 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Outbreak of Multidrug-Resistant Salmonella Typhimurium Associated with Rodents Purchased at Retail Pet Stores --- United States, December 2003--October 2004During 2004, the Minnesota Department of Health (MDH) Public Health Laboratory notified CDC about the isolation of multidrug-resistant Salmonella enterica serotype Typhimurium from ill hamsters from a Minnesota pet distributor. This report describes two of the first identified human cases associated with this outbreak, summarizes the multistate investigation of human S. Typhimurium infections associated with exposure to rodents (e.g., hamsters, mice, and rats) purchased at pet stores, and highlights methods for reducing Salmonella transmission from pet rodents to their owners. This is the first documented salmonellosis outbreak associated with pet rodents. Findings demonstrate that the handling of pet rodents is a potential health risk, especially for children. Public health practitioners should consider pet rodents a potential source of salmonellosis. Case ReportsSouth Carolina. During June 2004, a boy aged 4 years was hospitalized for 5 days with fever (105ºF [40.6ºC]), watery diarrhea, and abdominal cramping. A stool culture yielded S. Typhimurium. Nine days before the boy's illness, his family had purchased a hamster from a retail pet store supplied by an Arkansas distributor; the hamster was found dead 2 days after purchase. Minnesota. During August 2004, a boy aged 5 years had diarrhea of 14 days' duration (initially bloody), abdominal cramps, vomiting, and fever (103ºF [39.4ºC]). A stool culture yielded S. Typhimurium. Four days before the boy became ill, his family had purchased a mouse from a retail pet store supplied by a Minnesota distributor. The mouse became lethargic and had diarrhea immediately after purchase. Even though the mouse was ill, the boy frequently handled and kissed the mouse. One week after purchase, the mouse died; the mouse was frozen and later submitted for testing at MDH. Cultures of the mouse's lungs, pooled liver and spleen, and intestines yielded growth of S. Typhimurium, with a pulsed-field gel electrophoresis (PFGE) pattern indistinguishable from the boy's isolate. Hamster SalmonellosisOn August 30, a veterinarian for the Minnesota pet distributor called MDH about isolation of Salmonella from two ill hamsters submitted to the University of Minnesota Veterinary Diagnostic Laboratory (UMVDL). The hamsters were part of a shipment of 780 received on August 1 from an Iowa pet distributor (Figure). A total of 243 hamsters from this shipment were subsequently sent from the Minnesota distributor to 15 retail pet stores in four midwestern states. Distribution of rodents from the Minnesota distributor ceased on August 23 after numerous hamster deaths. Diarrhea was present in the majority of ill hamsters. By August 29, approximately 320 (60%) of the remaining 537 hamsters at the Minnesota distributor had died; the other hamsters were euthanized. UMVDL cultured S. Typhimurium from the internal organs of seven hamsters submitted for necropsy from the affected shipment. Isolates from these hamsters were submitted to MDH for testing; they were indistinguishable by PFGE from each other and from the Minnesota patient isolate that was submitted to MDH through routine surveillance. A subsequent MDH query of the PulseNet National Salmonella Database revealed that these S. Typhimurium isolates were indistinguishable by PFGE from a Kentucky patient's isolate and from the South Carolina patient isolate. Historical data from the database confirmed that this PFGE pattern was uncommon, representing 23 of 17,737 isolates in the S. Typhimurium database since 1998, providing additional evidence that this cluster potentially represented a common source outbreak. Epidemiologic InvestigationBased on recognition of the Minnesota and South Carolina human cases, CDC and MDH conducted a national search for additional human cases associated with exposure to rodents. A review of isolates submitted to the PulseNet National Salmonella Database in 2004 revealed 28 matching human case-isolates of S. Typhimurium from 19 states; patient illness onset dates ranged from December 2003 to October 2004. Of 22 patients interviewed, 13 (59%) had been exposed to rodents purchased from retail pet stores (Figure); all exposures occurred during the 8 days before illness onset. Two (9%) patients acquired salmonellosis through secondary exposure. Seven (32%) of the 22 patients had no identified rodent exposure. Four patients remained under investigation, and two were lost to follow-up. The 15 patients with primary or secondary rodent exposure were from Illinois, Kentucky, Missouri, Pennsylvania, and South Carolina (two cases each), Georgia, Michigan, Minnesota, New Jersey, and North Carolina (one case each) (Figure). Dates of illness onset ranged from December 23, 2003, to September 28, 2004. The median age of patients was 16 years (range: 0--43 years), and seven (47%) were aged <7 years. Symptoms reported by patients with confirmed rodent exposure included abdominal cramping (77%), fever (67%), vomiting (53%), and bloody diarrhea (20%). Six (40%) patients were hospitalized; no deaths occurred. Rodent exposures of primary patients included mice/rats purchased to feed snakes (seven cases), pet mice/rats (four), and pet hamsters (two). Human and animal S. Typhimurium isolates were uniformly resistant to ampicillin, chloramphenicol, streptomycin, sulfizoxazole, and tetracycline (R-type ACSSuT). Phage typing was performed on three human isolates at CDC; all were determined to be definitive phage type (DT) 120. Animal Traceback Investigations and Environmental TestingCDC, state and local health departments, and the U.S. Department of Agriculture Animal Care conducted traceback investigations of rodents from patients with confirmed rodent exposure and identified retail pet stores, rodent distributors, and breeders (Figure). Rodents purchased at retail pet stores were most frequently traced to a pet distributor in Georgia or Arkansas. In October 2004, a retail pet store in Illinois identified ill hamsters traced back to shipments originating from the Iowa distributor; S. Typhimurium was isolated from 16 of 22 necropsied hamsters. Seven isolates were submitted to the Illinois Department of Public Health Division of Laboratories for PFGE testing; all were indistinguishable from previous S. Typhimurium isolates obtained from hamsters received in Minnesota from the Iowa distributor (Figure). In November 2004, S. Typhimurium was isolated at the Georgia distributor from environmental cultures of rodent transport cages, rat bins, mice pellets/bedding, and rat pellets. Three of the four isolates were indistinguishable from each other by PFGE and closely related to the outbreak PFGE pattern. The Georgia Public Health Laboratory performed susceptibility testing of isolates from the Georgia distributor; tests indicated multidrug-resistance (R-type ACSSuT). Systematic environmental cultures were not obtained at the implicated Arkansas and Iowa pet distributors or other breeders. Information on use of antimicrobials (e.g., spectinomycin, leptomycin, tetracycline, and nitrofurazone) was obtained from five rodent breeders/distributors. Routine use of antimicrobials was documented in four facilities that use them for prevention of nonspecific rodent enteritis. Delivery of antimicrobial agents in drinking water occurred at the time of rodent weaning, before transport, and/or on arrival at the pet distributor. One pet distributor used rodent feed containing tetracycline for all rodent feedings. No common link was identified among the three main implicated pet distributors (Arkansas, Georgia, and Iowa); the source of infected rodents for this multistate outbreak is unknown. A systematic review of shipping and commercial records was not possible at many of the potentially involved rodent breeders and distributors. Reported by: K Smith, DVM, D Boxrud, MSc, F Leano, MS, Minnesota Dept of Health; C Snider, MHS, C Braden, MD, J Lockett, S Montgomery, DVM, Div of Bacterial and Mycotic Diseases, National Center for Infectious Diseases; S Swanson, MD, C O'Reilly, PhD, EIS officers, CDC. Editorial Note:Each year, an estimated 1.4 million persons in the United States have salmonellosis, leading to approximately 14,800 hospitalizations and 415 deaths (1,2). Salmonella is found in the intestinal tract of animals and is transmitted by ingestion of feces, which might occur from eating contaminated foods or through contact with animals or their environments. Exposure to animals with higher frequency of Salmonella shedding in their feces increases the risk for acquiring salmonellosis; among pets, these include reptiles, young animals, and animals with diarrhea (3,4). In addition to reptiles, salmonellosis outbreaks have been reported after handling of pet chicks, ducklings, kittens, and hedgehogs (5--7). Cases described in this outbreak were dispersed temporally and geographically, and rodent purchases occurred through multiple retail pet store chains and pet distributors; these factors might reflect the geographic spread of S. Typhimurium from a common source of infection occurring earlier in the chain of pet distributors or breeders. The recovery of S. Typhimurium from reusable transport containers, cages, and bins contaminated with rodent droppings offers a potential mechanism for both the environmental persistence and geographic spread of Salmonella. Rodents subsequently transported or housed in contaminated containers might have been exposed to Salmonella and become infected without direct contact with infected rodents. Phage typing of the S. Typhimurium isolates indicated DT120. S. Typhimurium DT120 is known to be multidrug-resistant, possessing antibiotic-resistance gene cassettes found also in S. Typhimurium DT104 (8). Thus, identifying pet rodents as an additional reservoir of multidrug-resistant S. Typhimurium is of public health importance. Antimicrobial agents are ineffective at preventing Salmonella shedding and likely prolong such shedding. The dissemination of multidrug-resistant Salmonella in pet rodents might have been facilitated by the use of prophylactic antimicrobials within the pocket-pet (e.g., hamsters, mice, and rats) animal industry. This use might have contributed to disease in colonized animals and increased shedding of Salmonella, thus facilitating increased transmission among animals and from animals to their human caretakers. Public health practitioners should consider pet rodents a potential source of salmonellosis and, when indicated, should obtain cultures from pet rodents during an investigation. Veterinarians, animal breeders, and distributors should consider submitting specimens to clinical laboratories for Salmonella isolation if substantial diarrhea-associated morbidity or mortality occurs among pet rodents intended for sale. Heightened infection-control practices by pet stores and distributors, including routine sanitizing of animal transport containers and cages, might reduce transmission. Preventive strategies (e.g., appropriate animal husbandry and hygiene practices) could reduce the need for nontherapeutic antimicrobials to prevent disease in rodents (9). Consumers and animal workers should be aware that rodents, like reptiles, can shed Salmonella; therefore, they should expect rodent feces to be potentially infectious. Salmonella transmission to humans can be reduced by thoroughly washing hands with soap and water after handling rodents or their cages or bedding. Young children who are unable to reliably wash their hands should avoid contact with rodent feces. Additional public health recommendations for preventing salmonellosis from reptiles might also be appropriate for preventing salmonellosis from pet rodents (3). References
Acknowledgments The findings of this report are based, in part, on contributions by A McKelvey, MSPH, Arkansas Dept of Health. C Shuler, DVM, C Burnett, MPH, Georgia Div of Public Health. M Auslander, DVM, Kentucky Dept for Public Health. C Austin, DVM, Illinois Dept of Public Health. S Brockus, DVM, Iowa Dept of Public Health. S Bidol, MPH, Michigan Dept of Community Health. A Covington, DVM; A Wünschmann, DVM, Univ of Minnesota College of Veterinary Medicine, Dept of Veterinary Diagnostic Medicine, St. Paul; H Hull, MD, State Epidemiologist, Minnesota Dept of Health. JA Rudroff, K Walker, Missouri Dept of Health and Senior Svcs. F Sorhage, VMD, New Jersey Dept of Health and Senior Svcs. M Deasy, Pennsylvania Dept of Health. MA Wenck, DVM, South Carolina Dept of Public Health. R Willems, DVM, Eastern Region, Animal Care, US Dept of Agriculture. S Van Duyne, MA, E Lyszkowicz, MSc, Div of Bacterial and Mycotic Diseases, National Center for Infectious Diseases, CDC.
Figure 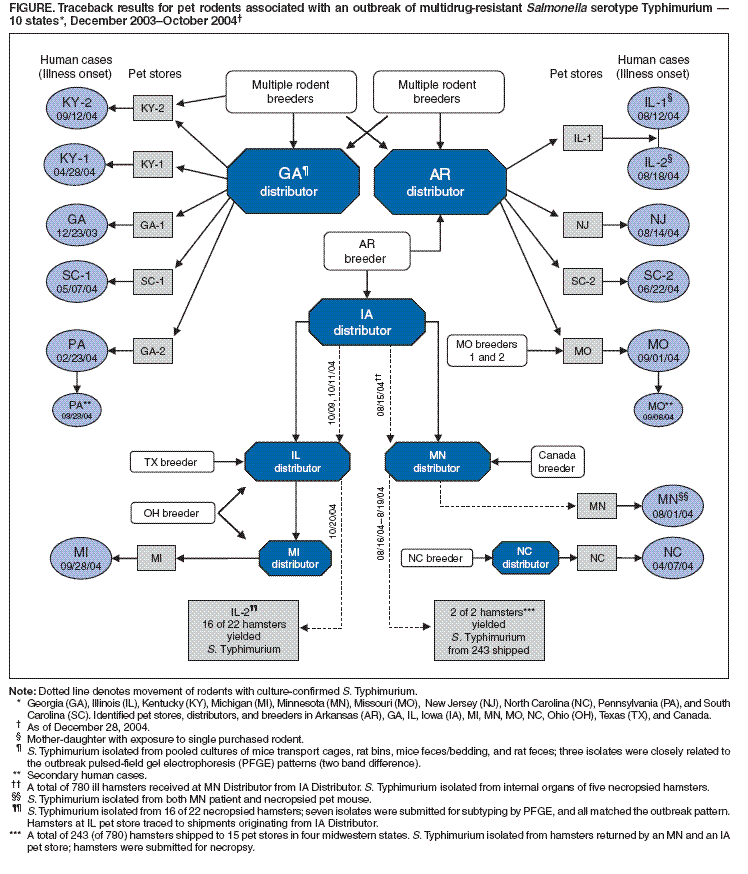 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 5/5/2005 |
|||||||||
|