 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Progress Toward Poliomyelitis Eradication --- Afghanistan, 1999--2000In 1988, the World Health Assembly of the World Health Organization (WHO) resolved to eradicate poliomyelitis globally by 2000. During the same year, the Eastern Mediterranean Region* (EMR) of WHO passed a resolution to join the global initiative. Since then, substantial progress has been made worldwide and in EMR member countries (1,2). Afghanistan, with ongoing civil conflict, initiated polio eradication activities in 1994. Since then, a countrywide surveillance system for acute flaccid paralysis (AFP) was established and National Immunization Days (NIDs)† were implemented (3). This report summarizes the achievements toward polio eradication in Afghanistan during 1999--2000. Routine VaccinationIn 1996, an estimated 30% of infants aged <1 year had received three doses of oral poliovirus vaccine (OPV) (3). In 1998, a review of the Expanded Program on Immunization (EPI) documented wide variations in vaccination coverage by geographic area; levels were particularly low in the north as a result of civil conflict. In 1999, EPI acceleration campaigns provided vaccinations to 82,000 unvaccinated children aged <2 years. In 2000, a comprehensive 5-year plan was drafted to set targets and strategies for the coming years. Supplemental OPV VaccinationDuring 1994--1996, supplemental vaccination activities against polio began with multivaccine subnational campaigns that delivered diphtheria and tetanus toxoids and pertussis vaccine, OPV, and measles vaccine to children aged <5 years. NIDs using OPV were initiated during April--May 1997, and since have been conducted annually. High coverage was achieved during four NID rounds in 1999 and another four in 2000 (Table 1). Of 330 districts in Afghanistan, 325 were reached during the fall 1999 NIDs. During the spring 2000 NIDs, all districts were reached except two north of the capital (Kabul) where most of the population had left the area because of ongoing civil conflict. Supplemental vaccination activities in Afghanistan have been coordinated with neighboring countries, particularly Iran and Pakistan. Because surveillance data indicate that Afghanistan and Pakistan are one epidemiologic block, supplemental campaigns have been conducted simultaneously in both countries when possible. Since the fall of 1999, careful district level NID planning and well-supervised house-to-house vaccination have led to incremental improvements in the quality and coverage of each NID. AFP SurveillanceIn 1997, 37 AFP sentinel reporting sites were established. Since then, surveillance has expanded to 234 sites with emphasis on areas with high population density. In 2000, Afghanistan exceeded the WHO established target for a nonpolio AFP rate indicative of sensitive surveillance (i.e., >1.0 per 100,000 population aged <15 years) with a rate of 1.2 (Table 1). During 1999--2000, the number of AFP cases increased from 230 to 253, and the number of wild polioviruses isolated from AFP cases decreased from 63 to 28 (Figure 1). The National Institute of Health (NIH), Islamabad, Pakistan, has provided laboratory support for the Afghanistan program. All stool specimens are flown from Afghanistan to Islamabad on United Nations' flights and transported to the NIH laboratory. A remaining challenge is the timely collection of adequate stool specimens§ from AFP case-patients. In 2000, 50% of AFP cases reported nationally had adequate stool specimens, which was substantially short of the WHO target of 80%. This low level is partly the result of AFP being identified late in patients' illness, which precludes the collection of stool specimens soon after paralysis onset. Intensified efforts are being made to improve surveillance quality by the immediate investigation of all AFP cases and weekly active surveillance visits to major hospitals and shrines. Reported by: Afghanistan Country Office, World Health Organization, Islamabad, Pakistan. Eastern Mediterranean Regional Office, World Health Organization, Cairo, Egypt. Vaccines and Biologicals Dept, World Health Organization, Geneva, Switzerland. Respiratory and Enteric Viruses Br, Div of Viral and Rickettsial Diseases, National Center for Infectious Diseases; Vaccine Preventable Disease Eradication Div, National Immunization Program, CDC. Editorial Note:Although polio remains endemic in Afghanistan, progress during 1999--2000 demonstrates that key strategies can be implemented successfully in countries experiencing internal strife. During 1999--2000, the nonpolio AFP rate almost doubled and the number of districts reached by NIDs increased steadily. Careful planning and supervision of house-to-house vaccination and support from an increasing number of local partners resulted in the largest number of children ever being reached. Monitoring by nongovernment organizations, United Nations' agencies, and local authorities has increased the quality of NIDs. During the spring 2000, the days of tranquility were respected by all warring factions and their local commanders, greatly facilitating the implementation of NIDs. Civil conflict, massive population shifts (returning refugees and traditional nomadic movements), a drought, rebuilding the public health infrastructure, geographic barriers, extreme climate, and the need to access areas that can be reached only by several days' travel on muleback are some of the obstacles facing eradication efforts in Afghanistan. Until 2000, negotiated cease-fires and days of tranquility agreements during NIDs had been only partly successful. Cessation of polio vaccination activities in mid-1997 in northern Afghanistan as a result of ongoing conflict may have facilitated the large polio outbreak that occurred in Kunduz province in 1999 (4). Innovative measures and local peace initiatives will continue to be needed to create opportunities for reaching and vaccinating isolated populations. Afghanistan is preparing the implementation of five NID rounds in 2001. Plans are being developed to conduct focal mass campaigns in large, high-risk areas during the summer of 2001. Improved and timely stool specimen collection from AFP case-patients will be necessary to obtain data for targeting these campaigns and eliminating the last reservoirs of poliovirus circulation. Meeting these challenges will require the continued support of polio eradication partners¶. References
* Djibouti, Egypt, Libya, Morocco, Somalia, Sudan, and Tunisia in northern and eastern Africa; Bahrain, Kuwait, Oman, Qatar, Saudi Arabia, United Arab Emirates, and Yemen in the Arabian peninsula; Iraq, Jordan, Lebanon, Syria, and the Palestinian National Authority in the Middle East; Afghanistan, Iran, and Pakistan in Asia; and Cyprus. †Mass campaigns over a short period (days to weeks) in which two doses of OPV are administered to all children in the target group (usually aged 0--4 years), regardless of vaccination history, with an interval of 4--6 weeks between doses. § Two stool specimens collected 24 to 48 hours apart within 14 days of onset of paralysis that arrive in the laboratory in good condition. ¶ Polio eradication in Afghanistan is supported by the national government. External support is provided by global polio eradication partners, including Rotary International, United Nations Children's Fund (UNICEF), WHO, the governments of the United States, Great Britain, Denmark, Norway, Netherlands, Sweden, Luxemburg, Germany, and the European Community. Table 1 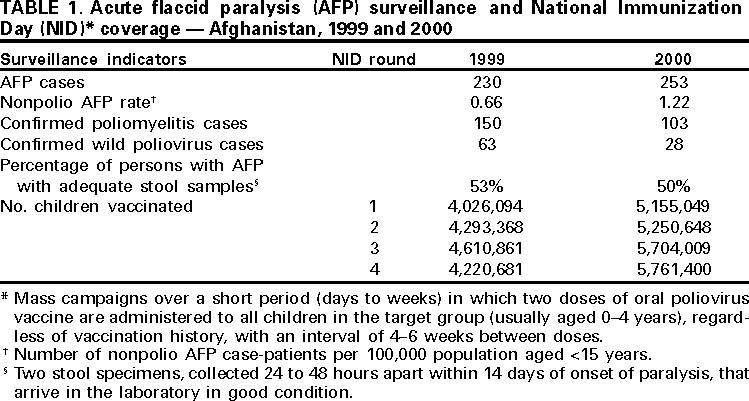 Return to top. Figure 1 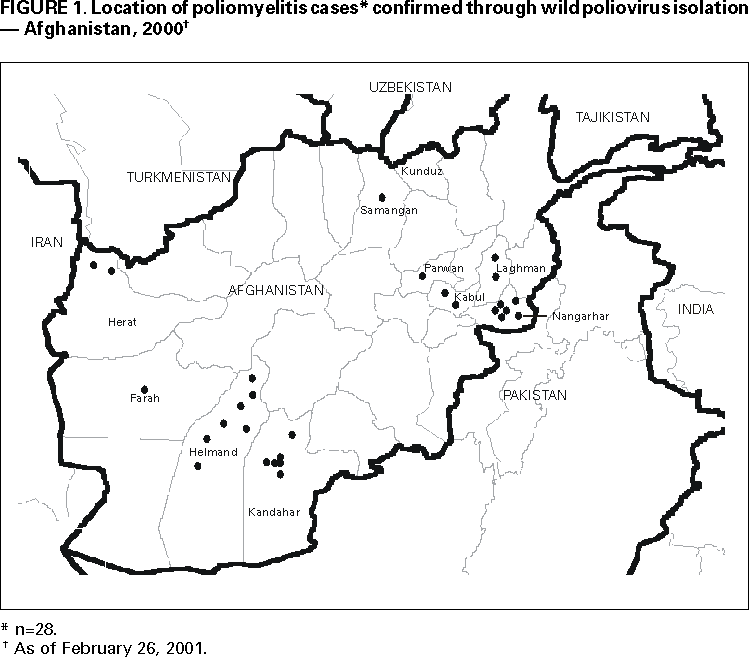 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 3/1/2001 |
|||||||||
This page last reviewed 5/2/01
|