 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Legionnaires Disease Associated with Potable Water in a Hotel --- Ocean City, Maryland, October 2003--February 2004During October 2003--February 2004, eight cases (seven confirmed cases and one possible) of Legionnaires disease (LD) were identified among guests at a hotel in Ocean City, Maryland. This report summarizes the subsequent investigation conducted by the Worcester County Health Department (WCHD), Maryland Department of Health and Mental Hygiene (DHMH), and CDC, which implicated the potable hot water system of the hotel as the most likely source of infection. The detection of this outbreak underscores the importance of enhanced, state-based surveillance for timely detection of travel-associated LD and implementation of control measures. On December 1, 2003, a local health department (LHD) notified DHMH of two LD cases in Maryland residents who had stayed at hotel A during the 2--10-day incubation period. The two patients had stays in hotel A of 3 and 4 days; their onsets of illness occurred 8 and 5 days, respectively, after leaving hotel A. Both patients had radiographically confirmed pneumonia and positive Legionella urinary antigen tests that were consistent with L. pneumophila serogroup 1 (Lp1) infection. The two patients had stayed at hotel A within 1 day of each other and were linked epidemiologically through travel information collected by LHDs in Maryland by using the DHMH report form for LD. This form collects information regarding location, accommodations, and dates of travel for the 10 days preceding illness. Review of LD case report forms revealed six additional LD patients with reported travel to Ocean City during the preceding year; however, none had stayed at hotel A. After environmental inspections and water sampling of hotel A by WCHD, multiple samples from multiple sites in the hotel revealed the presence of Lp1. On January 26, 2004, hotel A attempted remediation by superheating water systems, flushing all water taps, and hyperchlorinating the cooling tower. Showers and faucets were reportedly disinfected, and shower heads and sink aerators were replaced in rooms where patients had stayed. Case FindingsAfter the initial cases were identified, enhanced surveillance was conducted, including postings on the CDC Epidemic Information Exchange (Epi-X) and a rapid review of all DHMH case report forms for LD. In February 2004, two additional LD patients were identified, including one person who had stayed at hotel A after remediation. On the basis of this finding and the potential for ongoing but undetected transmission of Legionella, CDC was invited to join the investigation. To identify additional cases, neighboring jurisdictions, acute care hospital emergency departments, and all LHDs in Maryland were notified. Press releases and hotel A guest notifications were issued by DHMH, WCHD, and hotel A. Reports of persons with illness after a visit to Ocean City were reviewed by WCHD and DHMH to determine whether criteria for the LD case definition were met. A confirmed case of LD was defined as radiographically confirmed pneumonia with laboratory evidence of Legionella infection in a resident or visitor to Ocean City during October 2003--February 2004, whose illness began within 10 days of time spent in Ocean City. Laboratory confirmation included identification of Legionella by culture, direct fluorescent antibody testing, urine antigen assay, or an increase in antibody titer indicating recent infection. Possible LD cases were defined similarly but without laboratory confirmation of Legionella infection or other infectious etiology. Enhanced surveillance identified approximately 50 ill persons with exposure to hotel A. Further investigation resulted in identification of three additional confirmed cases and one possible case, for overall totals of seven confirmed and one possible case of LD during October 2003--February 2004 (Figure). The median length of stay at hotel A was 3 nights (range: 1--4 nights). Symptom onset occurred a median of 7.5 days (range: 4--9 days) after leaving hotel A. The median age of the eight patients was 63 years (range: 37--70 years), and six (75%) patients were men. Underlying medical conditions associated with increased risk for LD included smoking (five patients), diabetes (four patients), and an immunocompromised condition (one patient). Five cases were confirmed by urine antigen testing and two by serology. Seven patients were hospitalized; none died (Table). A review of possible exposures at hotel A among the patients with confirmed LD revealed that all had showered or bathed in their respective rooms, and one had used the whirlpool spa. Six patients reported exposure to the swimming pool and whirlpool area. No other common sources of exposure linking all cases were identified. Environmental InvestigationDuring December 2003--February 2004, WCHD, DHMH, and CDC conducted three environmental inspections and four rounds of water testing at hotel A. The hotel remained open during the inspections and testing. The rooms in which the seven confirmed patients stayed were located in different areas and on different floors of the hotel. During all rounds of testing, water temperatures in multiple locations were in an ideal range for growth and amplification of Legionella (77ºF--108ºF [25ºC--42ºC]). Lp1 was recovered from multiple sites in hotel A, including the hot water storage tank; cooling tower; multiple hot water heaters; and showers and faucets in rooms occupied by patients and well guests. All environmental Lp1 isolates were the same monoclonal antibody type 1,2,5,* (testing for type 6 was not conducted). Despite isolation of Lp1 from sites in hotel A, cultured isolates from patients were not available to link with environmental isolates through use of monoclonal antibody testing. After the third and fourth cases of LD were identified, a second superheating remediation was conducted at hotel A in February 2004. In addition, shower necks and faucets in all hotel rooms and condominiums were reportedly disinfected with a bleach solution. The whirlpool spa sand filter was cleaned. In March 2004, given the apparent inadequacy of the initial remediation, the potable water system was hyperchlorinated, and a postremediation plan for water testing for Legionella was instituted. Since the hyperchlorination treatment, no further cases of LD associated with hotel A have been identified. During postremediation follow-up testing, one Lp1 isolate from the cooling tower was identified at a low level, and the cooling tower was hyperchlorinated. DHMH continues to monitor for additional cases associated with hotel A and for all travel-associated LD cases. Reported by: D Goeller, MS, Worcester County Health Dept, Snow Hill; D Blythe, MD, M Davenport, MD, M Blackburn, MPH, Maryland Dept of Health and Mental Hygiene. B Flannery, PhD, C Lucas, PhD, B Fields, PhD, M Moore, MD, Div of Bacterial and Mycotic Diseases, National Center for Infectious Diseases; AD Castel, MD, L Hicks, DO, EIS officers, CDC. Editorial Note:Hotels have been common locations for LD outbreaks since the disease was first recognized among hotel guests in Philadelphia in 1976 (1,2). In this report, the exposure of patients to the hotel's potable water system, the lack of other epidemiologic links, and the recovery of Legionellae from multiple points in the system suggest that the hotel potable water system was the source of the outbreak. Approximately 8 million visitors travel to Ocean City each year; therefore, a link between the first two cases was not immediately evident. Available data were searched to identify additional cases associated with the hotel or travel to Ocean City. Active surveillance activities led to more rapid identification of other cases. The retrospective identification of these cases prompted further investigation and subsequent control and remediation efforts at hotel A. In 2003, DHMH began conducting enhanced surveillance because of increased reports of LD. All patients reported to DHMH are administered a follow-up questionnaire by local or state health departments. The questionnaire identifies travel that preceded the illness, including location, accommodations, dates, and information about exposures to common sources for infection, such as whirlpool spas and cooling towers. Surveillance data submitted to CDC indicate that approximately 21% of LD cases each year are travel associated (3). However, several factors hinder identification of travel-associated clusters of the disease. The LD incubation period is long enough for persons to disperse from the point source of infection. In addition, LD can be treated successfully with empiric antibiotics, which obviates the need for confirmatory testing. When diagnostic testing is performed, isolation of the organism is rare, preventing comparison of environmental isolates with clinical isolates. Improved national surveillance for travel-associated LD might help detect clusters of the disease. Surveillance for LD in the United States consists of two systems, a national, paper-based system and an electronic system reported through the National Electronic Telecommunications System for Surveillance. Only the paper case-report form collects information on location of travel and lodging. Although the paper case-report form is useful for tracking overall trends, a lack of timeliness and sensitivity, often resulting in an inability to link cases, limits its usefulness in identifying clusters (4). The European Working Group for Legionella Infections, established in 1986, has developed a successful surveillance system for identifying clusters of travel-associated LD. The European Surveillance Scheme for Travel-Associated Legionnaires Disease, which consists of 36 collaborating countries, compiles case data electronically and cross-checks travel accommodations with other cases to identify clusters. During 2000--2002, a total of 113 travel-associated LD clusters were reported, with the majority linked to hotels. Since introduction of the European group's guidelines in July 2002, all LD clusters are investigated, and remediation and control measures are instituted when necessary (5,6). The European and DHMH programs demonstrate how timely, sensitive surveillance can identify clusters of travel-associated LD. Prompt recognition and investigation of clusters can implicate a point source for infection and guide remediation and control efforts. Recognizing the benefits of enhanced surveillance, CDC plans to work with state health departments on new strategies to improve surveillance for travel-associated LD at the national, state, and local levels. Acknowledgments The findings in this report are based, in part, on contributions by R Thompson, P Dietrich, Baltimore County Health Dept, Baltimore; R Shockley, D Stevens, E Potetz, K Malloy, T Possident, Worcester County Health Dept, Snow Hill, Maryland. J Li, Office of Workforce and Career Development, CDC. References
Table 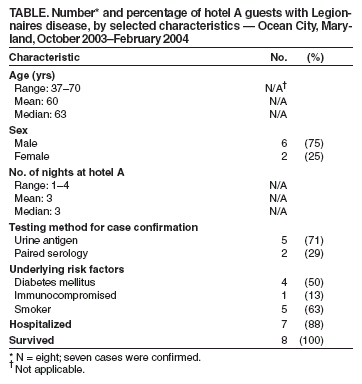 Return to top. Figure 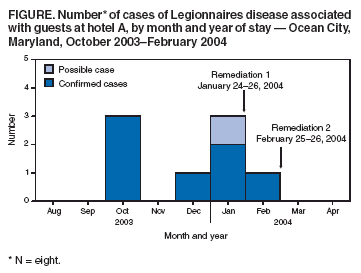 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 2/24/2005 |
|||||||||
This page last reviewed 2/24/2005
|