 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Update: Respiratory Syncytial Virus Activity -- United States, 1997-98 SeasonRespiratory syncytial virus (RSV) is the single most important cause of serious lower respiratory tract disease in infants and young children worldwide (1). In temperate climates, infections primarily occur during yearly outbreaks that usually peak during the winter months (2). RSV activity in the United States is monitored by the National Respiratory and Enteric Virus Surveillance System (NREVSS), a voluntary, laboratory-based system. This report summarizes trends for RSV reported to NREVSS from July 1997 to June 1998 and presents preliminary surveillance data from July 1 to November 18, 1998. Since July 1, 1990, 107 clinical and public health laboratories in 47 states and the District of Columbia have contributed data to NREVSS. Laboratories report weekly to CDC the number of specimens tested for RSV by antigen-detection and/or virus-isolation methods and the number of positive results. RSV activity is considered widespread by NREVSS when at least half of laboratories report any RSV detections for at least 2 consecutive weeks and when greater than 10% of all specimens tested by antigen detection for RSV are positive. From July 1990 through June 1998, widespread RSV activity began each November and continued for a mean of 22 weeks (range: 20-26 weeks), until April to mid-May (Figure_1). Peak activity for most laboratories occurred in January or February. For the 1997-98 season, 141,444 tests were performed, and 19,591 were positive for RSV. Median peak activity was observed in late December with peak activity occurring slightly earlier in southern sites* (November-December) than in northern sites** (January-February). Since the week ending November 13, 1998, 60% of the 59 laboratories reporting RSV test results have identified specimens positive for RSV, and 20% of reporting laboratories had greater than 10% of all tests positive for RSV, indicating the onset of widespread RSV activity for the 1998-99 season. Reported by: National Respiratory and Enteric Virus Surveillance System collaborating laboratories. Respiratory and Enteric Viruses Br, Div of Viral and Rickettsial Diseases, National Center for Infectious Diseases, CDC. Editorial NoteEditorial Note: The 1997-98 RSV season featured a longer period of widespread activity (26 weeks) than usual. The total specimens positive for RSV, months of peak activity, and south-to-north trend were consistent with data reported during previous years. Although not a population-based system, NREVSS consists of a large number of widely distributed laboratories and is an important tool for characterizing the spatiotemporal trends of RSV infections in the United States and can alert public health officials and physicians to seasonal RSV activity. During the RSV season (November-May), health-care providers should consider RSV as a cause of acute respiratory disease in both children and adults. RSV causes repeated symptomatic infections throughout life because of limited protective immunity induced by natural infection. Severe manifestations of RSV infection (e.g., pneumonia and bronchiolitis) most commonly occur in infants aged 2-6 months. In addition, RSV infection also can result in serious complications in older children and adults, especially those who have underlying cardiac or pulmonary disease or who are immunocompromised or elderly (3,4). Infection in immunocompromised persons can result in high death rates (5). RSV is a common but preventable cause of nosocomially acquired infection; the risk for nosocomial transmission increases during community outbreaks (6). Nosocomial infection may be acquired from infected patients, staff, visitors, or contaminated items in the patient's environment. Nosocomial outbreaks or transmission of RSV can be controlled with strict attention to contact-isolation procedures (6). Although vaccines are under development, none have been demonstrated to be safe and effective in preventing RSV-associated disease. RSV intravenous immune globulin and a recently licensed, humanized murine anti-RSV monoclonal antibody are available as prophylaxis for serious RSV infections in some high-risk infants and young children (e.g., those born prematurely or with chronic lung disease) (7). Ribavirin is the only available antiviral agent for treating RSV infection and may be considered for some patients (8). References
Figure_1 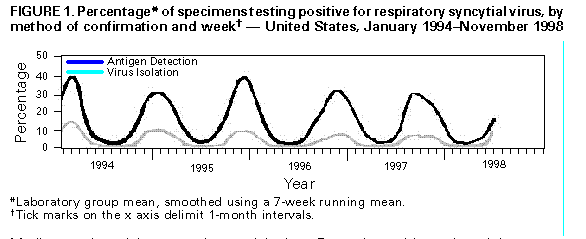 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 12/14/98 |
|||||||||
This page last reviewed 5/2/01
|