 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Update: Investigation of Anthrax Associated with Intentional Exposure and Interim Public Health Guidelines, October 2001On October 4, 2001, CDC and state and local public health authorities reported a case of inhalational anthrax in Florida (1). Additional cases of anthrax subsequently have been reported from Florida and New York City. This report updates the findings of these case investigations, which indicate that infections were caused by the intentional release of Bacillus anthracis. This report also includes interim guidelines for postexposure prophylaxis for prevention of inhalational anthrax and other information to assist epidemiologists, clinicians, and laboratorians responding to intentional anthrax exposures. For these investigations, a confirmed case of anthrax was defined as 1) a clinically compatible case of cutaneous, inhalational, or gastrointestinal illness* that is laboratory confirmed by isolation of B. anthracis from an affected tissue or site or 2) other laboratory evidence of B. anthracis infection based on at least two supportive laboratory tests. A suspected case was defined as 1) a clinically compatible case of illness without isolation of B. anthracis and no alternative diagnosis, but with laboratory evidence of B. anthracis by one supportive laboratory test or 2) a clinically compatible case of anthrax epidemiologically linked to a confirmed environmental exposure, but without corroborative laboratory evidence of B. anthracis infection. Laboratory criteria for diagnosis of anthrax consist of 1) isolation and confirmation of B. anthracis from a clinical specimen collected from an affected tissue or site or 2) other supportive laboratory tests, including (a) evidence of B. anthracis DNA by polymerase chain reaction (PCR) from specimens collected from an affected tissue or site, (b) demonstration of B. anthracis in a clinical specimen by immunohistochemical staining, or (c) other laboratory tests (e.g., serology) that may become validated by laboratory confirmation. FloridaOn October 2, the Palm Beach County Health Department (PBCHD) and the Florida Department of Health (FDOH) were notified of a possible anthrax case in Palm Beach County. The suspected case was identified when a gram stain of cerebrospinal fluid (CSF) revealed a gram-positive bacilli. An epidemiologic investigation was initiated by FDOH, PBCHD, and the FDOH state laboratory. The state laboratory and CDC confirmed B. anthracis from a culture of CSF on October 4. Later the same day, FDOH and CDC epidemiologists and laboratory workers arrived in Palm Beach County to assist PBCHD with the investigation. As of October 16, two confirmed cases of inhalational anthrax have been identified. The index patient was a 63-year-old male resident of Palm Beach County who sought medical care at a local hospital on October 2 with fever and altered mental status. Despite antibiotic therapy, his clinical condition deteriorated rapidly, and he died on October 5. An autopsy performed on October 6 confirmed the cause of death as inhalational anthrax. An investigation revealed no obvious exposures to B. anthracis. On October 1, the second patient, a 73-year-old co-worker of the index patient, was admitted to a local hospital for pneumonia. On October 5, a nasal swab was obtained from the patient that yielded a positive culture for B. anthracis. Subsequent testing revealed positive PCR tests for B. anthracis in hemorrhagic pleural fluid and reactive serologic tests. The patient remains hospitalized on antibiotic therapy. Enhanced case finding and retrospective and prospective surveillance systems were initiated in Palm Beach, and surrounding counties. Environmental assessments and sampling were performed at the index patient's home, work site, and travel destinations for the 60 days preceding symptom onset. Environmental sampling revealed B. anthracis contamination of the work site, specifically implicating mail or package delivery. Environmental samples of other locations the patient visited, including extensive sampling of his home, were negative. Questionnaires were administered to employees at the index patient's work site. Postexposure prophylaxis was administered, and nasal swabs were obtained from those with exposure to the work site for >1 hour since August 1. Of 1,075 nasal swabs performed, one was positive for B. anthracis. Environmental and co-worker testing indicated contamination of specific locations at the work site. The investigation and environmental sampling are ongoing. New YorkOn October 9, the New York City Department of Health notified CDC of a person with a skin lesion consistent with cutaneous anthrax. CDC sent a team to New York City to provide epidemiologic and laboratory support to local health officials. As of October 16, two persons with confirmed cases of cutaneous anthrax have been identified. One person with confirmed anthrax was a 38-year-old woman who had handled a suspicious letter postmarked September 18 at her workplace. The letter contained a powder that subsequently was confirmed to contain B. anthracis. On September 25, the patient had a raised lesion on the chest, which over the next 3 days developed surrounding erythema and edema. By September 29, the patient developed malaise and headache. On October 1, a clinician examined the patient and described an approximately 5 cm long oval-shaped lesion with a raised border, small satellite vesicles, and profound edema. The lesion was nonpainful and was associated with left cervical lymphadenopathy. Serous fluid from the lesion was obtained and was negative by gram stain and culture. The patient was prescribed oral ciprofloxacin. Over the next several days, the lesion developed a black eschar, and a biopsy was obtained and sent to CDC for testing. The tissue was positive by immunohistochemical staining for the cell wall antigen of B. anthracis. The other person with confirmed cutaneous anthrax was a 7-month-old infant who visited his mother's workplace on September 28. The next day, the infant had an apparently nontender, massively edematous, weeping skin lesion on his left arm; he was treated with intravenous antibiotics. Over the next several days, the lesion became ulcerative and developed a black eschar; clinicians presumptively attributed the lesion to a spider bite. The infant's clinical course was complicated by hemolytic anemia and thrombocytopenia, requiring intensive care. The diagnosis of cutaneous anthrax was first considered on October 12 after the announcement of the other confirmed anthrax case in New York City. A serum specimen collected on October 2 was positive for B. anthracis by PCR testing at CDC; a skin biopsy obtained on October 13 was positive by immunohistochemical staining at CDC for the cell wall antigen of B. anthracis. No suspicious letter with powder was identified at the mother's workplace. Both patients were treated with ciprofloxacin and are clinically improving. B. anthracis grew from swabs (two nasal and one facial skin swab) from three other persons, suggesting exposure to anthrax. One of the exposures was in a law enforcement officer who brought the letter containing B. anthracis from the index patient's workplace to the receiving laboratory. The other two exposures were in technicians who had processed the letter in the laboratory. Environmental sampling in both workplaces is ongoing and investigations of other exposed persons continue. Reported by: L Bush, MD, Atlantis; J Malecki, MD, Palm Beach County Health Dept, Palm Beach; S Wiersma, MD, State Epidemiologist, Florida Dept of Health. K Cahill, MD, R Fried, MD; M Grossman, MD, Columbia Presbyterian Medical Center; W Borkowsky, MD, New York Univ Medical Center, New York, New York; New York City Dept of Health. National Center for Infectious Diseases; and EIS officers, CDC. Editorial Note:The findings in this report indicate that four confirmed cases of anthrax have resulted from intentional delivery of B. anthracis spores through mailed letters or packages. These are the first confirmed cases of anthrax associated with intentional exposure in the United States and represent a new public health threat. Anthrax is an acute infectious disease caused by the spore-forming bacterium B. anthracis. It occurs most frequently as an epizootic or enzootic disease of herbivores (e.g., cattle, goats, or sheep) that acquire spores from direct contact with contaminated soil. Humans usually become infected through direct contact with B. anthracis spores from infected animals or their products (e.g., goat hair), resulting in cutaneous anthrax (2) (Box 1). Inhalational and gastrointestinal are other forms of the disease in the natural setting (4,5). Human-to-human transmission has not been documented. Clinical laboratorians should be alert to the presence of Bacillus species in patient specimens. In particular, laboratorians should suspect B. anthracis when the specimen is from a previously healthy patient with a rapidly progressive respiratory illness or a cutaneous ulcer. If B. anthracis is suspected, laboratories should immediately notify the health-care provider and local and state public health staff. For rapid identification of B. anthracis, state and local health departments should access the Laboratory Response Network for Bioterrorism (LRN). LRN links state and local public health laboratories with advanced capacity laboratories---including clinical, military, veterinary, agricultural, water, and food-testing laboratories. Laboratorians should contact their state public health laboratory to identify their local LRN representative. Postexposure prophylaxis is indicated to prevent inhalational anthrax after a confirmed or suspected aerosol exposure. When no information is available about the antimicrobial susceptibility of the implicated strain of B. anthracis, initial therapy with ciprofloxacin or doxycycline is recommended for adults and children (Table 1). Use of tetracyclines and fluoroquinolones in children has adverse effects. The risks for these adverse effects must be weighed carefully against the risk for developing life-threatening disease. As soon as penicillin susceptibility of the organism has been confirmed, prophylactic therapy for children should be changed to oral amoxicillin 80 mg/kg of body mass per day divided every 8 hours (not to exceed 500 mg three times daily). B. anthracis is not susceptible to cephalosporins or to trimethoprim/sulfamethoxazole, and these agents should not be used for prophylaxis. CDC is assisting other states and local areas in assessing anthrax exposures. Additional information about anthrax and the public health response is available at <http://www.bt.cdc.gov>. This information was current as of 4 p.m., eastern daylight time, October 17, 2001. References
* Cutaneous illness is characterized by a skin lesion evolving from a papule, through a vesicular stage, to a depressed black eschar; edema, erythema, or necrosis without ulceration may be present. Inhalational illness is characterized by a brief prodrome resembling a "nonspecific febrile" illness that rapidly progresses to a fulminant illness with signs of sepsis and/or respiratory failure, often with radiographic evidence of mediastinal widening; signs of bacterial meningitis may be present. Gastrointestinal illness is characterized by severe abdominal pain usually accompanied by bloody vomiting or diarrhea followed by fever and signs of septicemia. BOX 1. Clinical forms of anthrax Clinical Forms of Anthrax The following clinical descriptions of anthrax are based on experience in adults. The clinical presentation of anthrax in infants is not well defined. Inhalational. Inhalational anthrax begins with a brief prodrome resembling a viral respiratory illness followed by development of hypoxia and dyspnea, with radiographic evidence of mediastinal widening. Inhalational anthrax is the most lethal form of anthrax and results from inspiration of 8,000--50,000 spores of Bacillus anthracis (3). The incubation period of inhalational anthrax among humans typically ranges from 1--7 days but may be possibly up to 60 days. Host factors, dose of exposure, and chemoprophylaxis may affect the duration of the incubation period. Initial symptoms include mild fever, muscle aches, and malaise and may progress to respiratory failure and shock; meningitis frequently develops. Case-fatality estimates for inhalational anthrax are extremely high, even with all possible supportive care including appropriate antibiotics. Cutaneous. Cutaneous anthrax is characterized by a skin lesion evolving from a papule, through a vesicular stage, to a depressed black eschar. The incubation period ranges from 1--12 days. The lesion is usually painless, but patients also may have fever, malaise, headache, and regional lymphadenopathy. The case fatality rate for cutaneous anthrax is 20% without, and <1% with, antibiotic treatment. Gastrointestinal. Gastrointestinal anthrax is characterized by severe abdominal pain followed by fever and signs of septicemia. This form of anthrax usually follows after eating raw or undercooked contaminated meat and can have an incubation period of 1--7 days. An oropharyngeal and an abdominal form of the disease have been described. Involvement of the pharynx is usually characterized by lesions at the base of the tongue, dysphagia, fever, and regional lymphadenopathy. Lower bowel inflammation typically causes nausea, loss of appetite, and fever followed by abdominal pain, hematemesis, and bloody diarrhea. The case-fatality rate is estimated to be 25%--60%. The effect of early antibiotic treatment on the case-fatality rate is not established. Return to top.Table 1 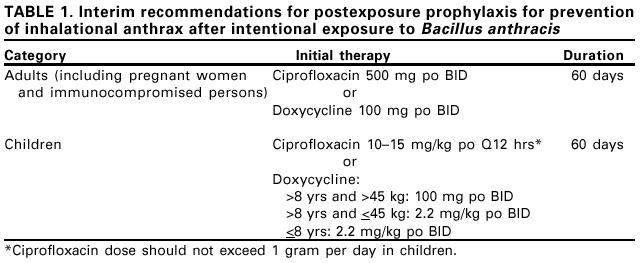 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 10/19/2001 |
|||||||||
This page last reviewed 10/19/2001
|