 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. A Comprehensive Immunization Strategy to Eliminate Transmission of Hepatitis B Virus Infection in the United StatesRecommendations of the Advisory Committee on Immunization Practices (ACIP) Part II: Immunization of AdultsPlease note: An erratum has been published for this article. To view the erratum, please click here. Prepared by
The material in this report originated in the National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention (proposed), Kevin A. Fenton, MD, PhD, Director; the Division of Viral Hepatitis, John W. Ward, MD, Director; the Division of STD Prevention, John M. Douglas, Jr., MD, Director; and the Division of HIV/AIDS Prevention, Robert S. Janssen, MD, Director; the National Center for Immunization and Respiratory Diseases, Anne Schuchat, MD, Director; and the Immunization Services Division, Lance E. Rodewald, MD, Director. Corresponding preparer: Eric E. Mast, MD, Division of Viral Hepatitis, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention (proposed), 1600 Clifton Road, NE, MS G-37, Atlanta, GA 30333. Telephone: 404-718-8500; Fax: 404-718-8595; E-mail: emast@cdc.gov.
SummaryHepatitis B vaccination is the most effective measure to prevent hepatitis B virus (HBV) infection and its consequences, including cirrhosis of the liver, liver cancer, liver failure, and death. In adults, ongoing HBV transmission occurs primarily among unvaccinated persons with behavioral risks for HBV transmission (e.g., heterosexuals with multiple sex partners, injection-drug users [IDUs], and men who have sex with men [MSM]) and among household contacts and sex partners of persons with chronic HBV infection. This report, the second of a two-part statement from the Advisory Committee on Immunization Practices (ACIP), provides updated recommendations to increase hepatitis B vaccination of adults at risk for HBV infection. The first part of the ACIP statement, which provided recommendations for immunization of infants, children, and adolescents, was published previously (CDC. A comprehensive immunization strategy to eliminate transmission of hepatitis B virus infection in the United States: recommendations of the Advisory Committee on Immunization Practices [ACIP]. Part 1: immunization of infants, children, and adolescents. MMWR 2005;54[No. RR-16]:1--33). In settings in which a high proportion of adults have risks for HBV infection (e.g., sexually transmitted disease/human immunodeficiency virus testing and treatment facilities, drug-abuse treatment and prevention settings, health-care settings targeting services to IDUs, health-care settings targeting services to MSM, and correctional facilities), ACIP recommends universal hepatitis B vaccination for all unvaccinated adults. In other primary care and specialty medical settings in which adults at risk for HBV infection receive care, health-care providers should inform all patients about the health benefits of vaccination, including risks for HBV infection and persons for whom vaccination is recommended, and vaccinate adults who report risks for HBV infection and any adults requesting protection from HBV infection. To promote vaccination in all settings, health-care providers should implement standing orders to identify adults recommended for hepatitis B vaccination and administer vaccination as part of routine clinical services, not require acknowledgment of an HBV infection risk factor for adults to receive vaccine, and use available reimbursement mechanisms to remove financial barriers to hepatitis B vaccination. IntroductionHepatitis B is a disease caused by the hepatitis B virus (HBV), which is transmitted through percutaneous (i.e., puncture through the skin) or mucosal (i.e., direct contact with mucous membranes) exposure to infectious blood or body fluids. HBV can cause chronic infection, resulting in cirrhosis of the liver, liver cancer, liver failure, and death. Persons with chronic infection also serve as the main reservoir for continued HBV transmission. Although chronic infection is more likely to develop in persons infected as infants or young children, rates of new infection and acute disease are highest among adults. Hepatitis B vaccination is the most effective measure to prevent HBV infection and its consequences. Since recommendations for hepatitis B vaccination were first issued in 1982, a comprehensive strategy to eliminate HBV transmission in the United States has evolved (1--5). This strategy includes 1) universal vaccination of infants beginning at birth, 2) prevention of perinatal HBV infection through routine screening of all pregnant women for hepatitis B surface antigen (HBsAg) and postexposure immunoprophylaxis of infants born to HBsAg-positive women or to women with unknown HBsAg status, 3) vaccination of all children and adolescents who were not vaccinated previously, and 4) vaccination of previously unvaccinated adults at risk for HBV infection (Box 1). To date, immunization strategies for infants, children, and adolescents have been implemented with considerable success. Recent estimates indicate that approximately 95% of pregnant women are tested for HBsAg and that case management has been effective in ensuring high levels of initiation and completion of postexposure immunoprophylaxis among infants born to HBsAg-positive women (6). Hepatitis B vaccine has been integrated successfully into the childhood vaccination schedule, and infant vaccination coverage levels now are equivalent to those of other vaccines in the childhood schedule (7). Vaccination coverage among adolescents also has increased substantially; preliminary data from 2003 indicated that approximately 50%--60% of adolescents aged 13--15 years have records indicating vaccination (with 3 doses) against hepatitis B (8). During 1990--2005, incidence of acute hepatitis B in the United States declined 78%. The greatest decline (96%) occurred among children and adolescents, coincident with an increase in hepatitis B vaccination coverage. This success can be attributed in part to the established infrastructure for vaccine delivery to children and to federal support for perinatal hepatitis B prevention programs. Among adults, ongoing HBV transmission occurs primarily among unvaccinated adults with risk behaviors for HBV transmission (e.g., heterosexuals with multiple sex partners, injection-drug users [IDUs], and men who have sex with men [MSM]) and among household contacts and sex partners of persons with chronic HBV infection. During 2000--2004, self-reported hepatitis B vaccination coverage among adults at risk for HBV infection increased from 30% to 45% (9); this increase in vaccination coverage likely contributed to the 35% decline in acute hepatitis B incidence that occurred during this period (from 3.7 to 2.4 per 100,000 population). However, incidence of acute hepatitis B remains highest among adults, who accounted for approximately 95% of an estimated 51,000 new HBV infections in 2005. Although acceptance of vaccination is high among adults offered vaccination (10), the low adult vaccination coverage reflects the lack of hepatitis B vaccination services in settings in which a high proportion of adults have risk factors for HBV infection (e.g., sexually transmitted disease [STD]/human immunodeficiency virus [HIV] testing and treatment facilities, drug-abuse treatment and prevention settings, health-care settings targeting services to IDUs, health-care settings targeting services to MSM, and correctional facilities) and missed opportunities to vaccinate adults at risk for HBV infection in primary care and specialty medical settings. Although hepatitis B incidence among adults is expected to continue to decline during the next decade as successive cohorts of persons vaccinated in infancy, childhood, and adolescence reach adulthood, new implementation strategies are needed to protect unvaccinated adults at risk for HBV infection. This report provides updated guidance from the Advisory Committee on Immunization Practices (ACIP) to increase hepatitis B vaccination coverage among adults. It includes recommendations regarding which adults should receive hepatitis B vaccine and outlines implementation strategies to ensure that those adults are vaccinated. The first part of this statement, which provided recommendations for immunization of infants, children, and adolescents, was published previously (11). MethodsIn response to continuing low rates of hepatitis B vaccination among adults at risk for HBV infection, ACIP's Hepatitis Vaccines Work Group met multiple times during October 2004--September 2005 to review previous guidelines and make recommendations for improving vaccination coverage in adults. The work group examined the progress made since 1991 in implementing the U.S. strategy to eliminate HBV transmission (e.g., vaccination coverage data and hepatitis B disease rates), surveillance data on missed opportunities for hepatitis B vaccination among adults with acute hepatitis B, and results of cost-effectiveness analyses. In addition, demonstration projects conducted in settings in which a high proportion of clients were at risk for HBV infection identified the components of successful adult hepatitis B vaccination programs and ongoing challenges to implementing adult hepatitis B vaccination. In January 2005, the proposed recommendations were posted online for public comment. In May 2005, CDC convened a meeting of external consultants, including researchers, physicians, state and local public health professionals, immunization program directors, and directors of viral hepatitis, STD, and HIV/AIDS prevention programs, to obtain input into the draft recommendations and consider the feasibility of the recommended strategies. In October 2005, the revised recommendations were approved by ACIP. Major Updates to the RecommendationsThis report updates ACIP recommendations published previously for hepatitis B vaccination of adults (3). The primary changes from previous recommendations are as follows:
BackgroundClinical Features and Natural History of HBV InfectionHBV is a 42-nm DNA virus classified in the Hepadnaviridae family. The liver is the primary site of HBV replication. After a susceptible person is exposed, the virus enters the liver via the bloodstream; no evidence exists indicating that the virus replicates at mucosal surfaces. HBV infection can produce either asymptomatic or symptomatic infection. The average incubation period is 90 days (range: 60--150 days) from exposure to onset of jaundice and 60 days (range: 40--90 days) from exposure to onset of abnormal serum alanine aminotransferase (ALT) levels (12,13). The onset of acute disease typically is insidious. Infants, children aged <5 years, and immunosuppressed adults with newly acquired HBV infection typically are asymptomatic, whereas 30%--50% of children aged >5 years and adults have initial clinical signs or symptoms (14). When present, clinical symptoms and signs can include anorexia, malaise, nausea, vomiting, abdominal pain, and jaundice. Extrahepatic manifestations of disease (e.g., skin rashes, arthralgias, and arthritis) also can occur (15). The fatality rate among persons with reported cases of acute hepatitis B is 0.5%--1.0%, with the highest rates in adults aged >60 years; however, because a substantial number of infections are asymptomatic and therefore are not reported, the overall fatality rate among all persons with HBV infection likely is lower (16). Approximately 95% of primary infections in adults with normal immune status are self-limited, with elimination of virus from blood and subsequent lasting immunity to reinfection. Chronic infection occurs in <5% of infected persons aged >5 years, approximately 30% of infected children aged <5 years, and approximately 90% of infected infants, with continuing viral replication in the liver and persistent viremia (14,17--19). Primary infections become chronic more frequently in immunosuppressed persons (e.g., hemodialysis patients and persons with HIV infection) (19,20) and persons with diabetes (21). Overall, approximately 25% of persons who become chronically infected during childhood and 15% of those who become chronically infected after childhood die prematurely from cirrhosis or liver cancer; the majority remain asymptomatic until onset of cirrhosis or end-stage liver disease (22). No specific treatment exists for acute hepatitis B; supportive care is the mainstay of therapy. Persons who have chronic HBV infection require medical evaluation and regular monitoring (23--25). Therapeutic agents approved by the Food and Drug Administration (FDA) for treatment of chronic hepatitis B can achieve sustained suppression of HBV replication and remission of liver disease in certain persons (24). Periodic screening with ultrasonography and alfa-fetoprotein has been demonstrated to enhance early detection of hepatocellular carcinoma (HCC) (25). Certain chronically infected persons with HCC have experienced long-term survival after resection of small hepatocellular carcinomas, and persons who were screened had HCC detected at an earlier stage and had a substantial survival advantage compared with historical controls (25); however, data from controlled studies are lacking. Guidance for the diagnosis and management of hepatitis B is available (26). Interpretation of Serologic Markers of HBV InfectionAntigens and antibodies associated with HBV infection include HBsAg and antibody to HBsAg (anti-HBs), hepatitis B core antigen (HBcAg) and antibody to HBcAg (anti-HBc), and hepatitis B e antigen (HBeAg) and antibody to HBeAg (anti-HBe). At least one serologic marker is present during each of the different phases of HBV infection (13,27). The serologic markers typically used to differentiate between acute, resolving, and chronic infection are HBsAg, anti-HBc, and anti-HBs (Table 1). HBeAg and anti-HBe screening typically is used for the management of patients with chronic infection. Serologic assays are available commercially for all markers except HBcAg because no free HBcAg circulates in blood. The presence of a confirmed HBsAg-positive result in serum indicates active HBV infection. All HBsAg-positive persons should be considered infectious. In newly infected persons, HBsAg is the only serologic marker detected during the first 3--5 weeks after infection. The average time from exposure to detection of HBsAg is 30 days (range: 6--60 days) (12,13). Highly sensitive single-sample nucleic acid tests can detect HBV DNA in the serum of an infected person 10--20 days before detection of HBsAg (28). Transient HBsAg positivity has been reported for up to 18 days after hepatitis B vaccination and is clinically insignificant (29,30). Anti-HBc appears at the onset of symptoms or liver-test abnormalities in acute HBV infection and persists for life. Acute or recently acquired infection can be distinguished by the presence of the immunoglobulin M (IgM) class of anti-HBc, which is detected at the onset of acute hepatitis B and persists for up to 6 months if the disease resolves. In patients who have chronic HBV infection, IgM anti-HBc can persist during viral replication at low levels that typically are not detectable by assays used in the United States. However, persons with exacerbations of chronic infection can test positive for IgM anti-HBc (31). Using IgM anti-HBc testing for diagnosis of acute hepatitis B should be limited to persons for whom clinical evidence of acute hepatitis or an epidemiologic link to a case has been identified because the positive predictive value of this test is low in asymptomatic persons. In persons who recover from HBV infection, HBsAg is eliminated from the blood, and anti-HBs develops, typically within 3--4 months. The presence of anti-HBs typically indicates immunity from HBV infection. Infection or immunization with one serotype of HBV confers immunity to all serotypes. In addition, anti-HBs can be detected for several months after hepatitis B immune globulin (HBIG) administration. Persons who recover from natural infection typically will be positive for both anti-HBs and anti-HBc, whereas persons who respond to hepatitis B vaccine have only anti-HBs. In persons who become chronically infected, HBsAg and anti-HBc persist, typically for life. HBsAg will become undetectable in approximately 0.5%--2% of persons with chronic infection yearly; anti-HBs will occur in the majority of these persons (32--35). In certain persons, the only HBV serologic marker detected in serum is anti-HBc. Isolated anti-HBc can be detected after HBV infection in persons who have recovered but whose anti-HBs levels have waned. Certain chronically infected persons with anti-HBc alone have circulating HBsAg not detectable by commercial serology. HBV DNA has been detected in the blood of <10% of persons with isolated anti-HBc (36,37). These persons are unlikely to be infectious except under circumstances in which they are a source for direct percutaneous exposure of susceptible recipients to substantial quantities of virus (e.g., through blood transfusion or organ transplantation) (38). An isolated anti-HBc result also can be a false-positive. Typically, the frequency of isolated anti-HBc relates directly to the prevalence of HBV infection in the population. In populations with a high prevalence of HBV infection, isolated anti-HBc likely indicates previous infection, with loss of anti-HBs. For persons in populations with a low prevalence of HBV infection, isolated anti-HBc is found in approximately 10%--20% of persons with serologic markers of HBV infection (37) and often represents a false-positive reaction; the majority of these persons have a primary anti-HBs response after a 3-dose series of hepatitis B vaccine (39,40). HBeAg can be detected in the serum of persons with acute or chronic HBV infection. The presence of HBeAg correlates with high levels of viral replication (i.e., HBV DNA levels typically of 107--109 IU/mL, indicating high infectivity) (41,42). Loss of HBeAg correlates with low levels (i.e., HBV DNA levels of <105 IU/mL) of replicating virus, although certain HBeAg-negative persons have HBV DNA levels up to 108--109 IU/mL (43). A mutation in the precore region of the HBV genome has been identified in HBeAg-negative persons with high HBV DNA levels (44,45). Epidemiology of HBV InfectionHBV is transmitted by percutaneous or mucosal exposure to infectious blood or body fluids. Although HBsAg has been detected in multiple body fluids, only serum, semen, and saliva have been demonstrated to be infectious (46,47). HBV is concentrated most highly in serum, with lower concentrations in semen and saliva. All HBsAg-positive persons are infectious, but those who are also HBeAg positive are more infectious because their blood contains high titers of HBV (typically HBV DNA levels of 107--109 IU/mL) (41,42). HBV is comparatively stable in the environment and remains viable for >7 days on environmental surfaces at room temperature (48). HBV DNA at concentrations of 102--103 IU/mL can be present on environmental surfaces in the absence of any visible blood and still cause transmission (48,49). For adults, the two primary sources of HBV infection are sexual contact and percutaneous exposure to blood. Person-to-person transmission of HBV also can occur in settings involving nonsexual interpersonal contact over an extended period (e.g., among household contacts of a person with chronic HBV infection and developmentally disabled persons living in a long-term--care facility). HBV is transmitted efficiently by sexual contact among heterosexuals and among MSM. Risk factors associated with sexual transmission among heterosexuals include having unprotected sex with an infected partner, having unprotected sex with more than one partner, and history of another STD. Risk factors associated with sexual transmission among MSM include having multiple sex partners, history of another STD, and anal intercourse. Percutaneous transmission of HBV can occur from receipt of blood transfusion or organ or tissue transplant from an infectious donor; injection-drug use, including sharing of injection-preparation equipment; and frequent exposure to blood or needles among health-care workers. In the United States, donor selection procedures and routine testing of donors have made transmission of HBV via transfusion of whole blood and blood components a rare occurrence (50,51). Persons with hemophilia who received plasma-derived clotting factor concentrates were previously at high risk for HBV infection, but such transmission has been eliminated through viral inactivation procedures and use of recombinant clotting factor concentrates. Among persons with bleeding disorders treated at U.S. hemophilia treatment centers during 1998--2002, no infections with viral hepatitis, including HBV, were attributable to blood products received during that time (52). Outbreaks of HBV infection from exposure to contaminated equipment used for therapeutic injections and other health-care--related procedures, tattooing, and acupuncture also have been reported, although such exposures among patients with acute hepatitis B are reported rarely (53--57). In the majority of cases, transmission resulted from noncompliance with aseptic techniques for administering injections and recommended infection-control practices designed to prevent cross-contamination of medical equipment and devices. No infections have been demonstrated in susceptible persons who had oral mucous membrane exposure to HBsAg-positive saliva, but transmission has occurred through a human bite and has been demonstrated in animals by subcutaneous inoculation of saliva (46,58--60). Persons living with chronically infected persons are at risk for HBV infection through percutaneous or mucosal exposures to blood or infectious body fluids (e.g., sharing a toothbrush or razor, contact with exudates from dermatologic lesions, or contact with HBsAg-contaminated surfaces). Persons with chronic HBV infection also can transmit HBV in other settings (e.g., schools, child care centers, or facilities for developmentally disabled persons), especially if they behave aggressively or have medical problems (e.g., exudative dermatitis or open skin lesions) that increase the risk for exposure to blood or serous secretions. Adults at Risk for HBV Infection In the United States in 2005, the highest incidence of acute hepatitis B was among adults aged 25--45 years (Figure 1). Approximately 79% of newly acquired cases of hepatitis B are associated with high-risk sexual activity or injection-drug use; other known exposures (i.e., occupational, household, travel, and health-care--related) together account for 5% of new cases, and 16% deny a specific risk factor for infection (61; CDC, unpublished data, 2001--2005). Adults at risk for infection by sexual exposure. The most common source of HBV infection among adults in the United States is sexual contact. Heterosexual transmission accounts for approximately 39% of new HBV infections among adults, and transmission among MSM accounts for approximately 24% (CDC, unpublishe data, 2001--2005). Serologic evidence of HBV infection (i.e., anti-HBc positive) has ranged from 10% to 40% among adults seeking treatment in STD clinics (62--64) and from 10% to 25% among MSM aged <30 years (65; CDC, unpublished data, 1999--2000). Follow-up studies identified HBV infection in 20%--42% of susceptible heterosexual partners of persons with acute hepatitis B (66--68). Among susceptible heterosexual spouses of persons with chronic HBV infection, the seroprevalence of HBV infection ranged from 25% to 59% (69--71). Injection-drug users. IDUs account for approximately 16% of new HBV infections in the United States (CDC, unpublished data, 2001--2005). Incidence of HBV infection among unvaccinated IDUs is high, ranging from 10 to 31 per 100 person-years (72--74). Risk for HBV transmission increases with the number of years of drug use and is associated with frequency of injection and with sharing of drug-preparation equipment (e.g., cottons, cookers, and rinse water), independent of syringe sharing (73,75). In a study of the seroprevalence of HBV infection among IDUs admitted to drug treatment in six U.S. cities, 64% (range: 50%--81%) had serologic evidence of HBV infection, and seroprevalence increased with age (76). Studies of street-recruited IDUs (77,78) and female IDUs (79) have identified similar prevalence of HBV infection, whereas a lower prevalence (25%) was found in a study of young IDUs (aged 18--30 years) (74). Chronic HBV infection has been identified in 3.1% of IDUs in a detention setting (77) and 7.1% of IDUs with HIV coinfection (80). Household contacts of persons with chronic HBV infection. Seroprevalence of HBV infection among susceptible household contacts of persons with chronic infection has varied, ranging from 14% to 60% (69,71,81--85). The risk for infection is highest among sex partners of, and children living with, a person with chronic HBV infection in a household or extended family setting (83--85). Developmentally disabled persons in long-term--care facilities. Developmentally disabled persons in residential and nonresidential facilities historically have had high rates of HBV infection (86,87), but the prevalence of infection has declined substantially since the implementation of routine hepatitis B vaccination in these settings (88,89). Nonetheless, because HBsAg-positive persons reside in such facilities, clients and staff continue to be at risk for infection. Persons at risk for occupational exposure to HBV. Before hepatitis B vaccination was widely implemented, HBV infection was recognized as a common occupational hazard among persons who were exposed to blood while caring for patients or working in laboratories (90,91). Since then, routine hepatitis B vaccination of health-care workers and use of standard precautions to prevent exposure to bloodborne pathogens have made HBV infection a rare event in these populations (92--94). Since the mid-1990s, the incidence of HBV infection among health-care workers has been lower than that among the general population (94). Public safety workers with exposures to blood also might be at risk for HBV infection (95--97); however, the prevalence of HBV infection in occupational groups such as police officers, firefighters, and corrections officers generally does not differ from that in the general population when adjusted for race and age (97), and infection is associated most often with nonoccupational risk factors (97,98). No increased risk for occupationally acquired HBV infection has been documented in workers exposed infrequently to blood or body fluids (e.g., ward clerks, dietary workers, maintenance workers, housekeeping personnel, teachers, and persons employed in day care settings) (91). Hemodialysis patients. Since the initiation of strict infection-control practices and hepatitis B vaccination, the rate of HBV infection among patients undergoing hemodialysis has declined approximately 95% (99,100). Nonetheless, repeated outbreaks of HBV infection among unvaccinated patients underscore the continued risk for infection in this population (101). Persons with chronic liver disease. Persons with chronic liver disease are not at increased risk for HBV infection unless they have percutaneous or mucosal exposure to infectious blood or body fluids. Furthermore, studies of the outcomes of acute hepatitis B among patients with chronic liver disease provide little evidence that acute hepatitis B increases their risk for an acute liver failure. However, concurrent chronic HBV infection might increase the risk for progressive chronic liver disease in HCV-infected patients (102). Travelers to HBV-endemic regions. Short-term travelers to regions in which HBV infection is of high or intermediate endemicity (Box 2) typically are at risk for infection only through exposure to blood in medical, health-care, or disaster-relief activities; receipt of medical care that involves parenteral exposures; or sexual activity or drug use (103). Infection rates of 2%--5% per year among persons working in such regions for >6 months have been reported (104,105). HIV-positive persons. Published data on the overall prevalence of HBV and HIV coinfection in the United States are limited. Studies of certain subgroups have identified prevalence of previous or current HBV infection of 45% in HIV-infected MSM aged 22--29 years (CDC, unpublished data, 1998--2000), 24% in adolescent HIV-infected males (106), and 43% in HIV-infected women, including 76% among HIV-infected female IDUs (79). Chronic HBV infection has been identified in 6%--14% of HIV-positive persons from Western Europe and the United States, including 9%--17% of MSM, 7%--10% of IDUs, and 4%--6% of heterosexuals (107). The course of HBV infection can be modified in the presence of HIV, with a lower incidence of jaundice and a higher incidence of chronic HBV infection (20,108,109). Limited data also indicate that HIV-infected patients with chronic HBV infection have an increased risk for liver-related mortality and morbidity (110). Incidence of Acute Hepatitis B During 1990--2005, the overall incidence of reported acute hepatitis B declined 78%, from 8.5 to 1.9 per 100,000 population (Figure 2), and the estimated number of new HBV infections, after adjusting for underreporting and asymptomatic infections, declined from approximately 232,000 to approximately 51,000 infections (CDC, unpublished data, 1990--2005). Among children and adolescents aged <19 years, incidence declined 96%, from 2.4 to 0.1 per 100,000 population. Among adults aged >19 years, incidence declined 76%, from 9.9 to 2.4 per 100,000 population, and racial/ethnic disparities in incidence were nearly eliminated for Asians/Pacific Islanders, American Indians/Alaska Natives, and Hispanics (Figure 3). Incidence also declined substantially among blacks aged >19 years during this period, from 19.7 to 4.2 per 100,000 population; however, in 2005, incidence among blacks remained nearly three times higher than that among other racial/ethnic populations. After leveling during 1999--2002, acute hepatitis B incidence among adults decreased 35% during 2002--2005, from 3.7 to 2.4 per 100,000 population (CDC, unpublished data, 2006). In 2005, the highest incidence of acute hepatitis B occurred among persons aged 25--44 years. Prevalence of HBV Infection During 1988--1994, the overall age-adjusted prevalence of HBV infection (including previous or chronic infection) in the U.S. population was 4.9%, and the prevalence of chronic infection was 0.4% (111). Persons who have immigrated to the United States from countries in which HBV is endemic (Box 2, Figure 4) are affected disproportionately by chronic HBV infection; in particular, the majority of chronic HBV infections in the United States are among Asians/Pacific Islanders (112--114). The prevalence of chronic HBV infection among persons immigrating to the United States from Central and Southeast Asia, the Middle East, and Africa varies (range: 5%--15%) and reflects the patterns of HBV infection in the countries and regions of origin. During 1994--2003, approximately 40,000 immigrants with chronic HBV infection were admitted annually to the United States for permanent residence (115; CDC, unpublished data, 2005). Prophylaxis Against HBV InfectionHepatitis B VaccineHepatitis B vaccine is available as a single-antigen formulation and also in fixed combination with other vaccines. Two single-antigen vaccines are available in the United States: Recombivax HB® (Merck & Co., Inc., Whitehouse Station, New Jersey) and Engerix-B® (GlaxoSmithKline Biologicals, Rixensart, Belgium). Of the three licensed combination vaccines, one (Twinrix® [GlaxoSmithKline Biologicals, Rixensart, Belgium]) is used for vaccination of adults and two (Comvax® [Merck & Co., Inc., Whitehouse Station, New Jersey] and Pediarix® [GlaxoSmithKline Biologicals, Rixensart, Belgium]) are used for vaccination of infants and young children. Twinrix contains recombinant HBsAg and inactivated hepatitis A virus. Comvax contains recombinant HBsAg and Haemophilus influenzae type b (Hib) polyribosylribitol phosphate conjugated to Neisseria meningitidis outer membrane protein complex. Pediarix contains recombinant HBsAg, diphtheria and tetanus toxoids and acellular pertussis adsorbed (DTaP), and inactivated poliovirus (IPV). HBsAg is the antigen used for hepatitis B vaccination (116,117). Vaccine antigen can be purified from the plasma of persons with chronic HBV infection or produced by recombinant DNA technology. For vaccines available in the United States, recombinant DNA technology is used to express HBsAg in yeast, which then is purified from the cells by biochemical and biophysical separation techniques (118,119). Hepatitis B vaccines licensed in the United States are formulated to contain 10--40 µg of HBsAg protein/mL. Hepatitis B vaccines produced for distribution in the United States do not contain thimerosal as a preservative or contain only a trace amount (<1.0 µg mercury/mL) from the manufacturing process (120,121). Hepatitis B Immune GlobulinHBIG provides passively acquired anti-HBs and temporary protection (i.e., 3--6 months) when administered in standard doses. HBIG typically is used as an adjunct to hepatitis B vaccine for postexposure immunoprophylaxis to prevent HBV infection. For nonresponders to hepatitis B vaccination, HBIG administered alone is the primary means of protection after an HBV exposure. HBIG is prepared from the plasma of donors with high concentrations of anti-HBs. The plasma is screened to eliminate donors who are positive for HBsAg, antibodies to HIV and hepatitis C virus (HCV), and HCV RNA. In addition, proper manufacturing techniques for HBIG inactivate viruses (e.g., HBV, HCV, and HIV) from the final product (122,123). No evidence exists to indicate that HBV, HCV, or HIV ever has been transmitted by HBIG commercially available in the United States. HBIG that is commercially available in the United States does not contain thimerosal. Adult Vaccination Schedules and Results of VaccinationPreexposure VaccinationVaccination of Adults Primary vaccination consists of >3 intramuscular doses of hepatitis B vaccine (Table 2). The 3-dose vaccine series administered intramuscularly at 0, 1, and 6 months produces a protective antibody response in approximately 30%--55% of healthy adults aged <40 years after the first dose, 75% after the second dose, and >90% after the third dose (124,125). After age 40 years, the proportion of persons who have a protective antibody response after a 3-dose vaccination regimen declines below 90%, and by age 60 years, protective levels of antibody develop in only 75% of vaccinated persons (126). In addition to age, other host factors (e.g., smoking, obesity, genetic factors, and immune suppression) contribute to decreased vaccine response (127--130). Alternative vaccination schedules (e.g., 0, 1, and 4 months or 0, 2, and 4 months) have been demonstrated to elicit dose-specific and final rates of seroprotection similar to those obtained on a 0-, 1-, 6-month schedule (131). The combined hepatitis A--hepatitis B vaccine (Twinrix) is indicated for vaccination of persons aged >18 years with risk factors for both hepatitis A and hepatitis B. The dosage of the hepatitis A component in the combined vaccine is lower than that in the single-antigen hepatitis A vaccine, allowing it to be administered in a 3-dose schedule instead of the 2-dose schedule used for the single-antigen vaccine. Nonstandard Vaccine Schedules No apparent effect on immunogenicity has been documented when minimum spacing of doses (i.e., 4 weeks between doses 1 and 2, 8 weeks between doses 2 and 3, and 16 weeks between doses 1 and 3) is not achieved precisely. Increasing the interval between the first 2 doses has little effect on immunogenicity or final antibody concentration (132--134). The third dose confers the maximum level of seroprotection but acts primarily as a booster and appears to provide optimal long-term protection (135). Longer intervals between the last 2 doses result in higher final antibody levels but might increase the risk for acquisition of HBV infection among persons who have a delayed response to vaccination. No differences in immunogenicity are observed when vaccines from different manufacturers are used to complete the vaccine series. Response to Revaccination Although serologic testing for immunity is not necessary after routine vaccination of adults, postvaccination testing is recommended for persons whose subsequent clinical management depends on knowledge of their immune status, including certain health-care and public safety workers; chronic hemodialysis patients, HIV-infected persons, and other immunocompromised persons; and sex or needle-sharing partners of HBsAg-positive persons (Appendix A). Of persons who did not respond to a primary 3-dose vaccine series with anti-HBs concentrations of >10 mIU/mL, 25%--50% responded to an additional vaccine dose, and 44%--100% responded to a 3-dose revaccination series (136--141). Better response to revaccination occurs in persons who have measurable but low (<10 mIU/mL) levels of antibody after the initial series (136,137). Increased vaccine doses (e.g., double the standard dose) were demonstrated to enhance revaccination response rates in one study (140) but not in another (138). Intradermal vaccination has been reported to be immunogenic in persons who did not respond to intramuscular vaccination (142,143); however, intradermal vaccination is not a route of administration indicated in the manufacturers' package labeling. Persons who do not have protective levels of anti-HBs 1--2 months after revaccination either are primary nonresponders or are infected with HBV. Genetic factors might contribute to nonresponse to hepatitis B vaccination (130,137). Groups Requiring Different Vaccination Doses or Schedules Compared with immunocompetent adults, hemodialysis patients are less likely to have protective levels of antibody after vaccination with standard vaccine dosages; protective levels of antibody developed in 67%--86% (median: 64%) of adult hemodialysis patients who received 3--4 doses of either vaccine in various dosages and schedules (100). Higher seroprotection rates have been identified in patients with chronic renal failure, particularly those with mild or moderate renal failure, who were vaccinated before becoming dialysis dependent. After vaccination with a 4-dose series, the seroprotection rate among adult predialysis patients with serum creatinine levels of <4.0 mg/dL was 86%, compared with 37% among patients with serum creatinine levels of >4.0 mg/dL, 88% of whom were dialysis patients (144). Humoral response to hepatitis B vaccination also is reduced in other immunocompromised persons (e.g., HIV-infected persons, hematopoietic stem-cell transplant recipients, and patients undergoing chemotherapy) (145--147). Modified dosing regimens, including doubling the standard antigen dose or administering additional doses, might increase response rates (148--150). However, limited data regarding response to these alternative vaccination schedules are available. Immune Memory Anti-HBs is the only easily measurable correlate of vaccine-induced protection. Immunocompetent persons who achieve anti-HBs concentrations of >10 mIU/mL after preexposure vaccination have nearly complete protection against both acute disease and chronic infection, even if anti-HBs concentrations decline subsequently to <10 mIU/mL (151--154). Although immunogenicity is lower among immunocompromised persons, those who achieve and maintain a protective antibody response before exposure to HBV have a high level of protection from infection (155,156). After primary immunization with hepatitis B vaccine, anti-HBs levels decline rapidly within the first year and more slowly thereafter. Among young adults who respond to a primary vaccine series with antibody concentrations of >10 mIU/mL, 17%--50% have low or undetectable concentrations of anti-HBs (reflecting anti-HBs loss) 10--15 years after vaccination (155--157). In the absence of exposure to HBV, the persistence of detectable anti-HBs after vaccination depends on the concentration of postvaccination antibodies (158). Even when anti-HBs concentrations decline to <10 mIU/mL, nearly all vaccinated persons remain protected against HBV infection. The mechanism for continued vaccine-induced protection is thought to be the preservation of immune memory through selective expansion and differentiation of clones of antigen-specific B and T lymphocytes (159). Persistence of vaccine-induced immune memory among persons who responded to a primary adult vaccine series 4--23 years previously but then had anti-HBs concentrations of <10 mIU/mL has been demonstrated by an anamnestic increase in anti-HBs concentrations in 74%--100% of these persons 2--4 weeks after administration of an additional vaccine dose and by antigen-specific B and T cell proliferation (160). Although direct measurement of immune memory is not yet possible, these data indicate that a high proportion of vaccinees retain immune memory and would have an anti-HBs response upon exposure to HBV. Population-based studies of highly vaccinated populations have demonstrated elimination of new HBV infections for up to 2 decades after hepatitis B immunization programs were initiated (161--163). Breakthrough infections (detected by the presence of anti-HBc or HBV DNA) have been documented in a limited percentage of vaccinated persons (159,164), but these infections typically are transient and asymptomatic; breakthrough infections resulting in chronic HBV infection have been documented only rarely among infants born to HBsAg-positive mothers (165) and have not been observed among immunocompetent adults. Limited data are available on the duration of immune memory after hepatitis B vaccination in immunocompromised persons (e.g., HIV-infected patients, dialysis patients, patients undergoing chemotherapy, or hematopoietic stem-cell transplant patients). No clinically significant HBV infections have been documented among immunocompromised persons who maintain protective levels of anti-HBs. In studies of long-term protection among HIV-infected persons, breakthrough infections occurring after a decline in anti-HBs concentrations to <10 mIU/mL have been transient and asymptomatic (155). However, among hemodialysis patients who responded to the vaccine, clinically significant HBV infection has been documented in persons who have not maintained anti-HBs concentrations of >10 mIU/mL (166). Postexposure ProphylaxisBoth passive-active postexposure prophylaxis (PEP) using HBIG and hepatitis B vaccine and active PEP using hepatitis B vaccine alone are highly effective in preventing infection after exposure to HBV (167--170). HBIG alone has also been demonstrated to be effective in preventing HBV transmission (68,171--173), but with the availability of hepatitis B vaccine, HBIG typically is used as an adjunct to vaccination. Guidelines for PEP for adults with occupational (174) and nonoccupational exposures (Appendix B) to HBV have been developed. The major determinant of the effectiveness of PEP is early administration of the initial dose of vaccine. The effectiveness of PEP diminishes the longer after exposure it is initiated (27,175,176). Studies are limited on the maximum interval after exposure during which PEP is effective, but the interval is likely <7 days for needlestick (171,172,177) exposures and <14 days for sexual exposures (68,154,168,170,173). Substantial evidence suggests that adults who respond to hepatitis B vaccination are protected from chronic HBV infection for at least 20 years even if vaccinees lack detectable anti-HBs at the time of an exposure (151--153). For this reason, immunocompetent persons who have had postvaccination testing and are known to have responded to hepatitis B vaccination with anti-HBs concentrations of >10 mIU/mL do not require additional passive or active immunization after an HBV exposure and do not need further periodic testing to assess anti-HBs concentrations. Vaccine SafetyHepatitis B vaccines have been demonstrated to be safe when administered to infants, children, adolescents, and adults (178). Since 1982, an estimated 70 million adolescents and adults and 50 million infants and children in the United States have received >1 dose of hepatitis B vaccine (CDC, unpublished data, 2004). Vaccine ReactogenicityThe most frequently reported side effects in persons receiving hepatitis B vaccine are pain at the injection site (3%--29%) and temperature of >99.9°F (>37.7°C) (1%--6%) (124,125). However, in placebo-controlled studies, these side effects were reported no more frequently among persons receiving hepatitis B vaccine than among persons receiving placebo (179). Adverse EventsCDC and FDA continually assess the safety of hepatitis B vaccine and other vaccines through ongoing monitoring of data from the Vaccine Safety Datalink (VSD) project, the Vaccine Adverse Events Reporting System (VAERS), and other surveillance systems. A causal association has been established between receipt of hepatitis B vaccine and anaphylaxis (178). On the basis of VSD data, the estimated incidence of anaphylaxis among children and adolescents who received hepatitis B vaccine is one case per 1.1 million vaccine doses distributed (95% confidence interval = 0.1--3.9) (180). Early postlicensure surveillance of adverse events suggested a possible association between Guillain-Barré syndrome (GBS) and receipt of the first dose of plasma-derived hepatitis B vaccine among U.S. adults (181). However, in a subsequent analysis of GBS cases reported to CDC, FDA, and vaccine manufacturers, among an estimated 2.5 million adults who received >1 dose of recombinant hepatitis B vaccine during 1986--1990, the rate of GBS that occurred after hepatitis B vaccination did not exceed the background rate among unvaccinated persons (CDC, unpublished data, 1992). An Institute of Medicine review concluded that evidence was insufficient to reject or accept a causal association between GBS and hepatitis B vaccination (178,182,183). One retrospective case-control study (184,185) reported an association between hepatitis B vaccine and multiple sclerosis (MS) among adults. However, multiple studies (186--189) have demonstrated no such association. Reviews by scientific panels have favored rejection of a causal association between hepatitis B vaccination and MS (190,191). In rare instances, chronic illnesses have been reported after hepatitis B vaccination, including chronic fatigue syndrome (192), neurologic disorders (e.g., leukoencephalitis, optic neuritis, and transverse myelitis) (193--195), rheumatoid arthritis (196,197), type 1 diabetes (198), and autoimmune disease (199). However, no evidence of a causal association between these conditions or other chronic illnesses and hepatitis B vaccine has been demonstrated (183,190,200--203). Reported episodes of alopecia (hair loss) after rechallenge with hepatitis B vaccine suggest that vaccination might, in rare cases, trigger episodes of alopecia (204). However, a population-based study determined no statistically significant association between alopecia and hepatitis B vaccine (205). Contraindications and PrecautionsHepatitis B vaccination is contraindicated for persons with a history of hypersensitivity to yeast or any vaccine component (206--209). Despite a theoretic risk for allergic reaction to vaccination in persons with allergy to Saccharomyces cerevisiae (baker's yeast), no evidence exists to document adverse reactions after vaccination of persons with a history of yeast allergy. Persons with a history of serious adverse events (e.g., anaphylaxis) after receipt of hepatitis B vaccine should not receive additional doses. As with other vaccines, vaccination of persons with moderate or severe acute illness, with or without fever, should be deferred until illness resolves (210). Vaccination is not contraindicated in persons with a history of MS, GBS, autoimmune disease (e.g., systemic lupus erythematosis or rheumatoid arthritis), or other chronic diseases. Pregnancy is not a contraindication to vaccination. Limited data suggest that developing fetuses are not at risk for adverse events when hepatitis B vaccine is administered to pregnant women (211). Available vaccines contain noninfectious HBsAg and should cause no risk of infection to the fetus. Implementation Barriers and Rationale for New RecommendationsSoon after hepatitis B vaccine was licensed in 1982, ACIP recommended vaccination for adults at increased risk for HBV infection (212). However, the recommendations were not widely implemented, and coverage among adults at risk for HBV infection remained low. By the early 1990s, the difficulty in vaccinating adults at risk for HBV infection and the substantial burden of HBV-related disease resulting from infections acquired during childhood indicated that additional hepatitis B vaccination strategies were needed (213,214). In 1991, recommendations for vaccination of unvaccinated adults at high risk for HBV infection became part of the national strategy adopted by ACIP and professional medical organizations to eliminate HBV transmission in the United States (3). However, hepatitis B vaccine still is not offered routinely in medical settings serving adults, and a substantial number of adults at risk for HBV infection remain unvaccinated. Multiple factors contribute to low hepatitis B vaccination coverage among adults at risk. In contrast to vaccination of children, no national program exists to support vaccine purchase and infrastructure for vaccine delivery to uninsured and underinsured adults. Reimbursement mechanisms for vaccination of adults with health insurance also are not widely used. In addition, certain patients and health-care providers are reluctant to discuss risk behaviors (215), and providers might not make hepatitis B vaccination a priority compared with other clinical care services. One strategy demonstrated to be effective at increasing vaccination coverage among adults at risk for HBV infection is to offer vaccination to all adults as part of routine prevention services in settings in which a high proportion of adults have HBV risk factors (10,216--223). In STD and HIV treatment facilities, health-care settings serving IDUs, and health-care settings targeting services to MSM, nearly all patients have behavioral risk factors for HBV infection. Furthermore, a high proportion of persons receiving health care in HIV testing facilities or correctional facilities report sexual and drug-use risk behaviors (224,225). Therefore, providing hepatitis B vaccination in these settings offers an efficient and effective way to reach adults at highest risk. During 2001--2004, in a study of 760 adults with reported acute hepatitis B who participated in CDC's Sentinel Counties Study of Viral Hepatitis, 39% reported a history of STD treatment, 40% reported a history of incarceration, and 22% reported a history of drug treatment; overall, 61% would have had at least one opportunity to be vaccinated either during STD or drug treatment or at a correctional facility (226). Demonstration projects that supported the purchase of hepatitis B vaccine and its administration in settings in which a high proportion of adults have HBV risk factors have established the feasibility of providing the vaccine as part of comprehensive STD, HIV, and hepatitis prevention services (10). When clients were offered hepatitis B vaccination in such settings, first-dose acceptance rates of 70%--85% were achieved (10,216,223,227). These demonstration projects have identified the components of successful adult hepatitis B vaccination programs (Box 3). In addition, "one-stop" delivery of integrated prevention services was preferred by the majority of patients and typically resulted in enhanced delivery of all prevention services (227). Return visits for second and third doses of hepatitis B vaccine also provide opportunities for patients to receive other STD/HIV-related services (e.g., test results, additional counseling, and referral). Multiple studies have established the cost-effectiveness of providing hepatitis B vaccination at STD/HIV counseling and testing sites, correctional institutions, drug-abuse treatment centers, and other settings serving adults at risk for HBV infection (228--231). Universal vaccination of adults in settings in which a high proportion of persons have HBV risk factors will reach a substantial proportion of all adults at risk for HBV infection. However, not all adults with risk factors for HBV infection visit these settings. For example, an estimated 80%--95% of STDs are diagnosed in settings other than STD clinics (232,233). Therefore, primary care and clinical preventive service providers (e.g., physicians' offices, community health centers, family planning clinics, liver disease clinics, and travel clinics) also should provide hepatitis B vaccine whenever indicated or requested as part of regular preventive care. Limited data are available regarding best practices in primary care and specialty medical settings to achieve high vaccination coverage among adults at risk for HBV infection. In one project in which hepatitis B vaccine was made available free of charge to primary care clients in community clinics, low vaccination coverage rates were observed, compared with rates at other venues (10). This finding suggests that provision of free vaccine alone might not ensure increased use of hepatitis B vaccine and that other implementation strategies (e.g., education and training of clinicians and standing orders) are needed to prompt providers to offer vaccination to adults. In primary care settings, targeting vaccination to persons at risk is an efficient approach to preventing HBV infection. During 2001--2005, among persons with acute hepatitis B who participated in CDC's Sentinel Counties Study of Viral Hepatitis, 84% reported risk behaviors or characteristics, either during the incubation period (i.e., 6 weeks--6 months) or during their lifetimes, that placed them in a group for which hepatitis B vaccination was recommended (CDC, unpublished data, 2001--2005). Providers in primary care settings can ascertain patients' risks for HBV infection and identify candidates for hepatitis B vaccination during routine patient visits. Assessment of patients' sex- and drug-related risk factors is recommended by the U.S. Preventive Services Task Force and the American Medical Association (AMA) (234,235) and has the ancillary benefit of identifying candidates for other prevention interventions (e.g., screening for HIV infection and other STDs and drug-abuse treatment). However, risk-targeted approaches can miss persons in need of prevention services. Patients might be reluctant to report sex- and drug-related behaviors, particularly when these behaviors are not perceived as relevant to the clinical encounter. In addition, despite recommendations of the U.S. Preventive Services Task Force and AMA, providers might be reluctant to inquire about behavioral risk factors. For example, surveys of physicians and patients conducted during 1995--1999 indicated that fewer than half of patients were asked about sexual behaviors (236--238). Health-care providers should educate all patients about the health benefits of hepatitis B vaccination, including risk factors for HBV infection and the importance of vaccination for persons who engage in certain risk behaviors. This information might stimulate patients to request vaccination from their primary care providers, without requiring them to acknowledge a specific risk factor. Another possible strategy is to offer vaccination to all adults in age groups with the highest incidence of infection as part of routine medical care (Figure 1). An age-based approach might simplify vaccination-related decision-making by practitioners and remove the stigma associated with disclosure of risk behaviors. Other adult vaccines, including those for influenza and pneumococcal disease, are delivered on age-based schedules. However, the effectiveness of age-based strategies in increasing hepatitis B vaccination coverage among adults at risk is unknown. In addition, age-based strategies for adult vaccination would be substantially more costly than risk-targeted approaches (CDC, unpublished data, 2005). Lack of funding for vaccine and its administration is a major barrier to provision of hepatitis B vaccine to adults. Hepatitis B vaccine often is a reimbursable charge in health-care settings that bill insurance or Medicaid for services, and surveys of public and private insurers indicate high rates of coverage for hepatitis B vaccination (239,240). In one study, an estimated 74% of adults aged 18--49 years with risk factors for HBV infection had health insurance coverage (241). However, public clinics might not have systems in place to bill for vaccination services, and reimbursement of private providers might be inadequate to cover the purchase and administration of vaccine. Other adults either lack private insurance or are not eligible for reimbursement by Medicaid. Although the Vaccines for Children program provides federally funded vaccine and administration costs for vaccination of uninsured and underinsured children and youth aged <19 years, no similar program supports adult vaccination. Certain adult hepatitis B vaccination programs have been successful at identifying federal, state, or local funds to provide free or low-cost hepatitis B vaccination to uninsured or underinsured adults. For example, the Immunization Grant Program, created under Section 317 of the Public Health Service Act, provides funding to state, local, and territorial public health agencies for vaccine purchase and vaccination-program operation (242). Section 317 funds can be used to purchase childhood and adult vaccines, including adult hepatitis B vaccine. However, lack of adequate funding constrains efforts to increase vaccination coverage among adults. To maximize available resources for hepatitis B vaccination, public and private health-care providers should become familiar with insurance billing and reimbursement mechanisms that can be used for hepatitis B vaccine. AMA billing and reimbursement guidelines are available at http://www.ama-assn.org/ama1/pub/upload/mm/36/ama_hep_coding_trifo.pdf. Although HBV infections are expected to decline as a result of universal childhood immunization and increased vaccination of adults at risk, an estimated 1.25 million persons in the United States are living with chronic HBV infection and require essential prevention services and medical management. In particular, Asians/Pacific Islanders in the United States have a high prevalence of chronic HBV infection, representing a major health disparity. Persons with chronic HBV infection often are unaware of their infection status or do not receive needed care. Few programs have been implemented to identify HBsAg-positive persons, provide or refer these persons for appropriate medical management, and provide vaccination to their contacts (243). During delivery of hepatitis B vaccination and provision of other preventive services, health-care providers have opportunities to identify persons with chronic HBV infection, refer them for counseling and management, and ensure that their susceptible contacts receive vaccination. Guidelines to identify and manage HBsAg-positive persons have been developed (Appendix C). Implementation of the recommendations and strategies outlined in this report and the companion ACIP recommendations for infants, children, and adolescents (11) should lead ultimately to the elimination of HBV transmission in the United States. New information will have implications for this effort, and adjustments and changes are expected to occur. Recommendations and Implementation Strategies for Hepatitis B Vaccination of AdultsRecommendations
Implementation Strategies
Acknowledgments The following persons contributed to the development of this report: Guthrie S. Birkhead, MD, AIDS Institute, New York State Department of Health, Albany, New York; Anna S. F. Lok, MD, Division of Gastroenterology, University of Michigan, Ann Arbor, Michigan; Molli C. Conti, Hepatitis B Foundation, Doylestown, Pennsylvania; Josiah D. Rich, MD, Brown University, Providence, Rhode Island; Robert A. Gunn, MD, San Diego County Health and Human Services Agency, San Diego, California; Harold S. Margolis, MD, Joanna Buffington, MD, Alison Greenspan, MPH, Stephanie M. Neitzel, Kevin P. O'Connor, MA, Annemarie Wasley, PhD, Brigette F. Ulin, MPH, Ian T. Williams, PhD, Division of Viral Hepatitis, Matthew T. McKenna, MD, Division of HIV/AIDS Prevention, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention (proposed); William L. Atkinson, MD, Edward W. Brink, MD, Immunization Services Division, Susan A. Maloney, MD, Division of Global Migration and Quarantine, National Center for Immunization and Respiratory Diseases (proposed), CDC. References
Table 1 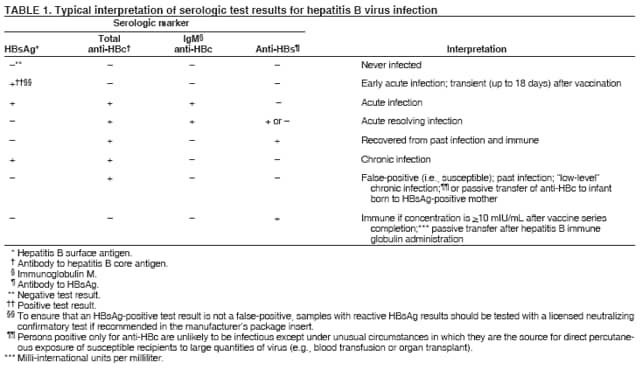 Return to top. Figure 1 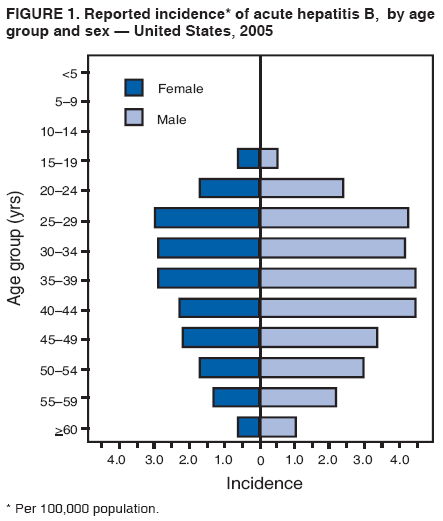 Return to top. Box 1  Return to top. Table 2 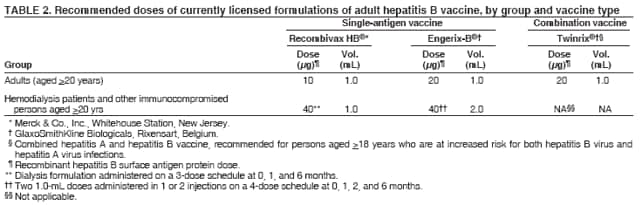 Return to top. Figure 2 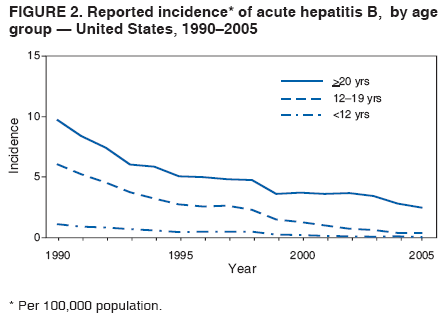 Return to top. Box 2  Return to top. Table 3  Return to top. Figure 3 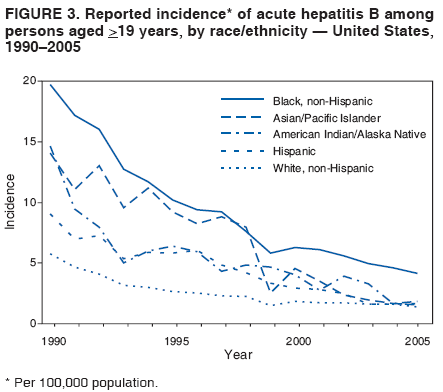 Return to top. Box 3  Return to top. Figure 4 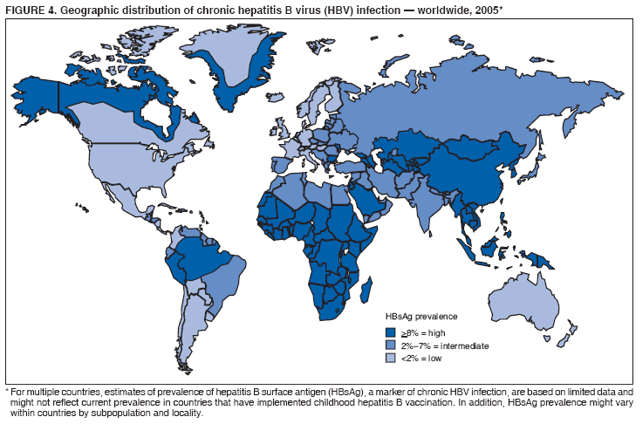 Return to top. Box 4  Return to top. Box 5  Return to top. Box 6  Return to top. Box 7  Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 11/21/2006 |
|||||||||
|