 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Spina Bifida and Anencephaly Before and After Folic Acid Mandate --- United States, 1995--1996 and 1999--2000Neural tube defects (NTDs) are serious birth defects of the spine (e.g., spina bifida) and the brain (e.g., anencephaly) that occur during early pregnancy, often before a woman knows she is pregnant; 50%--70% of these defects can be prevented if a woman consumes sufficient folic acid daily before conception and throughout the first trimester of her pregnancy (1). In 1992, to reduce the number of cases of spina bifida and other NTDs, the U.S. Public Health Service (USPHS) recommended that all women capable of becoming pregnant consume 400 µg of folic acid daily. Three approaches to increase folic acid consumption were cited: 1) improve dietary habits, 2) fortify foods with folic acid, and 3) use dietary supplements containing folic acid (1). Mandatory fortification of cereal grain products went into effect in January 1998; during October 1998--December 1999, the reported prevalence of spina bifida declined 31%, and the prevalence of anencephaly declined 16% (2). Other studies have indicated similar trends (3). To update the estimated numbers of NTD-affected pregnancies and births, CDC recently analyzed data from 23 population-based surveillance systems that include prenatal ascertainment of these birth defects. This report summarizes the results of that analysis, which indicate that the estimated number of NTD-affected pregnancies in the United States declined from 4,000 in 1995--1996 to 3,000 in 1999--2000. This decline in NTD-affected pregnancies highlights the partial success of the U.S. folic acid fortification program as a public health strategy. To reduce further the number of NTD-affected pregnancies, all women capable of becoming pregnant should follow the USPHS recommendation and consume 400 µg of folic acid every day. The numbers of annual NTD-affected birth defects were calculated from a 24-month prefortification period (1995--1996) and a 24-month postfortification period (1999--2000). To calculate the number of NTD-affected pregnancies (including live births, stillbirths, fetal deaths, and elective terminations), CDC estimated prevalence for spina bifida and anencephaly obtained from eight population-based surveillance systems that collect data systematically from sources that perform diagnostic prenatal ultrasounds as part of their surveillance programs (2). The numbers of spina bifida--affected pregnancies and anencephaly-affected pregnancies were calculated separately and then added together to provide an estimated total of NTD-affected pregnancies. Because the eight systems did not separate prenatally ascertained pregnancies from births, fetal deaths, and elective terminations, the remaining 15 population-based birth defects surveillance systems, which do not collect prenatally ascertained cases, were used to estimate the number of live births, stillbirths, and fetal deaths (occurring at >20 weeks' gestation) affected by NTDs (2). Previously published research on the ascertainment of NTD-affected pregnancies indicated that 9%--42% of such pregnancies were diagnosed prenatally (4). The number of live births used as the denominator for calculating the prevalence estimates is published by CDC's National Center for Health Statistics (5). In 1999, approximately 6.9 million pregnancies occurred in the United States, resulting in 4.0 million births, 1.3 million induced abortions, and 1.0 million fetal deaths (6). In one study, 87% of fetal deaths occurred during the embryonic period, when a diagnosis of an NTD would rarely be made or included in existing birth defect surveillance systems (7). Adjusting the denominator for those remaining (13%) fetal losses occurring at 14--20 weeks only slightly modified the prevalence and numbers of cases determined in the calculations. On the basis of data from the eight systems with prenatal ascertainment, an estimated 2,490 spina bifida--affected pregnancies and 1,640 anencephaly-affected pregnancies occurred annually before fortification of food with folic acid (Table). The total annual average number of NTD-affected pregnancies was 4,130. After fortification, an estimated 1,640 spina bifida--affected pregnancies and 1,380 anencephaly-affected pregnancies occurred, for an annual average of 3,020 NTD-affected pregnancies (a 27% decline). On the basis of data from the 15 systems without prenatal ascertainment, an estimated 1,980 spina bifida--affected births and 970 anencephaly-affected births occurred annually before fortification, for an annual average total of 2,950 NTD-affected live births, stillbirths, and fetal deaths at >20 weeks' gestation. After fortification, an estimated 1,340 births affected by spina bifida and 840 births affected by anencephaly occurred, for a total of 2,180 NTD-affected live births and stillbirths per year (a 26% decline). The difference between the number of cases reported from systems with and without prenatal ascertainment suggests that an estimated 1,180 fetal deaths (occurring at <20 weeks) or elective terminations occurred before fortification, compared with 840 after fortification. Reported by: P Mersereau, MN, K Kilker, Battelle Centers for Public Health Research and Evaluation; H Carter, MPH, E Fassett, MS, Assoc for Teachers of Preventive Medicine, Atlanta, Georgia. J Williams, MSN, A Flores, MPH, C Prue, PhD, L Williams, MPH, C Mai, MPH, J Mulinare, MD, Div of Birth Defects and Developmental Disabilities, National Center on Birth Defects and Developmental Disabilities, CDC. Editorial Note:The estimated decrease in the number of NTD-affected pregnancies after fortification highlights the success of folic acid fortification as a public health strategy. Periodic estimates of the number of NTD-affected pregnancies and births in the United States are needed to track the effects of ongoing and future public health activities. Although behavior changes (e.g., reported increased use of folic acid supplements) provide indirect evidence of the success of the folic acid mandate, the ultimate measure of success is the extent of the decline of NTD-affected pregnancies and births. The observed decrease in NTD-affected pregnancies of approximately 26% is less than what was estimated from research trials (1). More effort is needed if the 2010 national health objective of reducing the occurrence of spina bifida and other NTDs by 50% (objective no. 16-15) is to be achieved (8). The findings in this report are subject to at least two limitations. First, prevalence data were derived from surveillance systems. Because data collection is ongoing and data from the most recent years are likely to be incomplete because of reporting lags, only data through 2000 were used to calculate estimates. Second, because ascertainment of prenatally diagnosed cases varies by surveillance system, the number of cases probably is underestimated. However, prevalence of pregnancies or infants affected by NTDs collected for several years can provide reasonable estimates if the data are not too recent. The calculated prevalence data cover approximately one half of annual U.S. births in addition to prenatally diagnosed and electively terminated cases from eight surveillance systems (3). For the numbers of NTDs to be reduced, more effort is needed to increase consumption of 400 µg of folic acid each day by reproductive-aged women. Concerns include not only changing attitudes, knowledge, and behaviors of women, but also those of health-care providers regarding consumption of additional folic acid (e.g., folic acid--containing supplements or fortified breakfast cereals). Taking a vitamin with folic acid or eating a breakfast cereal fortified with 400 µg of folic acid per serving every day (9) are important components of the birth defects prevention efforts for women of childbearing age, regardless of pregnancy intention. Health-care providers can have a substantial impact on the increased use of folic acid--containing supplements. A total of 88% of women of reproductive age reported that they would take a folic acid--containing supplement if their health-care providers recommended it; however, only 37% of women reported that their health-care providers currently made that recommendation to them (Porter Novelli International, unpublished data, 2002). Ongoing monitoring of rates of NTDs and the assessment of the level of blood folates will help to quantify the effectiveness of the primary prevention effort for these serious birth defects in the United States. Additional studies will be needed to assess whether future NTD-affected pregnancies are the result of other causes besides folic acid insufficiency or a failure to consume the recommended amount of folic acid. References
Table 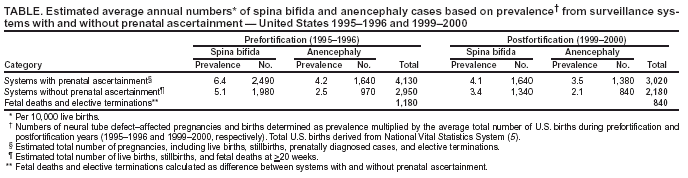 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 5/6/2004 |
|||||||||
This page last reviewed 5/6/2004
|