 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Update: Multistate Outbreak of Monkeypox --- Illinois, Indiana, Kansas, Missouri, Ohio, and Wisconsin, 2003CDC and state and local health departments continue to investigate cases of monkeypox among persons who had contact with wild or exotic mammalian pets or persons with monkeypox (1,2). This report updates epidemiologic, laboratory, and smallpox vaccine use data for U.S. cases. As of June 25, a total of 79 cases of monkeypox had been reported to CDC from Wisconsin (39), Indiana (20), Illinois (16), Missouri (two), Kansas (one), and Ohio (one) (Figure); these include 29 cases laboratory-confirmed at CDC and 51 cases under investigation by state and local health departments (Table). A total of 19 cases were excluded from those reported in the previous update because they met the exclusion criteria outlined in the updated case definition (2), and 11 were added. Of the 79 cases, 37 (47%) were among males; the median age was 28 years (range: 1--51 years). Age data were unavailable for two patients. Among 75 patients for whom data were available, 19 (25%) were hospitalized. Two patients have had a serious clinical illness. The first patient was a child with a previously reported laboratory-confirmed case of severe monkeypox-associated encephalitis (1,2); the child subsequently improved and was discharged after requiring hospitalization for 14 days. A second child, who was exposed to three ill prairie dogs, was hospitalized with profound painful cervical and tonsillar adenopathy and diffuse pox lesions, including lesions in the oropharynx. Although the child had difficulty breathing and swallowing, mechanical ventilation was not required. The adenopathy peaked 5 days after rash onset and 7 days after onset of initial prodromal symptoms of general malaise, myalgia, and fever. Preliminary testing of skin rash lesions was positive for orthopox virus; confirmatory testing for monkeypox virus is pending at CDC. Of the 79 reported cases, 29 (37%) have been laboratory confirmed at CDC for monkeypox by detection of virus in skin rash lesions by using culture, polymerase chain reaction (PCR), immunohistochemical testing, and/or electron microscopy. One patient had monkeypox virus detected by PCR and culture in throat and nasopharyngeal swabs obtained when the patient was ill with prodromal symptoms and a macular rash. In addition, an IgM response to orthopox viral antigen was detected in an acute serum sample. For these laboratory-confirmed cases, dates of illness onset ranged from May 16 to June 11. All confirmed patients reported a rash and at least one other clinical sign or symptom, including fever, respiratory symptoms, and/or lymphadenopathy. The median incubation period (i.e., first exposure date to illness onset date) was 12 days (range: 2--26 days). The majority of confirmed patients reported exposure to wild or exotic mammals, including prairie dogs; some patients also had contact with other persons with monkeypox virus infection in a household setting. No cases of monkeypox that could be attributed exclusively to person-to-person contact have been confirmed. Use of Smallpox VaccineTo prevent further transmission of monkeypox, 26 residents of five states have received smallpox vaccine since June 13; recipients included 24 adults and two children. Vaccine was administered to two laboratory workers pre-exposure and to 24 persons post-exposure (11 health-care workers, seven household contacts, three laboratory workers, two public health veterinarians, and one work contact). One adult who was vaccinated as a child did not have a major vaccine reaction or "take" 7 days after vaccination and required revaccination. CDC has issued updated interim guidance on the use of smallpox vaccine, cidofovir, and vaccinia immune globulin for prevention and treatment in the setting of an outbreak of monkeypox (3). Principal changes in the updated guidance include a revision of the definition of close contact with an ill animal, recommendations for vaccination of clinical laboratory workers handling specimens from ill animals and persons infected with monkeypox virus, and instructions for reporting smallpox vaccine--related serious adverse events to the Vaccine Adverse Event Reporting System (VAERS). Health-care providers, veterinarians, and public health officials who suspect monkeypox in animals or humans should report such cases to their state and local health departments. State health departments should report suspect cases to CDC, telephone 770-488-7100. Clinical specimens should be submitted for testing after consultation with the state and local health department. Interpretation of laboratory results requires completion of specimen submission forms, which are available at http://www.cdc.gov/ncidod/monkeypox/diagspecimens.htm. Additional information about monkeypox is available at http://www.cdc.gov/ncidod/monkeypox. Reported by: State and local health departments. Monkeypox investigation team, CDC. References
Acknowledgments This report is based on data contributed by MG Anderson, MD, Crusader Clinic; S Homann, MD, Rockford Infectious Disease Consultants, Rockford; L Frenkel, Dept of Pediatrics, Univ of Illinois, Chicago, Illinois. Table 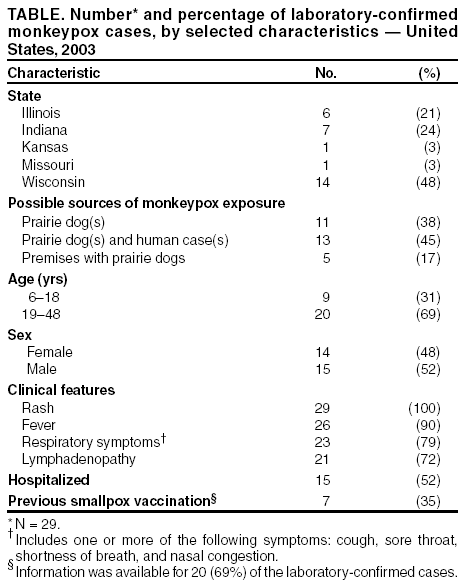 Return to top. Figure 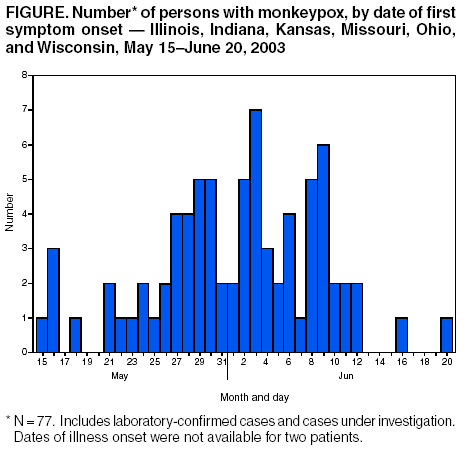 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 6/26/2003 |
|||||||||
This page last reviewed 6/26/2003
|