 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Hepatitis B Vaccination of Inmates in Correctional Facilities ---Texas, 2000--2002In December 2002, approximately 2.2 million persons were incarcerated in the United States (1); an estimated 8 million were released to the community that year (2). In 2001, approximately 22,000 acute hepatitis B cases and 78,000 new hepatitis B virus (HBV) infections occurred in the United States (3); an estimated 29% of these cases were in persons who had been incarcerated previously (4). The majority of HBV infections among incarcerated persons are acquired in the community; however, infection also is transmitted within correctional settings (2). Hepatitis B vaccination of incarcerated persons is recommended to prevent transmission in correctional facilities and in previously incarcerated persons on their return to the community (2). In May 2000, the Texas Department of Criminal Justice (TDCJ), which oversees custody of state jail and prison inmates, implemented a hepatitis B vaccination program. To determine hepatitis B vaccination rates of inmates during 2000--2002, TDCJ reviewed charts of inmates released during a 3-day period for documentation of vaccination. This report summarizes the results of that study, which indicated that rates of vaccine acceptance and vaccine series completion among inmates were high. Establishing hepatitis B vaccination programs in prisons and jails can prevent a substantial proportion of HBV infections among adults in the outside community. During 2000--2002, TDCJ housed approximately 151,000 inmates in 105 adult facilities, including prisons (median sentence of inmates: 9 years; range: 2--99 years) and jails (median sentence of inmates: 1.3 years; range: 3 months--2 years). Approximately 40,000 new offenders enter these facilities annually, and an estimated 1% of inmates are transferred between facilities daily (5,6). In 1999, state funds were appropriated for hepatitis B vaccination of all inmates in jails and prisons. Before implementation of the vaccination program, a cost-effectiveness model was developed that estimated the cost effectiveness of prevaccination testing for immunity to HBV infection among inmates. Stored serum specimens from 889 inmates incarcerated during 1998--1999 were tested for antibodies to hepatitis B core antigen (anti-HBc); HBV prevalence was 18%. The model estimated that at a threshold prevalence of 25%, the cost of a program with prevaccination testing was equivalent to that of vaccination without testing; at lower prevalence, prevaccination testing would not be cost effective (Figure). On the basis of these findings, all of the estimated 40,000 entering inmates were offered vaccine without prevaccination testing. Entering inmates were offered the first hepatitis B vaccine dose at the time of admission. Persons who were already incarcerated were offered the first dose at the time of their annual health evaluation, which occurred on their anniversary month of incarceration. After vaccination of incarcerated persons, only newly admitted inmates were offered vaccine. Vaccine was administered on a 0-, 2-, and 4-month schedule. An electronic pharmacy auto-renewal system was used to send second and third vaccine doses to the appropriate facility for each inmate. Health-care workers also recorded vaccine dose administration in each inmate's medical record, enabling inmates to complete the vaccination series despite frequent transfers within the system. In February 2002, TDCJ evaluated vaccine acceptance and series completion rates. Charts of 232 prison inmates and 211 jail inmates released during a 3-day period were audited for receipt of hepatitis B vaccine; 426 (96%) inmates with no record of previous vaccination or HBV infection were considered to be eligible for vaccination. Lack of documentation of a vaccination encounter was interpreted as a failure to offer vaccine, and only a signed informed refusal form was counted as a vaccination refusal. Hepatitis B vaccine was offered to 319 (75%) of 426 inmates. Prison inmates were more likely to be offered vaccine (185/220 [84%]) than jail inmates (134/206 [65%]) (p<0.001), which might be related to higher inmate turnover and lack of staff contact time in jails (Table). However, acceptance of the first vaccine dose was higher among jail inmates (114/134 [85%]) than among prison inmates (134/185 [72%]) (p = 0.005). Among 125 prison and 99 jail inmates who began vaccination and were incarcerated for >4 months, the 3-dose completion rate was 96% and 54%, respectively. In December 2002, the hepatitis B vaccination program was suspended because of a lack of funds. Reported by: M Kelley, MD, L Linthicum, MD, Texas Dept of Criminal Justice. A Spaulding, MD, K Billah, PhD, C Weinbaum, MD, Div of Viral Hepatitis, National Center for Infectious Diseases; R Small, Div of STD Prevention, National Center for HIV, STD, and TB Prevention, CDC. Editorial Note:Evaluation of the TDCJ hepatitis B vaccination program demonstrated that high vaccine coverage could be achieved for inmates in a state correctional system. Incarceration provides an opportunity to vaccinate persons at high risk typically not served by prevention services in the public or private sectors, and vaccination of incarcerated populations is cost effective (7). The findings in this report illustrate the need to tailor a program to a particular facility. Completion of the vaccine series is a more feasible goal for long-term facilities; short-term facilities should initiate the vaccine series, supply an immunization record and, where feasible, provide information at discharge about facilities offering the remaining vaccine doses. Vaccination also can be completed if the person returns to a correctional institution. Prevaccination testing to detect existing immunity can eliminate the cost of revaccinating persons who were vaccinated previously or infected. TDCJ's decision not to perform prevaccination testing was based on a model that included the costs of testing and vaccination and the series completion rate. The model assumed that all inmates who received the first vaccine dose would return for subsequent doses; if attrition caused by release was included in the model, prevaccination testing would only be cost effective if the prevalence of immunity was higher. Changes in prevalence of immunity to HBV infection or costs (e.g., vaccine, labor, and testing) also would change the cost effectiveness of prevaccination testing. In particular, immunity to HBV infection in young adults is changing rapidly within most communities because of an increase in vaccinated adolescents. If adequate immunization records are not available for inmates, periodic monitoring of the prevalence of immunity to HBV infection using a serologic marker to detect both infection (i.e., anti-HBc) and immunization (i.e., antibodies to hepatitis B surface antigen) will help corrections officials determine when prevaccination testing might reduce costs (2). The findings in this report are subject to at least two limitations. First, inmates with shorter sentences are more likely to be discharged and might be overrepresented by the sampling. Because inmates with short sentences might not have been incarcerated long enough to complete the vaccination series, more inmates might have completed the vaccination series than this study demonstrated. Second, lack of long-term follow-up precludes evaluation of the eventual series completion by jail inmates, who might have accessed additional doses outside the correctional system or during subsequent incarcerations. Hepatitis B vaccination of inmates in state correctional facilities is feasible if resources are available to purchase and administer vaccine. In 2000, a survey of state correctional facility medical directors indicated that the majority of prison systems would vaccinate inmates if resources were available (8). Although hepatitis B vaccination of inmates has been recommended since the vaccine first became available in 1982 (9), only five states (Hawaii, Michigan, New Mexico, Vermont, and Wisconsin) vaccinate inmates routinely (D. Burnett, M.D., Wisconsin Department of Corrections and F. Pullara, M.D., New Mexico Department of Corrections, personal communications, 2004) (8). Collaborations between public health and corrections authorities at the state and local level are essential to overcome barriers to vaccination program implementation. References
Table 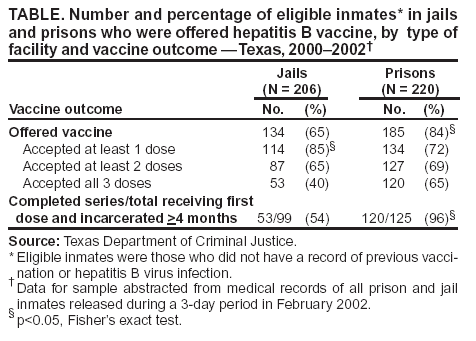 Return to top. Figure 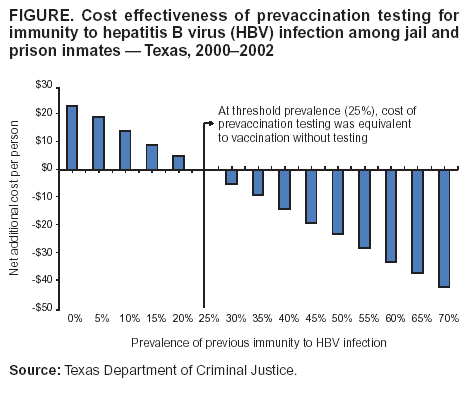 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 8/5/2004 |
|||||||||
This page last reviewed 8/5/2004
|