 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Progress Toward Poliomyelitis Eradication --- Eastern Mediterranean Region, 1999--September 2000In 1988, the Regional Committee for the Eastern Mediterranean Region* (EMR) of the World Health Organization (WHO) adopted a resolution to eradicate poliomyelitis from the region by 2000. Since then, substantial progress has been made in vaccination and surveillance and, by the end of the year, 19 of the 23 EMR countries are expected to have interrupted poliovirus transmission. This report summarizes progress toward this goal from January 1999 through September 2000. Routine vaccination coverage. In 1999, the regional reported coverage with at least three doses of oral poliovirus vaccine (OPV3) by age 1 year was 83% (range: 18%--100%), compared with 82% in 1998. OPV3 coverage of >90% was reported from 14 countries. Coverage levels of <80% were reported from Afghanistan (32%), Djibouti (27%), Pakistan (80%), Somalia (18%, only northern regions reporting), Sudan (77%), and Yemen (72%). These countries represent more than half of the total regional population. Compared with reported administrative data, surveys in some of these countries have identified lower coverage rates. Supplementary vaccination activities. During 1999, National Immunization Days (NIDs)† were conducted in 20 of the 23 countries of the region. Iran and Tunisia conducted targeted subnational campaigns in provinces at risk for poliovirus importation and/or with suboptimal vaccination coverage, and NIDs have not been considered necessary in Cyprus. In 2000, several countries that have been polio-free have scaled down the scope of supplementary vaccination activities from NIDs to subnational or local campaigns. During 1999--2000, NIDs and other supplementary vaccination activities have been intensified in countries with persistent poliovirus circulation (Afghanistan, Egypt, Iraq, Pakistan, Somalia, and Sudan). In 1999, each of these countries either conducted two pairs (four rounds) of NIDs (Afghanistan, Egypt, and Iraq) or one pair of NIDs and one pair of large-scale subnational campaigns (Pakistan, Somalia, and Sudan). During 2000, each of these six countries will conduct two pairs of NIDs and additional mopping up or subnational campaigns. The quality of campaigns in these remaining countries where polio is endemic has been improved substantially through house-to-house vaccination, greater emphasis on high-risk areas, improved planning and supervision, additional financial resources, and increased technical consultation. Campaigns are coordinated among groups of contiguous countries within EMR. Coordination with the European region has led to elimination of the poliovirus reservoir in the border areas of Iran, Iraq, Syria, and Turkey (1). Cross-border coordination will continue between Afghanistan, Pakistan, and Iran. Increasing attention is being focused on collaboration with the regional office of WHO for Africa to coordinate eradication activities among countries of the Horn of Africa and countries that border western and southern Sudan. Surveillance. All member countries have established acute flaccid paralysis (AFP) surveillance. Fifteen countries (Bahrain, Egypt, Iran, Iraq, Jordan, Lebanon, Libya, Oman, Pakistan, Palestine, Qatar, Saudi Arabia, Syria, Tunisia, and Yemen) achieved or exceeded the WHO-established minimum AFP reporting rate indicative of a sensitive surveillance system (>1 nonpolio AFP case per 100,000 children aged <15 years) during 1999 (Table 1). Among the eight remaining countries, the annualized nonpolio AFP reporting rates during 2000 have exceeded one in Afghanistan, Kuwait, Somalia, and Sudan. The regional average reporting rates for nonpolio AFP in 1999 and 2000 are 1.1 and 1.3 (annualized), respectively. During 1999 and 2000, two adequate stool samples were collected from 67% and 71% of the reported persons with AFP in EMR, respectively. During 1999, nine countries (Bahrain, Cyprus, Iraq, Jordan, Kuwait, Oman, Palestine, Syria, and Tunisia) achieved the WHO-recommended target of collecting two adequate stool specimens from at least 80% of persons with AFP. During 2000, an additional four countries (Egypt, Lebanon, Libya, and Saudi Arabia) achieved this target. EMR laboratory network. The EMR laboratory network consists of 12 laboratories (eight national and four regional reference laboratories). All network laboratories have been fully or provisionally accredited by WHO. As of September 2000, the EMR laboratory network tested 4129 stool specimens obtained from 1947 (96%) of 2028 persons with reported AFP (or their contacts) from 21 EMR countries. Specimens from an additional 142 persons with AFP reported from Somalia and southern Sudan were tested in the laboratory network of the African region. Laboratory results were reported on time (within 28 days of receipt of specimen) for >80% of stool specimens during 1999--2000. Genetic sequence analyses are performed routinely on all wild poliovirus isolates in the region. Recent sequence data have identified separate virus reservoirs shared between Pakistan and Afghanistan and between Chad and Sudan. With improvements in surveillance, independent and unique transmission chains of poliovirus types 1 and 3 have been identified in Afghanistan, Somalia, and Sudan. Communities with persistent foci of virus transmission have been better delineated in Egypt. Sequencing of a recent wild poliovirus isolate obtained in Syria confirmed that the strain was imported recently from southern Asia. Incidence of polio. Compared with the same period in 1999, the number of confirmed cases of polio reported through September 2000 in the EMR has decreased by approximately 50% (from 619 to 314) despite substantial improvements in AFP surveillance. Compared with 13 EMR countries in 1999, 16 have reported no cases during 2000. However, during 1996--2000, six countries (Afghanistan, Egypt, Iraq, Pakistan, Sudan, and Somalia) have reported cases with indigenous strains of wild poliovirus (Figure 1). In 1999, Iran and Syria reported cases associated with imported poliovirus strains. Intensive control measures composed of multiple NID rounds and mopping up campaigns have led to cessation of the polio outbreak in Iraq (2). The last virologically confirmed case-patient from this outbreak had paralysis onset in January 2000. Since late 1999, wild poliovirus transmission in Egypt has been localized to a few districts in four governorates. The latest person with virologically confirmed polio in Egypt had onset in late May 2000. Expansion of surveillance in southern and central Somalia has led to identification of an outbreak of polio caused by wild poliovirus types 1 and 3 in Mogadishu, where, since January 2000, 38 cases of virologically confirmed polio have been identified. During 1999--2000, Pakistan continued to report the largest number of cases and has contributed more than 60% of the total number of virologically confirmed cases in the region. However, from January through September 2000, the number of virologically confirmed cases has declined 46% in Pakistan compared with the same period in 1999. The Regional Commission for Certification of Poliomyelitis Eradication has reviewed national documentation of polio-free status from nine countries with high-quality AFP surveillance that have not reported cases of polio for several years. The commission has favorably reviewed reports from Bahrain, Iran, Jordan, Kuwait, Oman, Saudi Arabia, Syria, and Tunisia. Reported by: Regional Office for the Eastern Mediterranean Region, Cairo, Egypt. Dept of Vaccines and Biologicals, World Health Organization, Geneva, Switzerland. Respiratory and Enteric Viruses Br, Div of Viral and Rickettsial Diseases, National Center for Infectious Diseases; Vaccine Preventable Disease Eradication Div, National Immunization Program, CDC. Editorial Note:Remarkable progress toward polio eradication has occurred in the member states of EMR since 1988. By the end of 2000, poliovirus transmission probably will be interrupted in all but four EMR countries. Improved local level planning and supervision, house-to-house vaccination, community mobilization, and heightened political commitment have enabled vaccination of an increasing number of children, especially among hard-to-reach and high-risk populations. These activities have necessitated the mobilization of financial and human resources and the development of local administrative capacity. AFP surveillance in the region is increasingly guiding planning, coordination, and targeting of vaccination activities and has identified virus reservoirs shared between countries or previously unknown foci of virus transmission. Despite the progress, gaps remain in the quality of supplementary vaccination activities and in geographic representation of AFP surveillance in areas of conflict. Countries with armed conflict and/or high population density, poor sanitation, low OPV3 coverage, and weak or absent health infrastructure have posed obstacles to interruption of virus transmission (3--5). In polio-free countries of the EMR, maintenance of high OPV3 coverage and targeted supplementary vaccination activities will be necessary to minimize the spread of any poliovirus that may be introduced through importation. Polio eradication in the region has entered its final phase. High priority polio eradication activities planned for this phase include 1) rapid completion of program intensification and expansion in the remaining countries where polio is endemic to ensure interruption of poliovirus transmission in the region by the end of 2001 or soon after; 2) rapid geographic expansion of AFP surveillance in countries affected by conflict and difficult access to populations; 3) maintenance of high-quality surveillance in polio-free countries; 4) containment of poliovirus stocks and potentially infectious material in laboratories throughout the region; 5) documentation of polio-free status by each country for review by the regional commission and certification of polio eradication in the region by the end of 2004; and 6) an increased focus on strengthening routine vaccination programs and vaccine-preventable disease surveillance. Implementing these high priority activities to achieve polio eradication and its certification will require the continued support of national governments and partner agencies.§ References
* The 23 member countries are Djibouti, Egypt, Libya, Morocco, Somalia, Sudan, and Tunisia in northern and eastern Africa; Bahrain, Kuwait, Oman, Qatar, Saudi Arabia, United Arab Emirates, and Yemen in the Arabian peninsula; Iraq, Jordan, Lebanon, Syria, and the Palestinian National Authority in the Middle East; Afghanistan, Iran, and Pakistan in Asia; and Cyprus. † Mass campaigns over a short period (days to weeks) in which two doses of OPV are administered to all children in the target age group (usually age <5 years) regardless of previous vaccination history, with an interval of 4--6 weeks between doses. § Support of polio eradication activities in EMR is provided mainly by governments of member states and by Rotary International, CDC, the government of the United Kingdom through the Department of Foreign and International Development, the government of Japan through the Japanese International Cooperative Agency, the government of Canada through the Canadian International Development Agency, the government of Denmark through Danish International Development Assistance, Sultanate of Oman, the governments of Norway and Italy, the United Nations Foundation, and the U.S. Agency for International Development. Table 1 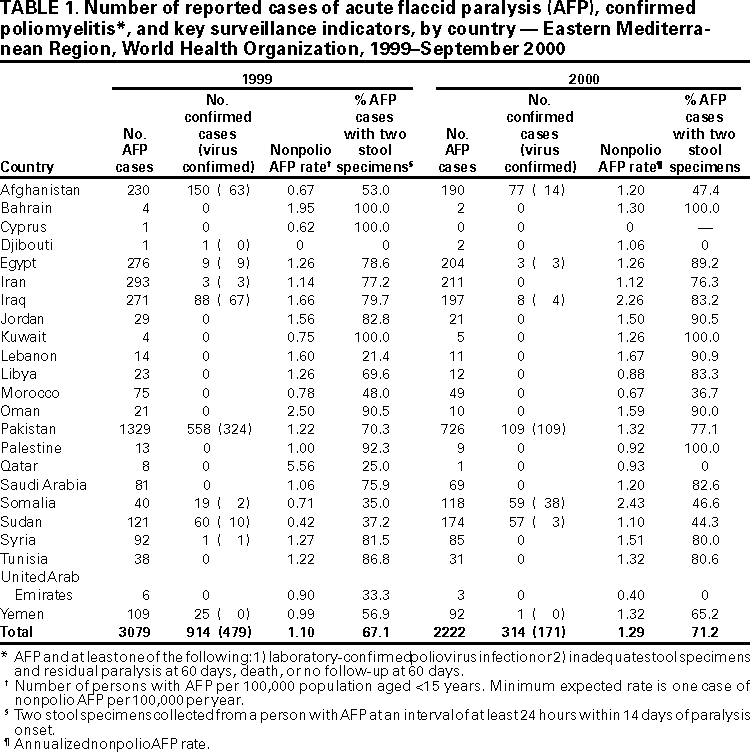 Return to top. Figure 1 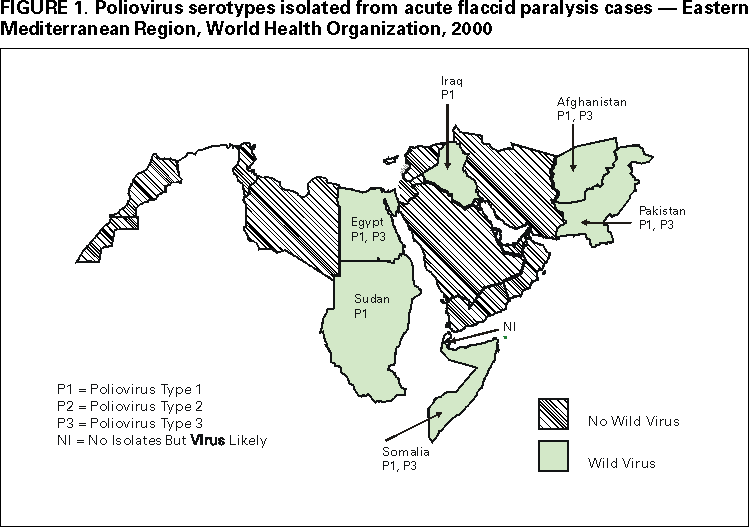 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 11/16/2000 |
|||||||||
This page last reviewed 5/2/01
|