 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Recommended Childhood and Adolescent Immunization Schedule --- United States, January--June 2004Each year, CDC's Advisory Committee on Immunization Practices (ACIP) reviews the recommended childhood and adolescent immunization schedule to ensure that it is current with changes in manufacturers' vaccine formulations and reflects revised recommendations for the use of licensed vaccines, including those newly licensed. The recommended childhood and adolescent immunization schedule for January--June 2004 (Figure), recommendations, and format have been approved by ACIP, the American Academy of Family Physicians, and the American Academy of Pediatrics. Catch-Up Childhood and Adolescent Immunization ScheduleA catch-up immunization schedule for children and adolescents who start late or who are >1 month behind was introduced in 2003 (1) and remains the same (Table). Minimum ages and minimum intervals between doses are provided for each of the routinely recommended childhood and adolescent vaccines. The schedule is divided into two age groups: children aged 4 months--6 years and children/adolescents aged 7--18 years. Hepatitis B VaccineThe schedule indicates a change in the recommendation for the minimum age for the last dose in the hepatitis B vaccination schedule. The last dose in the vaccination series should not be administered before age 24 weeks (updating the previous recommendation not to administer the last dose before age 6 months). Adolescent Tetanus and Diphtheria Toxoids (Td) VaccineThe range of recommended ages for the adolescent Td vaccine dose has been updated to emphasize a preference for vaccinating at ages 11--12 years with ages 13--18 years to serve as a catch-up interval. Clarification Regarding Certain Final DosesClarification was added to the footnotes regarding the timing of the final vaccine doses in the series for diphtheria and tetanus toxoids and acellular pertussis (DTaP) vaccine, Haemophilus influenzae type b (Hib) conjugate vaccine, and pneumococcal conjugate vaccine (PCV). The final dose in the DTaP series should be given at age >4 years. The final doses in the Hib and PCV series should be given at age >12 months. Influenza VaccineHealthy children aged 6--23 months are encouraged to receive influenza vaccine when feasible during the 2003--2004 influenza season. Children in this age group are at substantially increased risk for influenza-related hospitalizations (2). ACIP has indicated further that beginning in fall 2004, children aged 6--23 months will be recommended to receive annual influenza vaccine. An updated childhood and adolescent immunization schedule for July--December 2004 will be released to reflect this change. An intranasally administered, live, attenuated influenza vaccine (LAIV) was approved for use in the United States in June 2003. For healthy persons aged 5--49 years, LAIV is an acceptable alternative to the intramuscular trivalent inactivated influenza vaccine (TIV) (3). Vaccine Information StatementsThe National Childhood Vaccine Injury Act requires that all health-care providers give parents or patients copies of Vaccine Information Statements before administering each dose of the vaccines listed in the schedule. Additional information is available from state health departments and at http://www.cdc.gov/nip/publications/vis. Detailed recommendations for using vaccines are available from the manufacturers' package inserts, ACIP statements on specific vaccines, and the 2003 Red Book (4). ACIP statements for each recommended childhood vaccine can be viewed, downloaded, and printed from CDC's National Immunization Program website at http://www.cdc.gov/nip/publications/acip-list.htm; instructions on the use of the Vaccine Information Statements are available at http://www.cdc.gov/nip/publications/vis/vis-instructions.pdf. In addition, guidance on how to obtain and complete a Vaccine Adverse Event Reporting System (VAERS) form is available at http://www.vaers.org or by telephone, 800-822-7967. References
Table 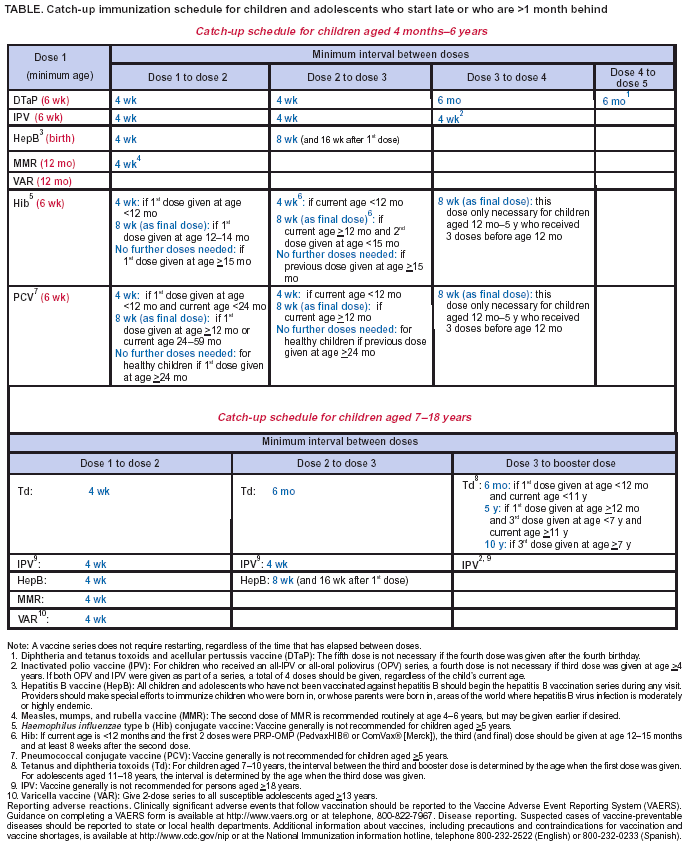 Return to top. Figure 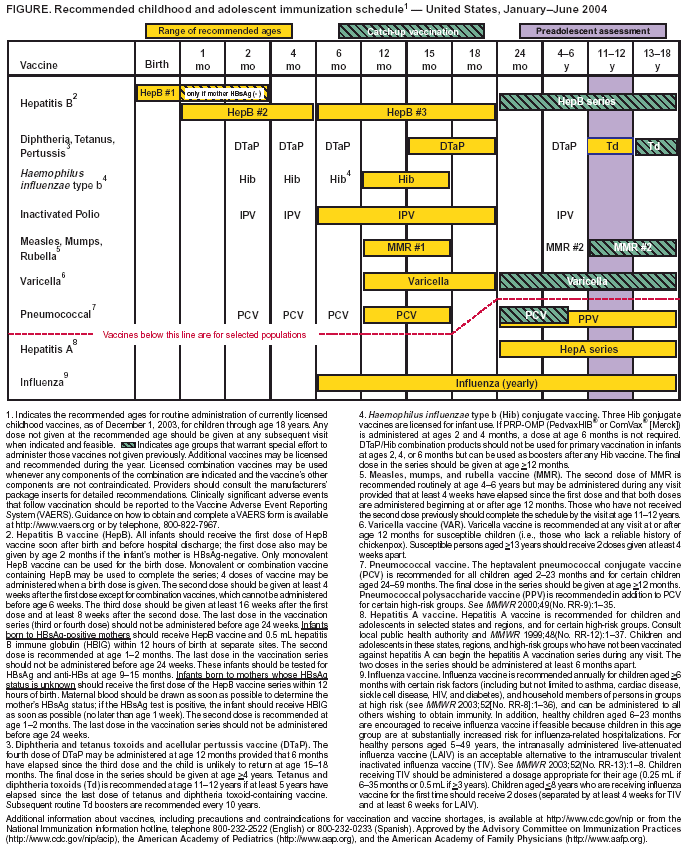 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 1/15/2004 |
|||||||||
This page last reviewed 1/15/2004
|