction of the treatment dosage of oseltamivir to 75 mg once daily and in the prophylaxis dosage to 75 mg every other day is recommended. No treatment or prophylaxis dosing recommendations are available for patients undergoing routine renal dialysis treatment. Persons with Liver Disease Amantadine. No increase in adverse reactions to amantadine has been observed among persons with liver disease. Rare instances of reversible elevation of liver enzymes among patients receiving amantadine have been reported, although a specific relation between the drug and such changes has not been established (295). Rimantadine. A reduction in dosage to 100 mg/day is recommended for persons with severe hepatic dysfunction. Zanamivir and Oseltamivir. Neither of these medications has been studied among persons with hepatic dysfunction. Persons with Seizure Disorders Amantadine. An increased incidence of seizures has been reported among patients with a history of seizure disorders who have received amantadine (296). Patients with seizure disorders should be observed closely for possible increased seizure activity when taking amantadine. Rimantadine. Seizures (or seizure-like activity) have been reported among persons with a history of seizures who were not receiving anticonvulsant medication while taking rimantadine (297). The extent to which rimantadine might increase the incidence of seizures among persons with seizure disorders has not been adequately evaluated. Zanamivir and Oseltamivir. Seizure events have been reported during postmarketing use of zanamivir and oseltamivir, although no epidemiologic studies have reported any increased risk for seizures with either zanamivir or oseltamivir use. RouteAmantadine, rimantadine, and oseltamivir are administered orally. Amantadine and rimantadine are available in tablet or syrup form, and oseltamivir is available in capsule or oral suspension form (298,299). Zanamivir is available as a dry powder that is self-administered via oral inhalation by using a plastic device included in the package with the medication. Patients will benefit from instruction and demonstration of correct use of this device (246). PharmacokineticsAmantadine Approximately 90% of amantadine is excreted unchanged in the urine by glomerular filtration and tubular secretion (258,300--303). Thus, renal clearance of amantadine is reduced substantially among persons with renal insufficiency, and dosages might need to be decreased (see Dosage) (Table 7). Rimantadine Approximately 75% of rimantadine is metabolized by the liver (251). The safety and pharmacokinetics of rimantadine among persons with liver disease have been evaluated only after single-dose administration (251,304). In a study of persons with chronic liver disease (the majority with stabilized cirrhosis), no alterations in liver function were observed after a single dose. However, for persons with severe liver dysfunction, the apparent clearance of rimantadine was 50% lower than that reported for persons without liver disease (286). Rimantadine and its metabolites are excreted by the kidneys. The safety and pharmacokinetics of rimantadine among patients with renal insufficiency have been evaluated only after single-dose administration (251,290). Further studies are needed to determine multiple-dose pharmacokinetics and the most appropriate dosages for patients with renal insufficiency. In a single-dose study of patients with anuric renal failure, the apparent clearance of rimantadine was approximately 40% lower, and the elimination half-life was approximately 1.6-fold greater than that among healthy persons of the same age (290). Hemodialysis did not contribute to drug clearance. In studies of persons with less severe renal disease, drug clearance was also reduced, and plasma concentrations were higher than those among control patients without renal disease who were the same weight, age, and sex (286,305). Zanamivir In studies of healthy volunteers, approximately 7%--21% of the orally inhaled zanamivir dose reached the lungs, and 70%--87% was deposited in the oropharynx (246,306). Approximately 4%--17% of the total amount of orally inhaled zanamivir is systemically absorbed. Systemically absorbed zanamivir has a half-life of 2.5--5.1 hours and is excreted unchanged in the urine. Unabsorbed drug is excreted in the feces (246,293). Oseltamivir Approximately 80% of orally administered oseltamivir is absorbed systemically (294). Absorbed oseltamivir is metabolized to oseltamivir carboxylate, the active neuraminidase inhibitor, primarily by hepatic esterases. Oseltamivir carboxylate has a half-life of 6--10 hours and is excreted in the urine by glomerular filtration and tubular secretion via the anionic pathway (247,307). Unmetabolized oseltamivir also is excreted in the urine by glomerular filtration and tubular secretion (308). Side Effects and Adverse ReactionsWhen considering use of influenza antiviral medications (i.e., choice of antiviral drug, dosage, and duration of therapy), clinicians must consider the patient's age, weight, and renal function (Table 7); presence of other medical conditions; indications for use (i.e., prophylaxis or therapy); and the potential for interaction with other medications. Amantadine and Rimantadine Both amantadine and rimantadine can cause CNS and gastrointestinal side effects when administered to young, healthy adults at equivalent dosages of 200 mg/day. However, incidence of CNS side effects (e.g., nervousness, anxiety, insomnia, difficulty concentrating, and lightheadedness) is higher among persons taking amantadine than among those taking rimantadine (308). In a 6-week study of prophylaxis among healthy adults, approximately 6% of participants taking rimantadine at a dosage of 200 mg/day experienced one or more CNS symptoms, compared with approximately 13% of those taking the same dosage of amantadine and 4% of those taking placebo (308). A study of older persons also demonstrated fewer CNS side effects associated with rimantadine compared with amantadine (287). Gastrointestinal side effects (e.g., nausea and anorexia) occur among approximately 1%--3% of persons taking either drug, compared with 1% of persons receiving the placebo (308). Side effects associated with amantadine and rimantadine are usually mild and cease soon after discontinuing the drug. Side effects can diminish or disappear after the first week, despite continued drug ingestion. However, serious side effects have been observed (e.g., marked behavioral changes, delirium, hallucinations, agitation, and seizures) (288,296). These more severe side effects have been associated with high plasma drug concentrations and have been observed most often among persons who have renal insufficiency, seizure disorders, or certain psychiatric disorders and among older persons who have been taking amantadine as prophylaxis at a dosage of 200 mg/day (258). Clinical observations and studies have indicated that lowering the dosage of amantadine among these persons reduces the incidence and severity of such side effects (Table 7). In acute overdosage of amantadine, CNS, renal, respiratory, and cardiac toxicity, including arrhythmias, have been reported (288). Because rimantadine has been marketed for a shorter period than amantadine, its safety among certain patient populations (e.g., chronically ill and older persons) has been evaluated less frequently. Because amantadine has anticholinergic effects and might cause mydriasis, it should not be used among patients with untreated angle closure glaucoma (288). Zanamivir In a study of zanamivir treatment of influenza-like illness among persons with asthma or chronic obstructive pulmonary disease where study medication was administered after use of a B2-agonist, 13% of patients receiving zanamivir and 14% of patients who received placebo (inhaled powdered lactose vehicle) experienced a >20% decline in forced expiratory volume in 1 second (FEV1) after treatment (246,248). However, in a phase-I study of persons with mild or moderate asthma who did not have influenza-like illness, 1 of 13 patients experienced bronchospasm after administration of zanamivir (246). In addition, during postmarketing surveillance, cases of respiratory function deterioration after inhalation of zanamivir have been reported. Certain patients had underlying airways disease (e.g., asthma or chronic obstructive pulmonary disease). Because of the risk for serious adverse events and because the efficacy has not been demonstrated among this population, zanamivir is not recommended for treatment for patients with underlying airway disease (246). If physicians decide to prescribe zanamivir to patients with underlying chronic respiratory disease after carefully considering potential risks and benefits, the drug should be used with caution under conditions of appropriate monitoring and supportive care, including the availability of short-acting bronchodilators (277). Patients with asthma or chronic obstructive pulmonary disease who use zanamivir are advised to 1) have a fast-acting inhaled bronchodilator available when inhaling zanamivir and 2) stop using zanamivir and contact their physician if they experience difficulty breathing (246). No definitive evidence is available regarding the safety or efficacy of zanamivir for persons with underlying respiratory or cardiac disease or for persons with complications of acute influenza (277). Allergic reactions, including oropharyngeal or facial edema, have also been reported during postmarketing surveillance (246,253). In clinical treatment studies of persons with uncomplicated influenza, the frequencies of adverse events were similar for persons receiving inhaled zanamivir and those receiving placebo (i.e., inhaled lactose vehicle alone) (236--241,253). The most common adverse events reported by both groups were diarrhea; nausea; sinusitis; nasal signs and symptoms; bronchitis; cough; headache; dizziness; and ear, nose, and throat infections. Each of these symptoms was reported by <5% of persons in the clinical treatment studies combined (246). Oseltamivir Nausea and vomiting were reported more frequently among adults receiving oseltamivir for treatment (nausea without vomiting, approximately 10%; vomiting, approximately 9%) than among persons receiving placebo (nausea without vomiting, approximately 6%; vomiting, approximately 3%) (242,243,247,309). Among children treated with oseltamivir, 14.3% had vomiting, compared with 8.5% of placebo recipients. Overall, 1% discontinued the drug secondary to this side effect (245), whereas a limited number of adults who were enrolled in clinical treatment trials of oseltamivir discontinued treatment because of these symptoms (247). Similar types and rates of adverse events were reported in studies of oseltamivir prophylaxis (247). Nausea and vomiting might be less severe if oseltamivir is taken with food (247,309). Use During PregnancyNo clinical studies have been conducted regarding the safety or efficacy of amantadine, rimantadine, zanamivir, or oseltamivir for pregnant women; only two cases of amantadine use for severe influenza illness during the third trimester have been reported (134,135). However, both amantadine and rimantadine have been demonstrated in animal studies to be teratogenic and embryotoxic when administered at substantially high doses (286,288). Because of the unknown effects of influenza antiviral drugs on pregnant women and their fetuses, these four drugs should be used during pregnancy only if the potential benefit justifies the potential risk to the embryo or fetus (see manufacturers' package inserts) (246, 247,286,288). Drug InteractionsCareful observation is advised when amantadine is administered concurrently with drugs that affect CNS, including CNS stimulants. Concomitant administration of antihistamines or anticholinergic drugs can increase the incidence of adverse CNS reactions (235). No clinically substantial interactions between rimantadine and other drugs have been identified. Clinical data are limited regarding drug interactions with zanamivir. However, no known drug interactions have been reported, and no clinically critical drug interactions have been predicted on the basis of in vitro data and data from studies using rats (246,310). Limited clinical data are available regarding drug interactions with oseltamivir. Because oseltamivir and oseltamivir carboxylate are excreted in the urine by glomerular filtration and tubular secretion via the anionic pathway, a potential exists for interaction with other agents excreted by this pathway. For example, coadministration of oseltamivir and probenecid resulted in reduced clearance of oseltamivir carboxylate by approximately 50% and a corresponding approximate twofold increase in the plasma levels of oseltamivir carboxylate (247,307). No published data are available concerning the safety or efficacy of using combinations of any of these four influenza antiviral drugs. For more detailed information concerning potential drug interactions for any of these influenza antiviral drugs, package inserts should be consulted. Antiviral Drug-Resistant Strains of InfluenzaAmantadine-resistant viruses are cross-resistant to rimantadine and vice versa (311). Drug-resistant viruses can appear in approximately one third of patients when either amantadine or rimantadine is used for therapy (257,312,313). During the course of amantadine or rimantadine therapy, resistant influenza strains can replace susceptible strains within 2--3 days of starting therapy (312,314). Resistant viruses have been isolated from persons who live at home or in an institution where other residents are taking or have recently taken amantadine or rimantadine as therapy (315,316); however, the frequency with which resistant viruses are transmitted and their effect on efforts to control influenza are unknown. Amantadine- and rimantadine-resistant viruses are not more virulent or transmissible than susceptible viruses (317). The screening of epidemic strains of influenza A has rarely detected amantadine- and rimantadine-resistant viruses (312,318,319). Persons who have influenza A infection and who are treated with either amantadine or rimantadine can shed susceptible viruses early in the course of treatment and later shed drug-resistant viruses, including after 5--7 days of therapy (257). Such persons can benefit from therapy even when resistant viruses emerge. Resistance to zanamivir and oseltamivir can be induced in influenza A and B viruses in vitro (320--327), but induction of resistance requires multiple passages in cell culture. By contrast, resistance to amantadine and rimantadine in vitro can be induced with fewer passages in cell culture (328,329). Development of viral resistance to zanamivir and oseltamivir during treatment has been identified but does not appear to be frequent (247,330--333). In clinical treatment studies using oseltamivir, 1.3% of posttreatment isolates from patients aged >13 years and 8.6% among patients aged 1--12 years had decreased susceptibility to oseltamivir (247). No isolates with reduced susceptibility to zanamivir have been reported from clinical trials, although the number of posttreatment isolates tested is limited (334) and the risk for emergence of zanamivir-resistant isolates cannot be quantified (246). Only one clinical isolate with reduced susceptibility to zanamivir, obtained from an immunocompromised child on prolonged therapy, has been reported (331). Available diagnostic tests are not optimal for detecting clinical resistance to the neuraminidase inhibitor antiviral drugs, and additional tests are being developed (334,335). Postmarketing surveillance for neuraminidase inhibitor-resistant influenza viruses is being conducted (336). Sources of Information Regarding Influenza and Its SurveillanceInformation regarding influenza surveillance, prevention, detection, and control is available at http://www.cdc.gov/flu/weekly/fluactivity.htm. Surveillance information is available through the CDC Voice Information System (influenza update) at 888-232-3228 or CDC Fax Information Service at 888-232-3299. During October--May, surveillance information is updated at least every other week. In addition, periodic updates regarding influenza are published in the MMWR Weekly (http://www.cdc.gov/mmwr). Additional information regarding influenza vaccine can be obtained by calling the CDC Immunization hotline at 800-232-2522 (English) or 800-232-0233 (Spanish). State and local health departments should be consulted concerning availability of influenza vaccine, access to vaccination programs, information related to state or local influenza activity, and for reporting influenza outbreaks and receiving advice concerning outbreak control. Additional Information Regarding Influenza Infection Control Among Specific PopulationsEach year, ACIP provides general, annually updated information regarding control and prevention of influenza. Other reports related to controlling and preventing influenza among specific populations (e.g., immunocompromised persons, health-care personnel, hospitals, and travelers) are also available in the following publications:
* These persons should receive inactivated influenza vaccine. † One dose equals 0.5 mL, divided equally between each nostril. References
Advisory Committee on Immunization Practices
Chairman: Myron J. Levin, M.D., Professor of Pediatrics and Medicine, University of Colorado Health Sciences Center, Denver, Colorado Executive Secretary: Stephen D. Hadler, M.D., Chief, Global Alliance Vaccine Initiative, Centers for Disease Control and Prevention, Atlanta, Georgia Members: Jon S. Abramson, M.D., Wake Forest University School of Medicine, Winston-Salem, North Carolina; Ban Mishu Allos, M.D., Vanderbilt University School of Medicine, Nashville, Tennessee; Guthrie S. Birkhead, M.D., New York State Department of Health, Albany, New York; Judith Campbell, M.D., Baylor College of Medicine, Houston, Texas; Jaime Deseda-Tous, M.D., San Jorge Children's Hospital, San Juan, Puerto Rico; Reginald Finger, M.D., Focus on the Family, Colorado Springs, Colorado; Janet Gildsdorf, M.D., University of Michigan, Ann Arbor, Michigan; Edgar Marcuse, M.D., Children's Hospital and Regional Medical Center, Seattle, Washington; Gregory Poland, M.D., Mayo Clinic College of Medicine, Rochester, Minnesota; John B. Salamone, National Italian American Foundation,Washington, D.C.; John J. Treanor, M.D., University of Rochester School of Medicine and Dentistry, Rochester, New York; Robin Womeodu, M.D., University of Tennessee Health Sciences Center, Memphis, Tennessee; and Richard Zimmerman, M.D., University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania. Ex-Officio Members: James E. Cheek, M.D., Indian Health Service, Albuquerque, New Mexico; Stephen Phillips, D.O., Department of Defense, Falls Church, Virginia; Geoffrey S. Evans, M.D., Health Resources and Services Administration, Rockville, Maryland; Bruce Gellin, M.D., National Vaccine Program Office, Washington, D.C.; Linda Murphy, Centers for Medicare and Medicaid Services, Baltimore, Maryland; George T. Curlin, M.D., National Institutes of Health, Bethesda, Maryland; Karen Midthun, M.D., Food and Drug Administration, Bethesda, Maryland; Kristin Lee Nichol, M.D., Department of Veterans Affairs, Minneapolis, Minnesota. Liaison Representatives: American Academy of Family Physicians, Martin Mahoney, M.D., Ph.D., Clarence, New York, and Richard Clover, M.D., Louisville, Kentucky; American Academy of Pediatrics, Margaret Rennels, M.D., Baltimore, Maryland, and Carol Baker, M.D., Houston, Texas; American Association of Health Plans, Robert Scalettar, M.D., North Haven, Connecticut; American College Health Association, James C. Turner, M.D., Charlottesville, Virginia; American College of Obstetricians and Gynecologists, Stanley Gall, M.D., Louisville, Kentucky; American College of Physicians, Kathleen Neuzil, M.D., Seattle, Washington; American Medical Association, Litjen Tan, Ph.D, Chicago, Illinois; American Pharmacists Association, Stephan L. Foster, Pharm.D., Memphis, Tennessee; Association of Teachers of Preventive Medicine, W. Paul McKinney, M.D., Louisville, Kentucky; Biotechnology Industry Organization, Clement Lewin, Ph.D., Orange, Connecticut; Canadian National Advisory Committee on Immunization, Monica Naus, M.D., Vancouver, British Columbia; Healthcare Infection Control Practices Advisory Committee, Jane D. Siegel, M.D., Dallas, Texas; Infectious Diseases Society of America, Samuel L. Katz, M.D., Durham, North Carolina, and William Schaffner, M.D., Nashville, Tennessee; London Department of Health, David M. Salisbury, M.D., London, United Kingdom; National Association of County and City Health Officials, J. Henry Hershey, M.D., Christiansburg, Virginia; National Coalition for Adult Immunization, David A. Neumann, Ph.D., Bethesda, Maryland; National Immunization Council and Child Health Program, Mexico, Romeo Rodriguez, Mexico City, Mexico; National Medical Association, Dennis A. Brooks, M.D., Baltimore, Maryland; National Vaccine Advisory Committee, Georges Peter, M.D., Providence, Rhode Island; and the Pharmaceutical Research and Manufacturers of America, Damian A. Braga, Swiftwater, Pennsylvania, and Geno Germano, St. Davids, Pennsylvania.
ACIP Influenza Working Group Chair: Richard Zimmerman, M.D., Pittsburgh, Pennsylvania. Members: Jon Abramson, M.D., Winston-Salem, North Carolina; Carolyn Bridges, M.D., Atlanta, Georgia; Robert Chen, M.D., Atlanta, Georgia; Richard Clover, M.D., Louisville, Kentucky; Nancy Cox, Ph.D., Atlanta, Georgia; Keiji Fukuda, M.D., Atlanta, Georgia; Stanley Gall, M.D., Louisville, Kentucky; Randolph Graydon, Baltimore, Maryland; Celine Hanson, M.D., Houston, Texas; Scott Harper, M.D., Atlanta, Georgia; J. Henry Hershey, M.D., Christiansburg, Virginia; John Iskander, M.D., Atlanta, Georgia; Marika Iwane, Ph.D., Atlanta, Georgia; Susan Lett, M.D., Boston, Massachusetts; Roland Levandowski, M.D., Bethesda, Maryland; Alison Mawle, M.D., Atlanta, Georgia; Kathleen Neuzil, M.D., Seattle, Washington; Kristin Lee Nichol, M.D., Minneapolis, Minnesota; Dennis O'Mara, Atlanta, Georgia; Margaret Rennels, M.D., Baltimore, Maryland; William Schaffner, M.D., Nashville, Tennessee; Jane Seward, M.B.B.S., Atlanta, Georgia; and Litjen Tan, Ph.D., Chicago, Illinois. Table 1  Return to top. Table 2 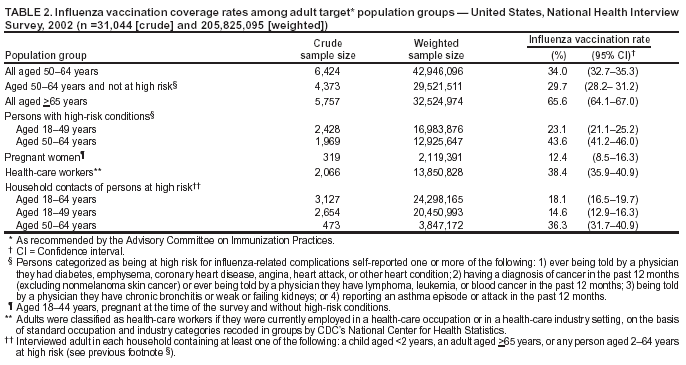 Return to top. Table 3 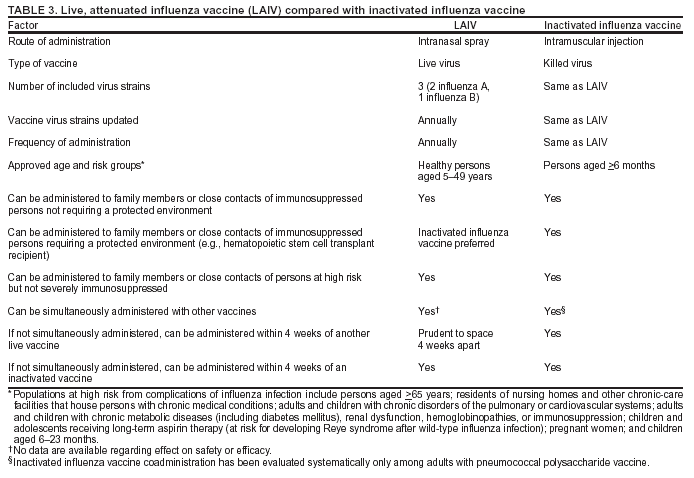 Return to top. Table 4  Return to top. Table 5 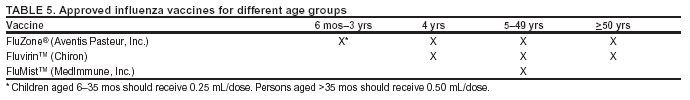 Return to top. Table 6  Return to top. Table 7 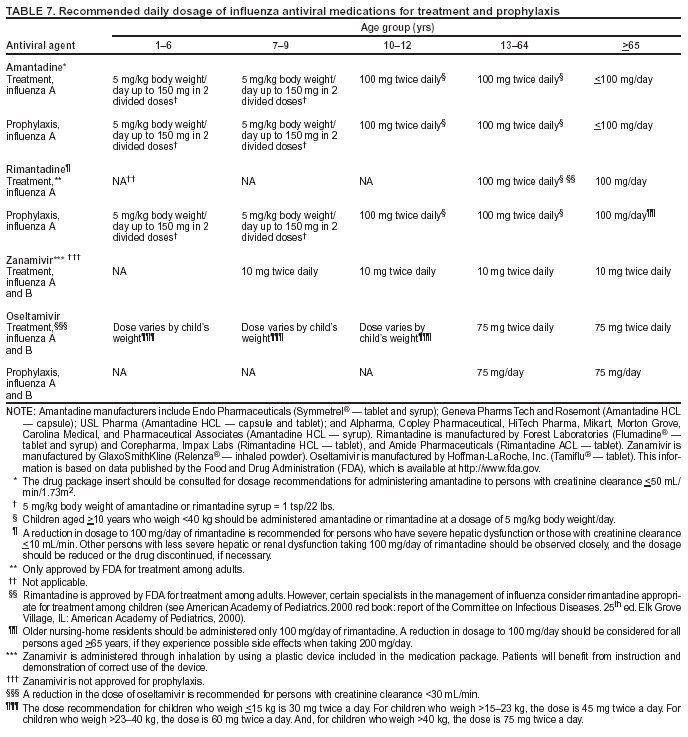 Return to top.
All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 4/30/2004 |
|||||||||
This page last reviewed 4/30/2004
|