 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. State Estimates of Neonatal Health-Care Costs Associated with Maternal Smoking --- United States, 1996Smoking during pregnancy can cause poor outcomes for both the pregnant woman and her unborn child (1) and also result in added health-care expenditures. To characterize costs by state, CDC analyzed pregnancy risk surveillance and birth certificate data to estimate the association between maternal smoking and the probability of infant admission to a neonatal intensive care unit (NICU). Neonatal health-care costs, in 1996 dollars, were assigned on the basis of data from private health insurance claims. This report summarizes the results of that analysis, which estimated smoking-attributable neonatal expenditures (SAEs) of $366 million in the United States in 1996, or $704 per maternal smoker (2), and indicated wide variations in SAEs among states. These costs are preventable. States can use these data to justify or support their prevention and cessation treatment strategies. CDC has incorporated this analysis into its Maternal and Child Health Smoking-Attributable Mortality, Morbidity, and Economic Costs (MCH SAMMEC) software, which generates estimates of the smoking-attributable fraction of neonatal expenditures and provides area-specific estimates of SAEs for all women giving birth and for selected subsets of that population. Costs for 1996 (the most recent data available when MCH SAMMEC was developed) for each area were estimated by mother's age, race, education level, health insurance status, and timing of initiation of prenatal care. Separate estimates for populations of Hispanic ethnicity were not provided; an update of MCH SAMMEC will allow for selection of Hispanic and other racial/ethnic populations. MCH SAMMEC used national and state-specific maternal smoking prevalence estimates for 1997 by using birth certificate data collected by the National Center for Health Statistics from all areas except California, Indiana, New York state (outside of New York City), and South Dakota. Mothers were asked whether they smoked during their pregnancy and, if they did, how many cigarettes daily. For California, MCH SAMMEC used Maternal and Infant Health Assessment data. For Indiana, New York state, and South Dakota, smoking prevalence data for women aged 18--44 years were obtained from the Behavioral Risk Factor Surveillance System. Data in MCH SAMMEC are derived by extrapolating the results of a multivariate model estimated on 1995 Pregnancy Risk Assessment Monitoring System (PRAMS) data for 13 states* to all 50 states. This model estimates the association between maternal smoking and the probability of admission to an NICU and infant length of stay, whether in an NICU or regular nursery bed. These measures of resource utilization (i.e., NICU admission and infant nights in hospital) were assigned dollar costs based on a 1996 private-sector claims database. Details of the MCH SAMMEC methodology and estimation procedure have been published previously (3). The MCH SAMMEC software provided a national estimate of smoking-attributable neonatal expenditures of $366 million in 1996, or $704 per maternal smoker (2). Across areas, higher total SAE estimates by MCH SAMMEC were associated with higher numbers of births and higher smoking prevalence. SAE totals ranged from $34 million in California to $537,661 in the District of Columbia (DC). After California, states with the highest SAEs were New York, Ohio, Pennsylvania, and Texas; states with the lowest SAEs were Alaska, Hawaii, Vermont, and Wyoming (Table 1). Although overall prevalence of maternal smoking was 13.2% in 1997, prevalence by area ranged from 4.8% in New York City to 25.3% in West Virginia (4). To focus on the population at risk for excess costs in and across areas, SAEs per maternal smoker were estimated. SAEs per maternal smoker ranged from $519 in Hawaii to $1,334 in DC (Table 1). Although higher smoking prevalence usually is associated with white mothers as compared with blacks, smoking prevalence was higher among black mothers in 13 states and DC. Among black mothers in these 13 states, smoking prevalence ranged from 6.7% to 21.3%, and SAE per maternal smoker ranged from $1,008 to $1,403. Among all states, the average SAE for black mothers was almost double that for white mothers ($1,207 versus $651). However, adverse outcomes among black infants are more likely regardless of maternal smoking; at least one study suggests stronger effects of smoking on birthweight among black women compared with white women (1). In every area except DC, Indiana, and Mississippi, smoking prevalence was higher for women aged <20 years than for women aged >20 years. In all areas, the prevalence of smoking among pregnant women with less education was more than double the prevalence of women with more education. Persons who were uninsured or who were on Medicaid while pregnant had greater smoking prevalence than those with private or other health insurance. In all areas except DC and Texas, the prevalence of smoking among Medicaid/uninsured mothers was >10%; prevalence was highest in Indiana (37.7%) (Table 2). The highest SAE per maternal smoker among the Medicaid/uninsured group was in DC ($1,355); the lowest was in Hawaii ($523). Among women in the private/other insurance group, smoking prevalence was <10% in 28 states. The average SAEs per maternal smoker, from all areas, were $753 for those in the Medicaid/uninsured group and $626 for those in the private/other insurance group. Mothers who are on Medicaid or uninsured are less likely to initiate prenatal care in the first trimester (5). Both the prevalence of maternal smoking and SAE per maternal smoker were higher for mothers who began prenatal care in the third trimester or who had no prenatal care, compared with mothers who received prenatal care in the first or second trimester. The average SAEs per maternal smoker ranged from $485 in Hawaii to $1,112 in DC for mothers beginning care in the first or second trimester and from $821 to $2,166 in the same two areas for women beginning prenatal care in the third trimester, or having no care at all. Reported by: CL Melvin, PhD, Univ of North Carolina-Chapel Hill and the Smoke-Free Families National Dissemination Office. EK Adams, PhD, Rollins School of Public Health, Emory Univ, Atlanta, GA; MF Ayadi, PhD, CC Rivera, Div of Reproductive Health, National Center for Chronic Disease Prevention and Health Promotion, CDC. Editorial Note:In 2001, the estimated prevalence of smoking during pregnancy for all U.S. women was 11.4%, ranging from 3.9% in DC to 26.2% in West Virginia (6,7). Maternal smoking prevalences were higher among women who were younger, white, had less education and lower incomes, and were either on Medicaid or had no insurance (6,8). However, factors other than maternal smoking can result in higher neonatal costs among certain populations. For example, mothers with late or no prenatal care might be more likely to engage in other risk behaviors that affect pregnancy outcomes and increase costs (9). In addition, black infants have a greater likelihood for lower birthweight and adverse outcomes, regardless of maternal smoking (1). The findings in this report are subject to at least three limitations. First, SAEs do not include neonatal costs incurred after the infant's initial hospital stay or any costs associated with secondhand smoke. Second, although PRAMS data include measures of resource utilization necessary to estimate costs, PRAMS data were not available for all areas. Thus, the models that used sociodemographic characteristics of pregnant women found in both PRAMS and birth certificate data were used to extrapolate SAEs to each area. Finally, SAEs in this report are in 1996 dollars (the most recent available when MCH SAMMEC was developed) and do not reflect cost-of-living variations across states or subsequent increases in costs. State-specific estimates of SAEs per maternal smoker can help states explore potential cost savings from smoking-cessation interventions and other policies (e.g., excise taxes) that can reduce smoking prevalence among pregnant women (10). All pregnant smokers should be treated according to Public Health Service recommendations. However, sociodemographic data can help states tailor smoking interventions to populations with the highest prevalence. References
* Alabama, Alaska, California, Florida, Georgia, Indiana, Maine, Michigan, New York (excluding New York City), Oklahoma, South Carolina, Washington, and West Virginia. Table 1 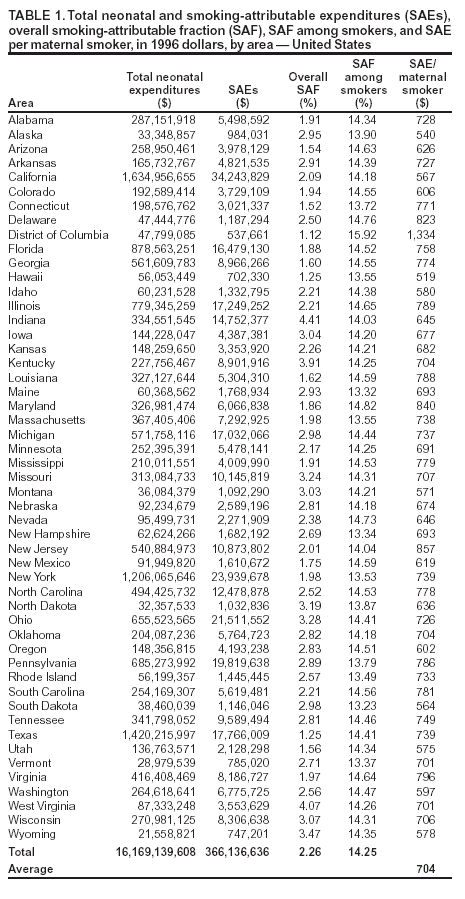 Return to top. Table 2 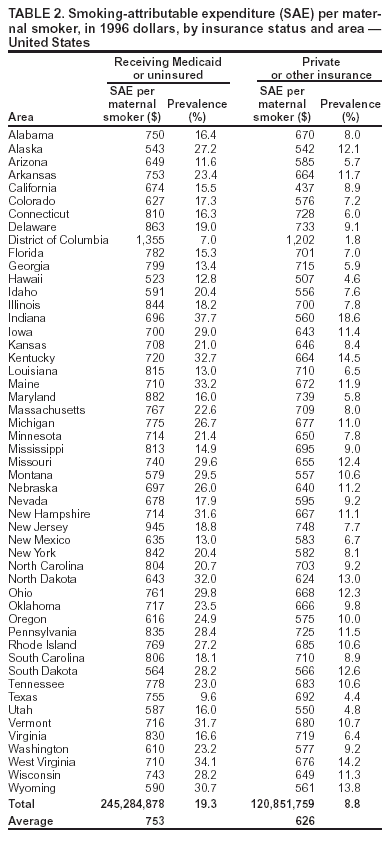 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 10/7/2004 |
|||||||||
This page last reviewed 10/7/2004
|