

|

|

Volume
2:
No. 1, January 2005
ESSAY
Diabetes and Mexicans: Why
the Two Are Linked
Reynaldo Martorell, PhD
Suggested citation for this article: Martorell R. Diabetes and Mexicans:
why the two are linked. Prev Chronic Dis [serial online] 2005 Jan [date
cited]. Available from: URL:
http://www.cdc.gov/pcd/issues/2005/
jan/04_0100.htm.
The Past
Obesity and diabetes were probably rare before the advent of agriculture.
Our ancestors, hunters and gatherers for millennia, had varied but
unpredictable diets. Studies of hunter-gatherers of the 20th
century suggest that animal sources dominated our ancient food basket, with
plants (fruits, vegetables, and nuts) providing only 20% to 40% of total energy (1). Modern and
presumably ancient hunter-gatherer populations, despite
a high-fat, high-protein diet, were free of the signs and symptoms of
noncommunicable diseases — a paradox. Perhaps energy needs were not always
met, thus keeping body sizes in check; also, the relative lack of salt and simple
carbohydrates, a mix of saturated and good fats, plenty of fiber, abundant micronutrients, a vigorous and active life, and less stress than we now endure may explain this finding. With the
food supply uncertain, one would expect individuals with “thrifty”
genotypes — genotypes that increase the ability to turn food to fat — to have a survival edge.
Agriculture brought a more predictable food supply but less variety.
Crops failed from time to time, bringing on famines when stores of grain
were depleted, but over time, agriculture allowed for increasingly larger
populations, with thrifty genotypes thriving as before. Super foods — such
as corn in Mesoamerica, the substance from which the Mayan gods in their
fourth attempt were finally able
to make man, according to the Popul Vuh, the sacred
book of the Maya — came to provide as much as 80% or more of energy needs.
Crowding brought new types of infections, which along with limited diets gave rise to
the nutritional deficiencies that have plagued humankind in recent millennia. Agriculture fostered
the development of highly stratified societies, and it became possible for a
few to lead a life of luxury. Until the 20th century, fatness was
a marker of wealth.
Back to top
The Present
Most of my professional career has been devoted to the study of hunger
and malnutrition in developing countries. As rates of child malnutrition
decline in Latin America and in other developing countries, the prevalence
of obesity is increasing rapidly, and I, like many of my colleagues, have
begun to study both ends of the spectrum — namely, deficiency and excess (2).
Economic development and urbanization are the engines
of the “nutrition transition” (3). Pathways include increased
food security, the availability of cheap sources of fat in the form of
vegetable oils, more eating away from home, the less arduous nature of
modern jobs, and increases in sedentary recreation (notably television).
These pathways have
transformed dietary and physical activity patterns and, as a result, tipped the balance in
favor of obesity (Figure 1).

Figure 1.
Possible causes of the nutrition transition and the emergence of obesity
in developing countries. Adapted from Martorell and Stein, 2001 (2), and Popkin, 1994 (3).
Some populations may be more susceptible to obesity (e.g., Pacific
Islanders, Native Americans) because of thrifty genotypes, as proposed by
the geneticist Neel some years ago (4). Thrifty phenotypes may also
increase susceptibility to obesity; some evidence suggests that poor intrauterine and
infant nutrition may also “program” individuals to be metabolically
thrifty, and if later times bring a life of abundance, these individuals
will be at risk for developing chronic diseases such as diabetes (5).
Back to top
The “Supersizing” of the Mexican People
Mexico is a country far along the nutrition transition. The Mexican National
Nutrition Survey 1999 showed that obesity (Body Mass Index [BMI] ≥30) among women
aged 18 to
49 increased from 9% in 1988 to 24% in 1999 (6). If we add
overweight (BMI = 25.0–29.9) to the mix, the percentage of overweight or obese
women increased from 33% to 59% in just one decade. The 1999 survey also
showed that the prevalence of stunting (low height-for-age, indicative of
child undernutrition) among preschool children in the indigenous rural south
of Mexico was 42%, as high as in many sub-Saharan African countries. Yet
the problem of obesity grew alarmingly among all sectors of society. All
socioeconomic groups, rural as well as urban areas, and all regions of
Mexico, including the impoverished South, showed equally
dramatic increases (Figure 2). Obesity and chronic diseases in Mexico can no longer be
dismissed as problems of the rich. However, poor Mexicans have a double
burden: child undernutrition in addition to obesity. As the nutrition transition
unfolds even further, as it has in Chile, obesity becomes more common among
the poor, as it is in the United States.
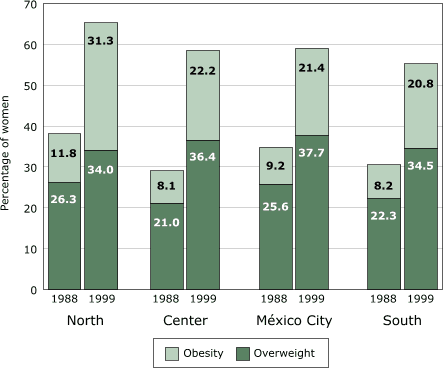
Figure 2.
Levels of overweight (BMI = 25.0–29.9) and obesity
(BMI ≥30.0) in 1988 and 1999 in women aged 18 to 49
in Mexico, by region. BMI indicates Body Mass Index. Data from Rivera et al, 2001 (6).
(A text description of this graph is also
available.)
Mexican Americans are one of the fattest groups in what is one of the
fattest nations on earth. Three out of four Mexican American adults (aged >20
years) were either overweight or obese at the end of the 20th
century (7).
Plentiful and unhealthy diets, many hours of television watching, and a
reluctance to exercise are some of the factors blamed. For example, a
study of Mexican children along the Mexico–U.S. border showed low intake of
fruits and vegetables and excessive consumption of soft drinks and high-fat
snacks (8).
Obesity is an easy, visible marker of the worldwide
pandemic of noncommunicable diseases for which considerable data from
around the world are available (2). Obesity is also a major risk factor for
type 2 diabetes, and where obesity is rising we can expect diabetes to
follow (9).
Back to top
The Type 2 Diabetes Pandemic
Diabetes is a growing problem worldwide. The prevalence of diabetes in
adults (aged >20 years) is projected to increase in developed countries from
6.0% in 1995 to 7.6% by 2025 (10). Diabetes in developing countries will
also increase from 3.3% to 4.9%, and because of initial population sizes and
growth, the increase in the number of people with diabetes will come
disproportionately from the developing world. The number of individuals with
diabetes will rise from 51 million to 72 million in developed countries, but
the number will rise from 84 million to 228 million in developing countries.
The three nations with the greatest numbers of individuals with diabetes in
1995 were India (19.4 million), China (16.0 million), and the United States
(13.9 million). In 2025, the rankings will be unchanged, but the absolute
number will increase dramatically in India (to 57.2 million) and China (to
37.6 million) and less so in the United States (to 21.9
million). Mexico, which was ninth in the world in 1995 (3.8 million), will
rise to seventh place by
2025 (11.7 million).
Diabetes is a serious public health problem among Mexicans and Mexican
Americans. Diabetes was found in 8.1% of Mexican adults in 2000 (11)
compared with 13.1% and 14.5% of Mexican American men and women in 1988–94
(12). In the United States, adults of Mexican origin, particularly men, had
higher rates of prevalence of diabetes than
non-Hispanic whites or blacks, as well as a greater degree of impaired
fasting glucose (Figure 3). The prevalence of diabetes in the United States is rising rapidly. The
prevalence of diabetes increased from 8.9% in 1976–1980 to 12.3% in
1988–94 among adults aged 40 to 74 (12). Mexican
Americans, the largest Hispanic/Latino subgroup in the United States, are
more than twice as likely to
have diabetes as non-Hispanic whites of similar age (13).
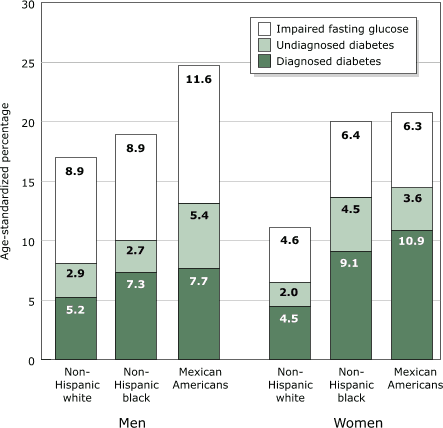
Figure 3.
Age-standardized prevalence of diagnosed and undiagnosed diabetes and
impaired fasting glucose in the U.S. population aged ≥20 years by sex and
ethnic group, based on the Third National Health and Nutrition Examination
Survey (NHANES III). Data from Harris et al, 1998 (12).
(A text description of this graph is also
available.)
Born in
Central America, I share a similar ancestry with Mexicans (Spanish and
Amerindian). Not surprisingly, diabetes runs in my family. Some statistics should scare me.
The lifetime risk of developing diabetes for U.S. individuals born in 2002
is about one in three for the general population, but about one in two for the Hispanic
population (14).
Back to top
Ancestry and Prenatal Exposure
Lifestyle characteristics are primarily responsible for the high levels
of obesity and diabetes among Mexicans, but other considerations are also
important. The San Antonio Heart Study began in 1979 and is a
population-based study of diabetes and cardiovascular disease in Mexican
Americans and non-Hispanic whites in San Antonio, Texas (9). One of the
interesting findings of the study is that the degree of Native American
ancestry is a major risk factor for diabetes, presumably because of
inherited thrifty genes (15).
The role of intergenerational mechanisms, specifically the risk of
developing diabetes in adulthood as a result of prenatal exposure to
diabetes, has become clear from studies of Pima Indians in Arizona (Figure
4). The prevalence of diabetes among adults aged 20 to 24 was found to be 1.4% if the mother
was free of diabetes, 8.6% if she was prediabetic (developed diabetes
after delivery), and 45.5% if she had gestational diabetes (16). Follow-up
studies over three decades reveal a steady rise in diabetes in Pima children
and adolescents. From 1967–76 to 1987–96, the prevalence of diabetes in
girls aged 10 to 14 years increased from 0.72% to 2.88%. In girls aged 15 to 19
years, the prevalence increased from 2.73% to 5.31% during the same period (17). The percentage of
youths (aged 10 to 19
years) who were exposed to gestational diabetes increased during this period (Figure 5). In 1967–76, 2.1% of youths were
exposed to gestational diabetes; by 1987–96, exposure had almost quadrupled
to 7.5% of pregnancies. The fraction of diabetes attributable to gestational
diabetes also rose markedly in youths aged 10 to 19 so that by
1987–96, more than one third of cases of diabetes could be attributed to
gestational diabetes. Also, more than 70% of persons with prenatal exposure
developed type 2 diabetes at 25 to 34 years of age (18). Clearly, the hyperglycemic intrauterine environment
brought on by gestational diabetes is an important determinant of early-onset type 2 diabetes
that is above any genetically transmitted susceptibility and is another example of fetal programming
(19). An
additional consequence is that 50% of women with gestational diabetes will
themselves develop diabetes within five years (20). The concern about
gestational diabetes is not limited to the Pima population. The incidence of
gestational diabetes increased from 4.9% in 1990 to 7.1% in 2000 in
California, where Asian and Hispanic women had higher incidences than whites
and African Americans (20).
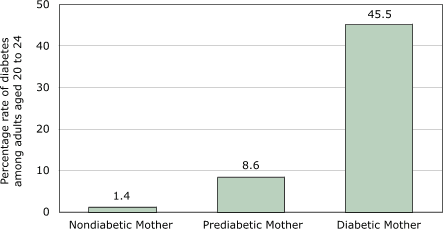
Figure 4.
Prevalence of type 2 diabetes among Pima Indian adults, Arizona, aged 20 to 24, by
diabetes status of the mother during pregnancy. A prediabetic mother is one who develops diabetes after
delivery. Data from Pettitt et al, 1988 (16).
(A text description of this graph is also
available.)
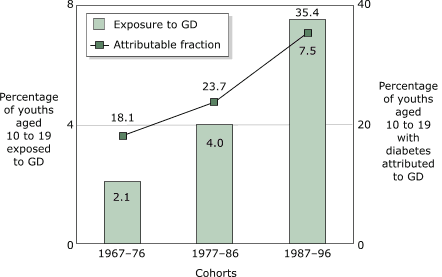
Figure 5. Exposure to gestational diabetes (GD) and
fraction of diabetes attributed to GD among cohorts of Pima
Indian adults, Arizona, aged 10 to 19 years (n = 6902). Data from Dabelea et al,
1998 (17).
(A text description of this graph is also
available.)
Gestational diabetes is adding fuel to an already raging epidemic of
diabetes. The intergenerational component operates through women and begins
with the interaction of genetic susceptibility and unhealthy lifestyle
practices that precipitate obesity in girls and women of reproductive age,
which in turn increases the risk of diabetes prior to or during pregnancy.
The percentage of women exposed to diabetes in their intrauterine life then
increases in each subsequent generation, driving rates of diabetes in the
general population higher and higher with each generation. This scenario is already
unfolding in the Mexican populations of North America and deserves serious
study.
Back to top
Where Do We Go From Here?
The costs of diabetes in the United States were estimated at $132 billion
for 2002 (21). Meeting the demand for public health care services caused by
diabetes will alone cost Mexico $318 million in 2005, 26% more than in 2003
(22). While the monetary costs are staggering, the suffering and disability
among those afflicted with diabetes and their families are incalculable.
We need to confront the diabetes pandemic with urgency.
Efficacy studies show that lifestyle changes can effectively reduce the
incidence of diabetes in persons at high risk (23). We need effective
programs that promote healthy lifestyles and make screening and sound case
management widely available. We also need to devote significant resources to
developing new drugs and therapies. Combating obesity and inactivity must
become a national priority. Preventive actions must be undertaken along a
broad front, impacting behavior as well as the physical environment — from how we design our cities to promote physical activity to what
agriculture and food policies we support to foster a healthier food basket.
We need to promote aggressively a love of physical activity and healthy
diets, particularly among our children. We need flexible programs that can
fit local settings and our diversity of cultures, including the mosaic of
Hispanic groups in the United States. Mexico, with far fewer resources, must do all of
the above while combating yesterday’s unresolved problems of undernutrition.
The future will be grim only if we let it become so.
Back to top
Author Information
Corresponding author: Reynaldo Martorell, PhD, Department of
Global Health, The Rollins School of Public Health of Emory
University, 1518 Clifton Rd, Room 754, Atlanta, GA 30322. Telephone:
404-727-9854. E-mail: rmart77@sph.emory.edu.
Back to top
References
- Cordain L, Eaton SB, Miller JB, Mann N, Hill K.
The paradoxical nature
of hunter-gatherer diets: meat-based, yet non-atherogenic. Eur J Clin Nutr
2002;56(Suppl 1):S42-S52.
- Martorell R, Stein AD. The emergence of diet-related chronic
diseases in developing countries. In: Bowman BA, Russel RM, editors.
Present knowledge in nutrition. 8th edition. Washington (DC): ILSI
Press; 2001. p. 665-685.
- Popkin BM.
The nutrition transition in low-income countries: an
emerging crisis. Nutr Rev 1994;52:285-98.
- Diamond J.
The double puzzle of diabetes.
Nature
2003 Jun;423(5):599-602.
- Hales CN, Barber DJP. The thrifty phenotype hypothesis. Br Med Bull
2001;60:5-20.
- Rivera DJ, Shamah LV, Villalpando HS, González de Cossío T, Hernández
PB, Sepúlveda J. Encuesta Nacional de Nutrición 1999. Estado nutricio de
niños y mujeres en México. Cuernavaca, Morelos, México: Instituto Nacional
de Salud Pública; 2001.
- Flegal KM, Carroll MD, Ogden CL, Johnson CL.
Prevalence and
trends in
obesity among US adults, 1999-2000. JAMA 2002;288:1723-7.
- Jiménez-Cruz A, Bacardí-Gascón M, Jones EG.
Consumption of
fruits,
vegetables, soft drinks, and high-fat-containing snacks among Mexican
children on the Mexico-U.S. border. Arch Med Res 2002;33:74-80.
- Haffner SM.
Obesity and the metabolic syndrome; the San Antonio Heart
Study. Br J Nutr 2000;83(Suppl 1):S67-S70.
- King H, Aubert RE, Herman WH.
Global
burden of diabetes, 1995-2025: prevalence, numerical estimates, and
projections.
Diabetes Care 1998;21(9):1414-31.
- Aguilar-Salinas CA, Velazquez Monroy O, Gómez-Pérez FJ, Gonzalez Chávez AG, Esqueda AL,
Molina Cuevas V, et al.
Characteristics of
patients with type 2 diabetes in México: results from a large
population-based nationwide survey.
Diabetes Care 2003;26(7):2021-6.
- Harris MI, Flegal KM, Cowie CC, Eberhardt MS, Goldstein DE, Little RR,
et al.
Prevalence of
diabetes, impaired fasting glucose, and impaired
glucose tolerance in U.S. adults. The third National Health and Nutrition Examination Survey, 1988-1994. Diabetes Care 1998;21(4):518-24.
-
Centers for Disease Control and Prevention. National
diabetes fact sheet, United States, 2003. Atlanta (GA): Centers for
Disease Control and Prevention; 2004.
- Narayan KM, Boyle JP, Thompson TJ, Sorensen SW, Williamson DF.
Lifetime
risk for diabetes mellitus in the United States. JAMA
2003;290(14):1884-90.
- Gardner LI, Stern MP, Haffner SM, Gaskill SP, Hazuda HP, Relethford JH,
et al. Prevalence of diabetes in Mexican Americans.
Relationship to
percent of gene pool derived from Native American sources. Diabetes
1984;33:86-92.
- Pettitt DJ, Aleck KA, Baird HR, Carraher MJ, Bennett PH, Knowler WC.
Congenital
susceptibility to NIDDM. Role of intrauterine environment.
Diabetes 1988;37:622-8.
- Dabelea D, Hanson RL, Bennett PH, Roumain J, Knowler WC, Pettitt DJ.
Increasing prevalence of
Type II diabetes in American Indian children. Diabetologia 1998;41:904-10.
- Dabelea D, Knowler WC, Pettitt DJ.
Effect of diabetes in pregnancy
on offspring: follow-up research in the Pima Indians. J Matern Fetal Med
2000;9(1):83-8.
- Dabelea D, Pettitt DJ.
Intrauterine
diabetic environment confers risks for type 2 diabetes mellitus and
obesity in the offspring, in addition to genetic susceptibility. J Pediatr Endocrinol Metab
2001;14(8):1085-91.
- Ferrara A, Kahn HS, Quesenberry CP, Riley C, Hedderson MM.
An
increase
in the incidence of gestational diabetes mellitus: Northern California,
1991-2000. Obstetrics & Gynecology 2004;103(3):526-33.
- Hogan P, Dall T, Nikolov P; American
Diabetes Association.
Economic
costs of diabetes in the US
in 2002. Diabetes Care 2003;26(3):917-32.
- Arredondo A, Zuniga A.
Economic
consequences of epidemiological
changes in diabetes in middle-income countries. The Mexican case. Diabetes
Care 2004;27(1):104-9.
- Knowler WC, Barrett-Connor E, Fowler
SE, Hamman RF, Lachin JM, Walker EA, et al; Diabetes Prevention Program Research Group.
Reduction in the
incidence
of type 2 diabetes with lifestyle intervention or metformin. N Engl J
Med 2002;346(6):393-403.
Back to top |
|
