 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Trichinellosis Surveillance --- United States, 1997--2001Sharon L. Roy, M.D. AbstractProblem/Condition: Trichinellosis is a parasitic disease caused by tissue-dwelling roundworms of the species Trichinella spiralis. The organism is acquired by eating Trichinella-infected meat products. The disease has variable clinical manifestations, ranging from asymptomatic to fatal. In the United States, trichinellosis has caused hundreds of preventable cases of illness and occasional deaths. The national trichinellosis surveillance system has documented a steady decline in the reported incidence of this disease, as well as a change in its epidemiology. Reporting Period Covered: This report summarizes surveillance data for trichinellosis in the United States for 1997--2001. Description of System: Trichinellosis became a nationally reportable disease in 1966, but statistics have been kept on the disease since 1947. The national trichinellosis surveillance system is a passive system that relies on existing resources at the local, state, and federal levels. Cases are diagnosed based on clinical history with laboratory confirmation. Cases are reported weekly to CDC through the National Electronic Telecommunications System for Surveillance (NETSS). Detailed data regarding signs and symptoms, diagnostic tests, and food consumption are gathered by using a supplementary standardized surveillance form and are reported to CDC by fax or mail. This information is compared with NETSS data several times a year by CDC staff. Discrepancies are reviewed with the state health departments. The purpose of the surveillance system is to determine the incidence of trichinellosis, to maintain awareness of the disease, to monitor epidemiologic changes, to identify outbreaks, to guide prevention efforts, and to measure the effectiveness of those efforts. Results: Although trichinellosis was associated historically with eating Trichinella-infected pork from domesticated sources, wild game meat was the most common source of infection during 1997--2001. During this 5-year period, 72 cases were reported to CDC. Of these, 31 (43%) cases were associated with eating wild game: 29 with bear meat, one with cougar meat, and one with wild boar meat. In comparison, only 12 (17%) cases were associated with eating commercial pork products, including four cases traced to a foreign source. Nine (13%) cases were associated with eating noncommercial pork from home-raised or direct-from-farm swine where U.S. commercial pork production industry standards and regulations do not apply. Interpretations: The majority of the decline in reported trichinellosis cases is a result of improved observance of standards and regulations in the U.S. commercial pork industry, which has altered animal husbandry practices resulting in reduced Trichinella prevalence among swine. Public Health Actions: Because of the change in epidemiology of trichinellosis and the continued occurrence of cases among consumers of wild game meat and noncommercial pork, more targeted public education is needed to further reduce the incidence of this disease. IntroductionTrichinellosis is a parasitic disease caused by a roundworm of the Trichinella genus that infects carnivorous and omnivorous animals, including domestic swine and wild game (e.g., bears, cougars, and wild boars) (1). Humans become infected when they eat meat cooked inadequately from these animals containing larval cysts of Trichinella spp. Trichinellosis has a broad clinical presentation, ranging from asymptomatic to fatal. Initial symptoms of abdominal pain, vomiting, diarrhea, constipation, and low-grade fever might occur within 1 week of ingestion of infected meat, as the larvae invade the bowel wall. These symptoms might be followed in approximately 1--8 weeks by facial edema, peripheral edema, headaches, intermittent fever, conjunctivitis, rashes, pruritus, cough, myalgia, and eosinophilia, as the larvae migrate through the tissues and encyst in the muscles (2,3). Fatal cases might result from neurologic and myocardial complications (3). In the United States, the national trichinellosis surveillance system has documented a steady decline in the reported incidence of this disease. During 1947--1951, a median of 393 cases (range: 327--487) was reported annually, including 57 trichinellosis-related deaths (4). During 1997--2001, the incidence decreased to a median of 12 cases annually (range: 11--23) and no reported deaths. No apparent change in the trichinellosis surveillance system can account for this decline. However, because the surveillance system is not designed to detect asymptomatic cases, the number of reported cases probably represents only a portion of the total number of infections. Historically, pork products were the most commonly identified source of Trichinella infection in the United States. However, the number of annual reported cases attributed to pork has declined for many years, largely because of changes implemented by the U.S. pork industry that have resulted in reduced prevalence of Trichinella among domestic swine. This report presents data for 1997--2001 on trichinellosis and describes the changing epidemiology of this disease. MethodsState health departments report new cases of trichinellosis by week to CDC through the National Electronic Telecommunications System for Surveillance (NETSS) and through trichinellosis surveillance case report forms (CDC 54.7 Rev. 02-90) containing additional epidemiologic information. Additional cases might be identified through laboratory testing for Trichinella performed at CDC or through investigations conducted by CDC. NETSS data are reviewed several times a year and matched to case report forms or laboratory reports. A confirmed case of trichinellosis is defined as one that is laboratory confirmed and is clinically compatible with the common signs and symptoms of trichinellosis among symptomatic persons. These signs and symptoms include eosinophilia, fever, myalgia, and periorbital edema. Laboratory confirmation is made either by identification of Trichinella larvae through muscle biopsy or by a positive serologic test. An outbreak of trichinellosis is defined as >2 cases, of which at least one must be laboratory confirmed. If associated patients share an epidemiologically implicated meal or meat product and have either positive serologic tests or clinically compatible illnesses, they should also be reported as confirmed (5). Cases reported by states through NETSS that were unaccompanied by case report forms or that did not meet the case definition for a confirmed case were excluded from this report. During 1997--2001, only two cases were unaccompanied by case report forms and excluded. For data analysis, commercial pork products were defined as pork purchased at supermarkets, butcher shops, or public eating places. This category includes pork products that were eaten or obtained in other countries. Noncommercial pork products were defined as pork purchased directly from a farm, obtained from home-raised swine, or obtained from wild boars (6). ResultsDuring 1997--2001, a total of 18 states reported 72 cases of trichinellosis to CDC, including nine outbreaks involving >2 persons. States in the Midwest and Mountain West accounted for 63 (88%) cases, of which 50 (69%) were reported from five states: California (21), Ohio (nine), Illinois (seven), Montana (seven), and Alaska (six) (Table 1). The nine common-source outbreaks occurred in these five states and accounted for 33 (46%) cases (Table 2). Of the 72 cases, 52 (72%) occurred among males. The median age of patients was 45 years (range: 3--81 years) (Figure 1). During 1997--2001, a total of 55 (76%) of the 72 patients had at least one common sign or symptom of trichinellosis; 46 (64%) had myalgia, 39 (54%) had eosinophilia, 33 (46%) had fever, and 22 (31%) had periorbital edema. A total of 40 (56%) patients reported more than one sign or symptom. Of the 17 patients who did not report signs or symptoms, one was a girl aged 3 years with a positive Trichinella antibody titre but no eosinophilia, fever, or periorbital edema; myalgia could not be assessed. The signs and symptoms of the remaining 16 patients were unknown. For the 24 patients for whom the dates of consumption of the implicated meat and the onset of symptoms were available, the median incubation period was 13 days (range: 1--50 days). Of the 72 patients, 54 (75%) had serologic testing performed; 50 (93%) were seropositive for Trichinella antibodies. Muscle biopsies were performed on four (6%) patients; all were positive. The source of infection was known or suspected for 57 (79%) patients. Pork products were associated with 22 (39%) cases: 12 with commercial pork, nine with home-raised or direct-from-farm swine, and one with a wild boar. Of the 12 cases associated with commercial pork, eight were linked to U.S. commercial pork, and four were linked to pork obtained in Egypt, Vietnam, and Yugoslavia. Nonpork products were associated with 30 (53%) cases: 29 with bear meat and one with cougar meat. The source of infection was unknown for 20 (35%) cases; these included five cases that were associated with both pork and nonpork products. Because a single type of meat could not be implicated, each patient was classified as having an unknown source of infection. None of the meats consumed by these five patients were examined for larvae (Table 3). During 1997--2001, implicated meat was examined for Trichinella larvae in 34 cases, which included cases from seven of the nine common-source outbreaks. Larvae were present in meat associated with 32 of the 34 cases, including all seven of the outbreaks for which meat was examined. These outbreaks accounted for 29 of the 32 cases. Five outbreaks were linked to larvae-positive bear meat (Montana 1997, five cases; Ohio 1998, eight cases; Alaska 2000, four cases; California 2001, two cases; and California 2001, two cases). Two outbreaks were linked to larvae-positive meat from home-raised swine (California 2001, six cases; California 2001, two cases). Three isolated cases also were associated with larvae-positive meat: two with bear and one with cougar. The method of cooking the meat was identified in 38 (53%) of 72 cases: uncooked, 20 cases; open fire roasted or barbequed, 14 cases; fried, 13 cases; stewed, six cases; baked, two cases; and microwaved, one case. Certain cases were associated with multiple cooking methods. The source of the meat was identified in 55 (76%) of 72 cases. Meat was purchased commercially in 12 cases: supermarket, nine cases; butcher shop, one case; restaurant, one case; and a county fair, one case. Meat was obtained noncommercially in 41 cases: directly from a farm or from a home-raised animal, nine cases (all pork); and hunted or trapped, 32 cases (29 bear meat, one cougar meat, one wild boar meat, and one moose/quail/pheasant meat). The meat was consumed in another country in four cases: two in Yugoslavia (pork) and one each in Egypt (pork) and Vietnam (pork). Of the nine trichinellosis outbreaks, five were associated with bear meat, two with home-raised swine, and two with commercial pork. In December 1997, an outbreak of five cases in Montana was associated with bear jerky. In October--December 1998, an outbreak of eight cases in Ohio was associated with meat from a bear killed in northern Ontario, Canada, in March 1998 and transported back to the United States. The meat was prepared as a roast, a spaghetti dish, and hamburgers and eaten on October 13, 1998, at a church barbeque. The hunter tested positive for Trichinella by muscle biopsy. Another member of the outbreak tested positive by enzyme immunoassay and bentonite flocculation antibody tests. The remaining six cases were linked epidemiologically through the church barbeque. In August--September 2000, an outbreak of four cases in Alaska was associated with steaks from a bear killed in Alaska in August 2000 and fried on a camp stove. In August 2001, an outbreak of two cases in California was associated with "medium-rare" barbequed meat from a bear killed in Alaska in June 2001. In November 2001, an outbreak of two cases in California was associated with uncooked bear meat. Of the two outbreaks associated with home-raised swine, one was associated with uncooked pork (California, May 2001, two cases), and the other was associated with a combination of fried, barbequed, stewed, and uncooked pork (California, May--June 2001, six cases). Of the two outbreaks associated with commercial pork, one was associated with homemade pork sausage and jerky (Illinois, March--May 1999, two cases), and the other was associated with sausage and smoked pork consumed in Yugoslavia (Illinois, January 2000, two cases). In previous years, seasonal peaks in trichinellosis in the United States were observed in December and January, corresponding to consumption of homemade pork sausage during the Christmas holidays (6). However, during 1997--2001, no such pattern was observed for patients with known dates of illness onset in which commercial pork products were the only implicated meats (Figure 2). For cases in which noncommercial pork products were the only implicated meats, seasonal peaks occurred in May and June and were associated with home-raised swine (Figure 3). For cases in which nonpork products were the only implicated meats, seasonal peaks occurred during August--September and November--December and were associated with bear meat (Figure 4). DiscussionThe reported incidence of trichinellosis has declined substantially since the U.S. Public Health Service began keeping records on the disease in 1947 (Figure 5). The decrease in cases has mirrored the decline in prevalence of Trichinella in commercial pork products. In 1900, the trichinellosis prevalence in U.S. swine was estimated to be 1.41%. The estimated prevalence decreased to 0.125% during 1966--1970 and to 0.013% in 1995 (7). This decline reflects changes in the U.S. pork industry that have reduced exposure of domestic swine to Trichinella. Historically, the major sources of Trichinella-infected pork were swine fed garbage containing animal waste products. Trichinellosis prevalence among swine decreased after garbage cooking laws were passed to control vesicular exanthema (1953--1954) and hog cholera (1962) (8,9). In 1980, Congress passed the Federal Swine Health Protection Act (Public Law 96-468) that prohibits feeding potentially contaminated garbage to swine. Other risk factors for Trichinella-infected pork include consumption by swine of small mammals infected with Trichinella, including rodents, raccoons, skunks, and opossums, and cannibalism among swine within an infected herd (10). These risk factors are being addressed through industry-wide emphasis on 1) maintaining effective rodent-control programs; 2) controlling access of wildlife and pets to swine, facilities, and feed stuffs; 3) maintaining good hygiene in facilities; and 4) removing dead swine immediately when they are found (10,11). The United States Department of Agriculture (USDA), the National Pork Producers Council, and the pork processing industry have developed the Trichinae Herd Certification Program. This is a voluntary pre-harvest pork safety program that provides documentation of swine management practices to minimize Trichinella exposure. The goal of the program is to establish a system under which pork production facilities that follow good production practices might be certified as Trichinella-safe (12). In addition to the reduction in Trichinella prevalence in commercial pork, processing methods also have contributed to the dramatic decline in human trichinellosis associated with pork products. Through the U.S. Code of Federal Regulations, USDA has stipulated specific cooking temperatures and times, freezing temperatures and times, and curing methods for processed pork products to control post-harvest human exposure to Trichinella (13). Increased public awareness regarding safe cooking and freezing of fresh pork also has reduced post-harvest exposure. Although the number of reported human trichinellosis cases related to eating pork has decreased, since 1982, the number of reported cases related to eating nonpork products has remained constant. During 1997--2001, the number of cases associated with eating nonpork products surpassed that of pork for the first time in the United States (Figure 6). Home-raised and direct-from-farm swine and pork from foreign sources are unaffected by regulations and standards followed by the U.S. commercial pork production industry that have helped to reduce Trichinella prevalence in commercial pork. Outbreaks of trichinellosis associated with noncommercial sources of pork have been reported previously (14,15) and continue to be reported. Trichinellosis cases associated with noncommercial pork now outnumber those cases associated with U.S. commercial pork, reflecting a changing risk pattern in pork consumption. The emergence of wild game meat as the most common source of trichinellosis and the continued occurrence of trichinellosis among consumers of pork obtained from small farms or other countries suggest that educational messages concerning the risks for eating meat cooked improperly, especially from noncommercial sources, are not reaching persons at risk for trichinellosis. USDA recommends that consumers of fresh pork cook the meat to an internal temperature of 160ºF (71ºC) (16). T. spiralis larvae in pork are killed at lower temperatures (e.g., 140ºF [60ºC] for 2 minutes or 131ºF [55ºC]) for 6 minutes (17); however, USDA has recommended a higher temperature to allow for different cooking methods that might result in uneven temperature distributions throughout the meat (e.g., microwave cooking) (13). Escherichia coli, Salmonella, Staphylococcus aureus, and Listeria monocytogenes, other foodborne microorganisms that also can be found in pork, are destroyed by thorough cooking to an internal temperature of 160ºF (71ºC) (16). Freezing kills T. spiralis larvae in pork. Pork <6 inches thick can be made safe if frozen to -20ºF (-29ºC) for 6 days, -10ºF (-23ºC) for 10 days, or 5ºF (-17ºC) for 20 days (18). However, freezing might not kill other species and types of Trichinella found in wild game. Infective Trichinella spp. larvae have been found in frozen bear meat: grizzly bear meat frozen at -4º--20.3ºF (-20º-- -6.5ºC) for 27 months (19) and polar bear meat frozen at -0.4ºF (-18ºC) for approximately 24 months (20,21). Infective Trichinella spp. larvae also have been found in other wild carnivore meat frozen at 5ºF (-15ºC): marten frozen for 5 months, wolverine frozen for 6 months, and arctic fox frozen for 14 months (22). In addition, viable Trichinella spp. larvae were found in black bear meat that had been processed into ham and jerky by dry curing with a commercial salt mixture at a USDA-licensed establishment by using procedures similar to those used to prepare pork for human consumption. However, no viable Trichinella spp. larvae were found in ground bear meat preparations (e.g., sausage, pepperoni, or salami) that had been processed according to standards mandated by USDA for processing pork (19). The dramatic decline of trichinellosis in the United States reflects changes in industrial practices, increased government regulations, and increased public awareness. As a result, the epidemiology of the disease has changed. Because of the successful reduction in Trichinella prevalence among swine in the U.S. commercial pork industry, the majority of cases of human trichinellosis are now associated with wild game meat, noncommercial pork, and foreign pork. Persons at risk need a greater understanding of the changing risks for trichinellosis, of the methods for safe meat preparation, and of the limitations of those methods in certain circumstances, if further reduction in the incidence of this disease is to be achieved in the United States. References
Table 1 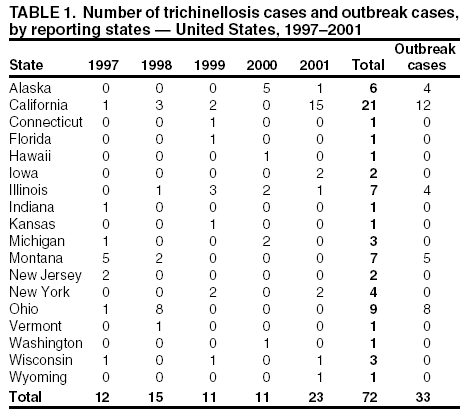 Return to top. Figure 1 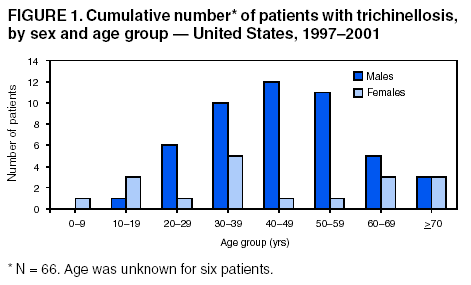 Return to top. Table 2  Return to top. Figure 2 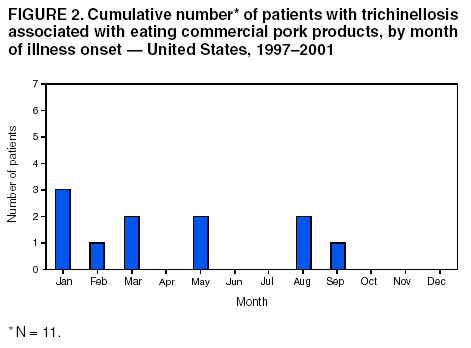 Return to top. Table 3 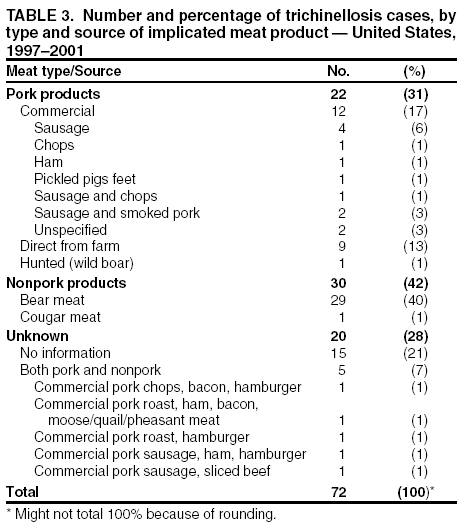 Return to top. Figure 3 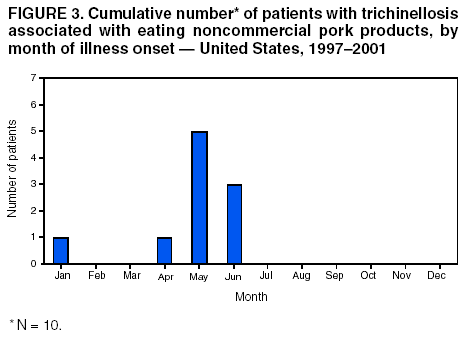 Return to top. Figure 4 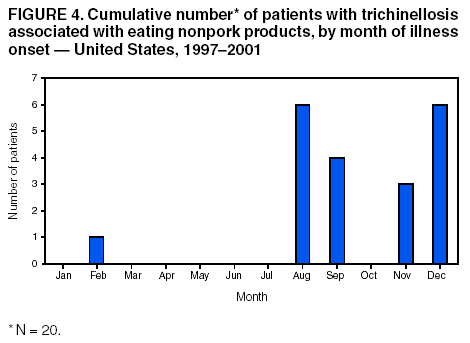 Return to top. Figure 5 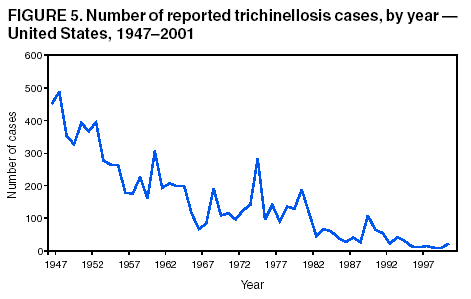 Return to top. Figure 6 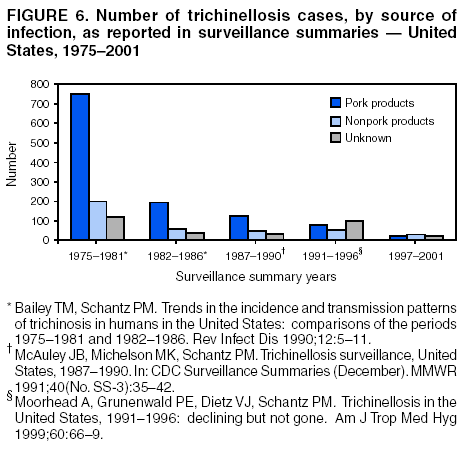 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 7/8/2003 |
|||||||||
This page last reviewed 7/8/2003
|