

|

|

Volume
3:
No. 1, January 2006
ORIGINAL RESEARCH
The Behavioral and Clinical Effects of Therapeutic Lifestyle Change on
Middle-aged Adults
Steven G. Aldana, PhD, Roger L. Greenlaw, MD, Hans A. Diehl, DrHSc, MPH,
Audrey Salberg, RN, Ray M. Merrill, PhD, MPH, Seiga Ohmine, Camille Thomas, MS
Suggested citation for this article: Aldana SG, Greenlaw RL, Diehl
HA, Salberg A, Merrill RM, Ohmine S, et al. The behavioral and clinical
effects of therapeutic lifestyle change on middle-aged adults. Prev Chronic
Dis [serial online] 2006 Jan [date cited]. Available from: URL: http://www.cdc.gov/pcd/issues/2006/
jan/05_0088.htm.
PEER REVIEWED
Abstract
Introduction
Chronic diseases such as cancer, cardiovascular disease, stroke, and diabetes
are responsible for most deaths in the United States. Lifestyle factors —
poor nutrition, sedentary living, and tobacco use — appear to play a
prominent role in the development of many chronic diseases. This study determined the
behavioral and clinical impact of a therapeutic lifestyle-modification
intervention on a group of community volunteers.
Methods
Participants included 348 volunteers aged 24 to 81 years from the Rockford,
Ill, metropolitan area who participated in a randomized clinical trial.
The intervention group attended a 40-hour educational course delivered as
lectures during a 4-week period. Participants learned the importance of making
better lifestyle choices and how to make improvements in nutrition and
physical activity. Changes in nutrition, physical activity behavior, and
several chronic disease risk factors were assessed at baseline and 6 months.
Results
Intervention participants showed significant 6-month improvement in all
nutrition and physical activity measures except calories from protein and
whole-grain servings and all clinical measures except blood glucose, total
cholesterol, triglycerides, and high-sensitivity C-reactive protein. Total
cholesterol and low-density lipoprotein cholesterol were worse after 6 months in both
groups but only significantly worse in the control group. The control group
experienced small but significant improvements in systolic and diastolic blood
pressure and high-density lipoproteins. Change-score comparisons between the
intervention and control groups were significant for all nutrition and
physical activity variables except total steps per week and daily sodium
intake and were also significant for the clinical measures of weight, body fat,
and body mass index.
Conclusion
This therapeutic lifestyle-modification program can significantly improve
nutrition and physical activity behavior and can reduce many of the risk
factors associated with common chronic diseases.
Back to top
Introduction
Chronic diseases such as cancer, cardiovascular disease, stroke, and
diabetes are responsible for most deaths in the United States (1). Between 70%
and 90% of these deaths are believed to be caused by poor nutrition, sedentary
living, and tobacco use and are preventable (2-4). These lifestyle factors
appear to play a prominent role in the mechanisms and processes that lead to
the development of many chronic diseases. The largest reductions in chronic
disease prevalence in the United States will be achieved when individuals
adopt and maintain lifestyles that include a healthy diet and regular physical
activity.
During the 1980s, Nathan Pritikin conducted several in-patient
lifestyle-change programs that documented how a low-fat, high–complex-carbohydrate,
high-fiber diet and regular exercise could improve blood lipid levels and insulin
sensitivity (5-7). Variations of this holistic approach to preventing and
arresting chronic diseases have more recently been evaluated in randomized
clinical trials such as the PREMIER clinical trial (8), the DASH dietary study
(9), and other trials in the United States, United Kingdom, and New Zealand
(10-12). Most of these trials used inpatient treatment or controlled feeding
to encourage and monitor changes in diet and physical activity. All of them
demonstrated reductions in cardiovascular risk factors, including obesity,
blood pressure, and blood lipid levels.
The Coronary Health Improvement Project (CHIP) was created with the goal of
reducing chronic diseases and improving the overall health of the public by
providing a lifestyle-change program to both the community and the workplace
(13). The CHIP is a 40-hour live-lecture educational course that highlights
the importance of making better lifestyle choices for reducing chronic disease
risk factors. A one-group pretest–posttest analysis of the program revealed
that after 4 weeks, participants significantly reduced their blood pressure, blood
glucose, body weight, and total and low-density lipoprotein (LDL) cholesterol
(13). This exploratory study demonstrated that the program had the potential
to improve not only coronary risk factors but also the risks associated with
cancer, diabetes, and the metabolic syndrome. These results were repeated in a
quasi-experimental design that included results from six groups of working
adults (14).
A large randomized clinical trial was initiated to further explore the
effect of the CHIP (15). Six-week results from this study revealed that adults
who completed the program improved their nutrition and physical activity behavior
and reduced cardiovascular disease risk factors (15). We present the behavioral and clinical changes that participants in this
therapeutic lifestyle-change program experienced after 6 months.
Back to top
Methods
Subject recruitment and study design
Recruitment was conducted by the SwedishAmerican Center for Complementary
Medicine (SACCM) using targeted advertising, marketing through the
SwedishAmerican Health System Centers of
Excellence, CHIP alumni groups, corporate client sites, and the SwedishAmerican
Health System. Recruitment efforts were aimed at adults (aged
at least 18 years) in the greater Rockford, Ill, metropolitan area. To be
enrolled in the study, each participant had to be willing to start participating in
the program in 1 month or in 7 months. Figure 1 shows participant
progress through the study. Eligible and interested participants provided
informed consent. Participants were highly encouraged to participate with a
spouse or significant other and were randomized as a paired unit. All other
participants were randomized as individual units. The allocation sequence was
created using a random number generator. Program sign-up, randomization, and
group assignments were made by the study coordinator. The study was approved
by the Institutional Review Board of the SwedishAmerican Health System on
August 29, 2002.
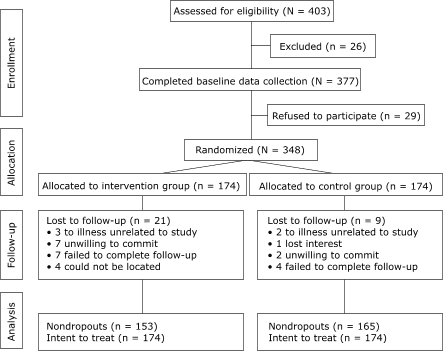
Figure 1. Process for a therapeutic
lifestyle-modification intervention with a group of community volunteers,
Rockford, Ill. [A text description of this model is
also available.]
Intervention
The intervention for this study was a live version of the CHIP (13).
Participants met for 4 weeks — 4 times each week for 2 hours — to
receive instruction during April 2003. The curriculum included the following topics: modern
medicine and health myths, atherosclerosis, coronary risk factors, obesity,
dietary fiber, dietary fat, diabetes, hypertension, cholesterol, exercise,
osteoporosis, cancer, lifestyle and health, the Optimal Diet, behavioral
change, and self-worth.
In conjunction with CHIP lectures, participants received a textbook and
workbook that closely followed the curriculum topics and included assignments
with learning objectives for every topic. Copies of these materials can be
obtained from CHIP at www.chipusa.org*. Assignments were designed to
help participants understand and integrate the information presented. Dietitians
and medical professionals spoke to the group weekly, introducing them to the
latest nutritional and medical information related to the prevention of
chronic diseases. Participants had access to scheduled shopping tours and
cooking demonstrations given by a dietitian.
The diet guidelines approximate the recommendations previously used in the
Pritikin program (5-7), and the exercise guidelines are from the Surgeon
General’s Report on Physical Activity and Health (16). Participants were
encouraged to follow preset dietary and exercise goals. The dietary goal
was to adopt a more plant-food–based diet that emphasizes as-grown,
unrefined food. Participants were encouraged to eat the following foods: whole
grains, legumes, vegetables, and fresh fruits. In addition, the diet was low
in fat (less than 20% of energy), animal protein, sugar, and salt; very low in
cholesterol; and high in fiber. Concurrently, program participants were
encouraged to work toward walking or exercising for at least 30 minutes each
day. Participants were given a pedometer and encouraged to keep an exercise
log to record the miles walked each day. In addition, at the completion of the
program, participants were encouraged to join the Rockford CHIP alumni
association for an annual cost of $25 for an individual or $35 for a couple.
The purpose of the alumni organization was to help prevent relapse and help
participants maintain their new behaviors. Alumni receive a monthly newsletter
that contains news of health-promoting community events such as healthy
dinners, walking groups, and support-group meetings. The alumni were
encouraged to attend special lectures on healthy living and ways to avoid
relapse.
The primary objectives of this therapeutic lifestyle-change program were to
improve cognitive understanding of the importance of healthy lifestyles,
nutrition, and physical activity behavior and reduce risk factors associated
with hypertension and cardiovascular disease. The cost to participate in the
entire CHIP was $395 per person or $595 per couple.
Measures
Variables gathered included cognitive and behavioral measurements and
physiologic outcomes related to chronic disease. Demographic data were
collected at baseline in April 2003. Attendance at each of the classes was tracked and
averaged. Participants attended an average of 89% of the classes.
The intervention was designed to assist individuals in adopting healthy
eating and physical activity behaviors. To assess dietary intake, the Block 98
full-length dietary questionnaire was used (Block 98.2, Block Dietary Data
Systems, Berkeley, Calif). The Block 98 questionnaire has been extensively
studied and validated (17). The questionnaire contains self-reported data and
is optically scanned and scored. The questionnaire measures the following
variables (in addition to others) on a per-day basis: nutrients obtained from food;
percentage of calories from fats, carbohydrates, and protein; fiber from different sources; and food group servings
per day.
To ascertain energy expenditure contributed by physical activity, a 7-day
self-recorded pedometer log was maintained by each participant. Participants
wore the Walk4Life Model 2000 Life Stepper pedometer (Walk4Life Inc,
Plainfield, Ill) on a belt at the right hip directly above the right kneecap
each day for 7 days. Immediately before going to bed, participants recorded
the number of steps for the day and reset the pedometer. Strike counts from
pedometers are a valid and reliable method of monitoring and measuring
free-living physical activity (18).
The primary outcome variables for this study included several chronic
disease risk factors. The following data were collected from April to October
2003. Blood was drawn from participants (after a 12-hour fast) by
phlebotomists from the SwedishAmerican Health System’s outpatient laboratory using a vacutainer
(Becton-Dickinson Vacutainer Systems, Rutherford, NJ). Samples were
allowed to clot and were centrifuged. Clinical analyses were completed at the SwedishAmerican Health System laboratory. Lipid analysis followed the lipid
standards provided by the Centers for Disease Control and Prevention. Glucose,
total cholesterol, high-density lipoprotein (HDL), and triglyceride
concentrations were determined using Beckman-Coulter LX-20 instrumentation
(Beckman Coulter, Inc, Fullerton, Calif). Glucose measurements were obtained
with the oxygen-rate method using a Beckman oxygen electrode; cholesterol
measurements were obtained with the timed-endpoint enzymatic method using
cholesterol oxidase; triglyceride measurements were obtained with the
timed-endpoint enzymatic method using glycerol kinase; and HDL measurements
were obtained with the homogeneous timed-endpoint method using polyanion
detergent to separate HDL and non-HDL lipids. For participants with
triglyceride values below 400, LDL values were calculated as follows: LDL =
total cholesterol – HDL – (triglycerides/5) (19). High-sensitivity
C-reactive protein (CRP) measurements were determined using a microplate
protocol based on a latex-bead–enhanced immunoturbidity assay (20). Glucose
measurements were determined using a Kodak Ektachem (Kodak, Rochester, NY).
Trained program staff took blood pressure measurements. Blood pressure was
measured in participants after a 5-minute rest, using the guidelines set forth
by the American Heart Association. Weight and height were measured using
standard medical weight and height scales recently calibrated by the
biometrics department of the SwedishAmerican Health System. Percentage of body
fat was estimated with Tanita TBF-300A Body Composition Analyzer/Scale using
bioelectrical impedance analysis (Tanita, Tokyo, Japan) (21). Body mass index
(BMI) was determined using the following formula: weight (kg)/height (m2).
Statistical analyses
Cross-tabulations were used to perform bivariate analyses between selected
variables, with statistical significance based on the chi-square test for
independence. For testing differences in means, t tests were used.
Because multiple pair-wise tests were performed, an adjusted α was used
to minimize the overall probability of committing a type I error. The modified
α is .0001, based on the Bonferroni correction, 28 pair-wise tests, and α =
.05. This conservative α was used to determine significance for
data in Tables 2 through 7. Risk factor cut-points (Tables 6 and 7) were previously
established (22,23) and categorized accordingly. Results are based on the
intent-to-treat method in which all participants were retained in the
analyses. Where participant data were lost to follow-up, the last-test
carry-forward method was applied to the participant’s most recent data. The
results did not differ significantly when participants lost to follow-up were
dropped from the analyses. These results are not reported. Analyses were
performed using SAS 9.0 (SAS Institute Inc, Cary, NC). Procedure statements
used in SAS for assessing the data were PROC UNIVARIATE, PROC FREQ, PROC TTEST,
and PROC GLM.
Back to top
Results
There were 318 participants who completed both baseline and 6-month
evaluations. An additional 30 completed the baseline evaluation but not the
6-month evaluation. Of these lost to follow-up, 21 were in the intervention
group, and 9 were in the control group (Figure 1).
Analyses were based on 348 participants. Ages ranged from 24 to 81 years,
with little difference in the mean age between intervention and control groups
(50.1 years, intervention group; 50.8 years, control group, t346 = −0.57,
P = .57). A description of participants in both intervention and
control groups is presented according to selected demographic characteristics in
Table 1. There were no statistically significant
differences between groups for these variables. Within each group, the
majority of participants had the following characteristics: female, white,
married, an annual family income of at least $60,000, and at least some college education. Of the
intervention participants, 47 (27%) joined the CHIP alumni association.
Because the unit of randomization was pairs for those who
participated with a partner and individuals for those who participated
as individuals, comparisons were made of the effect of the program between
pairs and individuals. Of the 348 randomized participants, 146 (42%)
participated as pairs. There were no significant differences in the outcomes of
pairs and individuals. After 6 months, participants in the intervention group
experienced significant improvements in all physical activity and nutrition
variables except calories from protein and whole-grain servings (Table 2). Changes in the control
group were generally not statistically significant, or they were much smaller
in magnitude than the changes in the intervention group. For each variable except total steps
per week and daily sodium intake, the change observed in physical activity or
nutrition was significantly greater for participants in the intervention group
compared with the control group (Table 3). The control group consumed
significantly more fat calories and fewer whole-grain servings at 6-month
follow-up compared with the control group at 6-month follow-up.
After 6 months, participants in the intervention group showed significant
reductions in BMI, weight, body fat, systolic and diastolic blood pressure,
and resting heart rate (Table 4).
The control group experienced significant improvements in systolic and
diastolic blood pressure and HDL, but total cholesterol and LDL were
significantly worse. For BMI, weight, and body fat, changes were significantly greater for
participants in the intervention group compared with the control group
(Table
5).
Mean baseline, 6-month, and change in mean scores are presented according
to standard health risk cut-points for the risk factor variables according to
intervention group (Table 6) and control group
(Table 7). This analysis
stratifies results according to risk status. Individuals with low risk would
not be expected to experience large changes, but risk values considered to be
high would be expected to change significantly. For the intervention group,
the distributions favorably changed between baseline and 6 months for BMI,
systolic blood pressure, and diastolic blood pressure. Corresponding
significant change in the distribution between baseline and 6 months was
observed in the control group for systolic blood pressure and diastolic blood
pressure but not for BMI. Favorable changes
in risk behaviors were generally higher and more likely to be significant for
individuals in the intervention group than for individuals in the control
group.
Whereas total cholesterol significantly increased between baseline and 6
months for participants in the control group, no significant difference was
observed in the intervention group. For both intervention and control groups,
total cholesterol significantly increased among participants with total cholesterol in the
normal range and decreased (but not significantly) for those with cholesterol in the
high-risk category. Cholesterol medication played a minimal role in the change
observed in cholesterol. At baseline, there were 77 participants in the
intervention group who reported using blood pressure medication. At 6 months,
60 participants (75%) indicated no change in their medication over the study
period, 9 participants (11.2%) indicated a dosage increase, and 11
participants (13.8%) indicated a dosage decrease. There was not a significant
difference in the use of blood pressure medication from baseline to 6 months
between the intervention and control groups (χ21 = 1.14, P =
.56).
Back to top
Discussion
Therapeutic lifestyle change can result in significant improvements in
nutrition and physical activity behavior and reductions in many cardiovascular
disease risk factors. Six months after the intervention began, program
participants continued to demonstrate dramatic improvements in nutrition and
physical activity behavior. Increases in the number of servings of fruit and
vegetables and whole grains, increases in physical activity, and decreases in
dietary sodium are likely responsible for the improvements in both systolic
and diastolic blood pressure. Intervention group participants consumed 2.3
more servings of fruit and vegetables per day at 6 months compared with
baseline. In the PREMIER study (8), participants who completed a
behavior-change program and adopted the DASH diet increased fruit and
vegetable servings by 3.0 servings after 6 months. Those PREMIER program
participants decreased their percentage of calories from fat by 9.5% and lost
an average of 5.8 kg of body weight. This compares to a percentage fat
reduction of 8.2% and a 4.5 kg weight loss for intervention participants in
the present study.
At baseline, the intervention group included 77 participants who were at
least diastolic prehypertensive at 6 months; this number decreased by 44% to
43 participants at 6 months (Table 6). The number of intervention-group participants who were at least systolic prehypertensive at
baseline declined by 20%, from 122 participants at baseline to 98 at 6 months. The average reductions in blood pressure were greater than the
reductions reported in the DASH study (9) and comparable with the results of
the PREMIER clinical trial (8).
Previous reports of the CHIP intervention showed sharp improvements in blood lipid
levels at 6 weeks,
but most of these changes
disappeared at 6 months (7). Other therapeutic lifestyle trials that lasted
longer than 3 months and included lipid outcomes reported similar findings
(10-12,24). In this study, dietary cholesterol among the intervention group
was reduced by 122 mg/day (a 56% reduction), and dietary saturated fat was cut
by half. Despite these favorable changes in dietary cholesterol precursors, a
return to previous lipid levels suggests that there is a significant increase
in endogenous cholesterol, most of which appears to be LDL cholesterol (25).
It is also possible that these changes in blood lipid levels were affected by
seasonal variation. Without more accurate measures of endogenous cholesterol
biosynthesis, it is impossible to determine the exact cause of the cholesterol
increase (26).
Pedometer data show that program participants increased physical activity
by 30%. The average number of steps for the intervention group after 6 months
did not meet the recommended 10,000 steps per day (27). For this predominately
middle-aged and obese population, however, an increase in physical activity of
30% likely contributed to risk factor reductions. When combined with diet
changes, improvement in physical activity is the likely explanation for
the percentage decreases in BMI (−5%), weight (−5%), and percentage body fat (−6%) among the
intervention group. Improved physical activity was also associated with a
significant decrease in resting heart rate, a correlated measure of cardiorespiratory fitness thought to be caused by increased heart size, blood
volume, stroke volume, and cardiac output (28).
Poor nutrition and sedentary living are associated with a constellation of
risk factors, some identified in the metabolic syndrome, and all linked to
common chronic diseases (29). Improvements in nutrition and physical
activity are associated with significant reductions in diabetes risk as whole
body glucose tolerance improves, insulin sensitivity increases, and the amount
of glucose transporter (GLUT4) increases (30). The number of individuals with
diabetes (glucose ≥126 mg/dL) in the intervention group was reduced by 19%,
demonstrating that this therapeutic lifestyle-change program improves insulin
sensitivity. Similar results were reported by other lifestyle trials reporting
glucose findings (11,12).
These improvements in behavior and risk are not unexpected because the
intervention lectures were structured on the health belief and
transtheoretical models. Video clips, testimonials, role playing, short
presentations from physicians, social support strategies, food selection and
planning activities, and other behavior-change–driven pedagogical activities
helped to encourage participants to enthusiastically evaluate personal
behaviors and commit to make lifestyle changes.
Most of the participants were white and sufficiently self-motivated to
volunteer to participate in the intervention. On average, participants were
slightly more educated than the community average. Participants had lifestyles
that permitted them to attend most, if not all, of the classes. This is
evident in the high rate of attendance to this time-intensive program. These
delimitations threaten the generalizability of these findings and make
application of the intervention to other populations problematic. Because the
participants were self-selected, the results from this intervention may
represent a best-case scenario.
Despite the apparent effect of this intervention, there are some
shortcomings associated with the study design. Both the physical activity and
nutrition data were self-reported. For some variables, the control group also
experienced significant improvement. Significant decreases were observed in
the control group in percentage of fat calories and dietary-fat grams, sodium grams,
and total calories as well as small increases in total steps. In addition, the
control group experienced similar improvement in blood pressure compared with
the intervention group. There are more than 27 restaurants in the Rockford
metropolitan area that offer healthy, CHIP-recommended menu items, which could
have contributed to improvements in the control group. When conducting
lifestyle trials, the question of what to do with the control group is
difficult to answer because there is no such thing as a lifestyle placebo.
After participants were assigned to an intervention or control group, some control-group participants
expressed happiness with their assignment because they had personal or
work-related conflicts that would have prohibited them from participating in
the intervention group. Others were
disappointed in their control-group assignment but realized when they agreed to participate in the research
study that there was always the chance that they would have to wait to
participate in the program.
This study indicates that an intervention that uses various behavior
modification tools, such as live lectures, workbooks, and professional advice,
and is implemented among a group of middle-aged volunteers can result in
reduced risk factors for cardiovascular disease after 6 months. Further
research is needed to examine the effects of the program on other populations.
Back to top
Author Information
Corresponding Author: Steven G. Aldana, PhD, 274 SFH, College of Health and
Human Performance, Brigham Young University, Provo, UT 84602-2214.
Telephone: 801-422-2145. E-mail: steve_aldana@byu.edu.
Author Affiliations: Roger L. Greenlaw, MD, Audrey Salberg, RN,
SwedishAmerican Center for Complementary Medicine, Rockford, Ill; Hans A.
Diehl, DrHSc, MPH, Lifestyle Medicine Institute, Loma Linda, Calif; Ray M.
Merrill, PhD, MPH, Camille Thomas, MS, College of Health and Human
Performance, Brigham Young University, Provo, Utah; Seiga Ohmine, Department of
Molecular and Microbiology, Brigham Young University, Provo, Utah.
Back to top
References
- Centers for Disease Control and Prevention. Deaths: leading causes for 2001
[Internet]. Atlanta (GA): Centers for Disease Control and Prevention,
National Center for Health Statistics; 2003. Available from: URL:
http://www.cdc.gov/nchs/data/nvsr/nvsr52/nvsr52_09.pdf.
- Stampfer MJ, Hu FB, Manson JE, Rimm EB, Willett WC.
Primary prevention
of coronary heart disease in women through diet and lifestyle.
N Engl J
Med 2000 Jul 6;343(1):16-22.
- Platz EA, Willett WC, Colditz GA, Rimm EB, Spiegelman D, Giovannucci E.
Proportion of colon cancer risk that might be preventable in a cohort of
middle-aged US men. Cancer Causes
Control 2000 Aug;11(7):579-88.
- Hu FB, Manson JE, Stampfer MJ, Colditz G, Liu S, Solomon CG, Willett WC.
Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. N Engl
J Med 2001 Sep 13;345(11):790-7.
- Barnard RJ, Lattimore L, Holly RG, Cherny S, Pritikin N.
Response of
non-insulin-dependent diabetic patients to an intensive program of diet and
exercise. Diabetes Care 1982 Jul-Aug;5(4):370-4.
- Barnard RJ, Massey MR, Cherny S, O'Brien LT, Pritikin N.
Long-term use
of a high-complex-carbohydrate, high-fiber, low-fat diet and exercise in the
treatment of NIDDM patients. Diabetes Care 1983 May-Jun;6(3):268-73.
- Rosenthal MB, Barnard RJ, Rose DP, Inkeles S, Hall J, Pritikin N.
Effects of a high-complex-carbohydrate, low-fat, low-cholesterol diet on
levels of serum lipids and estradiol. Am J Med 1985 Jan;78(1):23-7.
- Appel LJ, Champagne CM, Harsha DW, Cooper LS, Obarzanek E, Elmer PJ, et
al; Writing
Group of the PREMIER Collaborative Research Group.
Effects of comprehensive
lifestyle modification on blood pressure control: main results of the PREMIER
clinical trial. JAMA 2003;289(16):2083-93.
- Appel LJ, Moore TJ, Obarzanek E, Vollmer WM, Svetkey LP, Sacks FM, et al.
A clinical trial of
the effects of dietary patterns on blood pressure. DASH Collaborative Research
Group. N Engl J Med 1997 Apr 17;336(16):1117-24.
- Toobert DJ, Glasgow RE, Strycker LA, Barrera M Jr, Radcliffe JL, Wander
RC, et al.
Biologic and quality-of-life outcomes from the Mediterranean
Lifestyle Program: a randomized clinical trial.
Diabetes Care 2003
Aug;26(8):2288-93.
- Oldroyd JC, Unwin NC, White M, Imrie K, Mathers JC, Alberti KG.
Randomised controlled trial evaluating the effectiveness of behavioural
interventions to modify cardiovascular risk factors in men and women with
impaired glucose tolerance: outcomes at 6 months.
Diabetes Res Clin Pract
2001 Apr;52(1):29-43.
- McAuley KA, Williams SM, Mann JI, Goulding A, Chisholm A, Wilson N, et
al.
Intensive lifestyle changes are
necessary to improve insulin sensitivity: a randomized controlled trial.
Diabetes
Care 2002 Mar;25(3):445-52.
- Diehl HA.
Coronary risk reduction through intensive community-based
lifestyle intervention: the Coronary Health Improvement Project (CHIP)
experience. Am J Cardiol 1998;82:83T-87T.
- Aldana SG, Greenlaw R, Diehl HA, Englert H, Jackson R.
Impact of the
Coronary Health Improvement Project (CHIP) on several employee populations.
J
Occup Environ Med 2002;44(9):831-9.
- Aldana S, Greenlaw R, Diehl H, Salberg A, Merrill R, Ohmine S, Thomas
C. The effects of an intensive diet and physical activity modification program
on the health risks of adults. J Am Diet Assoc 2005 Mar;105(3):371-81.
- Centers for Disease Control and Prevention. Surgeon General’s Report on
physical activity and health [Internet]. Atlanta (GA): Centers for Disease
Control and Prevention, National Center for Chronic Disease Prevention and
Health Promotion. Available from: URL: http://www.cdc.gov/nccdphp/sgr/sgr.htm.
- Block G, Woods M, Potosky A, Clifford C.
Validation of a
self-administered diet history questionnaire using multiple diet records.
J
Clin Epidemiol 1990;43:1327-35.
- Hendelman D, Miller K, Baggett C, Debold E, Freedson P.
Validity of accelerometry for the assessment of moderate intensity physical activity in
the field. Med Sci Sports Exerc 2000 Sep;32(9 Suppl):S442-9.
- Sinha R, Block G, Taylor PR.
Determinants of plasma ascorbic acid in a
healthy male population. Cancer Epidemiol Biomarkers Prev
1992;1:297-302.
- Wu TL, Tsao KC, Chang CP, Li CN, Sun CF, Wu JT.
Development of ELISA on microplate for serum C-reactive protein and establishment of age-dependent
normal reference range. Clin Chim Acta 2002;322(1-2):163-8.
- Kyle UG, Genton L, Karsegard L, Slosman DO, Pichard C.
Single
prediction equation for bioelectrical impedance analysis in adults aged 20-94
years. Nutrition 2001;17(3):248-53.
- Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr, et
al; National Heart,
Lung, and Blood Institute Joint National Committee on Prevention, Detection,
Evaluation, and Treatment of High Blood Pressure; National High Blood Pressure
Education Program Coordinating Committee.
The Seventh Report of the Joint
National Committee on Prevention, Detection, Evaluation, and Treatment of High
Blood Pressure: the JNC 7 report. JAMA 2003 May 21;289(19):2560-72.
- National Institutes of Health. Third report of the National Cholesterol Education Program (NCEP)
expert panel on detection, evaluation, and treatment of high bood cholesterol
in adults (adult treatment panel III). Bethesda (MD): National Institutes
of Health, National Heart, Lung, and Blood Institute, National Cholesterol
Education Program; 2001 May (publication no. 01-3670).
- Tudor-Locke C, Bell RC, Myers AM, Harris SB, Ecclestone NA, Lauzon N,
et al.
Controlled outcome evaluation of the First Step Program: a daily
physical activity intervention for individuals with type II diabetes.
Int J
Obes Relat Metab Disord 2004 Jan;28(1):113-9.
- Sundram K, French MA, Clandinin MT.
Exchanging partially hydrogenated
fat for palmitic acid in the diet increases LDL-cholesterol and endogenous
cholesterol synthesis in normocholesterolemic women.
Eur J Nutr 2003
Aug;42(4):188-94.
- Di Buono M, Jones PJ, Beaumier L, Wykes LJ.
Comparison of deuterium
incorporation and mass isotopomer distribution analysis for measurement of
human cholesterol biosynthesis. J Lipid Res 2000 Sep;41(9):1516-23.
- Le Masurier GC, Sidman CL, Corbin CB.
Accumulating 10,000 steps: does
this meet current physical activity guidelines?
Res Q Exerc Sport 2003
Dec;74(4):389-94.
- McArdle WD, Katch FI, Katch, VL. Exercise physiology: energy,
nutrition, and human performance. Philadelphia (PA): Lea & Febiger
Publishers; 1991.
- Maki KC.
Dietary factors in the prevention of diabetes mellitus and
coronary artery disease associated with the metabolic syndrome.
Am J
Cardiol 2004 Jun 3;93(11A):12C-17C.
- Tuomilehto J, Lindstrom J, Eriksson JG, Valle TT, Hamalainen H,
Ilanne-Parikka P, et al.
Prevention of type 2
diabetes mellitus by changes in lifestyle among subjects with impaired glucose
tolerance. New Engl J Med 2001;344:1343-50.
Back to top
*URLs for nonfederal organizations are provided solely as a
service to our users. URLs do not constitute an endorsement of any organization
by CDC or the federal government, and none should be inferred. CDC is
not responsible for the content of Web pages found at these URLs.
|
|
