 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Serogroup B Meningococcal Disease -- Oregon, 1994In Oregon, the incidence of meningococcal disease has increased substantially, more than doubling from 2.2 cases per 100,000 persons in 1992 to 4.6 per 100,000 in 1994--the highest incidence in Oregon since 1943. This incidence was almost fivefold higher than recent estimates for the United States during 1989-1991 (approximately one case per 100,000 persons annually) (1). This report describes meningococcal disease surveillance data from 1994 and summarizes epidemiologic and laboratory data on serogroup B meningococcal disease in Oregon during 1987-1994. During 1994, a total of 143 cases of meningococcal disease was reported to the State Health Division. In 124 cases, Neisseria meningitidis was isolated from a normally sterile site (confirmed cases); in four cases, gram-negative diplococci were detected in specimens obtained from a normally sterile site or in persons who had classic symptoms after contact with a confirmed case (presumed cases). Characteristic symptoms (including petechial rash and hypotension) occurred in 14 cases; however, these cases were not culture confirmed (suspected cases). Of 115 isolates for which serogroup was known, 70 (61%) were serogroup B, 40 (35%) were serogroup C, four were serogroup Y, and one was serogroup W-135. When compared with 1992 and 1993, the serogroup-specific incidence in 1994 was higher for both serogroups B and C. Of the 70 culture-confirmed cases of serogroup B infection, 34 (49%) occurred in females. Seven (10%) cases were fatal; of these, one occurred in a child aged 2 years, and four deaths occurred in persons aged 55-88 years. During 1987-1992, 63% (84 of 133) of cases of serogroup B occurred in children aged less than 5 years; in comparison, in 1994, 27% (19 of 70) occurred in this age group. When compared with 1987-1992, the incidence of reported serogroup B disease in 1994 increased modestly among those aged less than 5 years (from 6.9 to 8.4), approximately 14-fold among those aged 15-19 years (from 0.4 to 5.4), and approximately fourfold among those aged greater than or equal to 60 years (from 0.3 to 1.1) Figure_1. In 1994, serogroup B cases occurred in 17 of the 36 counties in Oregon; these counties account for 83% of the total population of Oregon. The risk for disease was highest in counties in the Willamette Valley in the northwestern part of the state. Based on investigation of serogroup B cases, six (9%) were linked to other cases. Two co-primary cases (disease in a close contact within 24 hours of disease onset in a primary case) were linked to a single primary case. Four secondary cases (disease in a close contact greater than 24 hours after disease onset in a primary case) were identified; at least two occurred in patients for whom appropriate chemoprophylaxis had been prescribed but who were noncompliant with therapy. Of the 114 N. meningitidis serogroup B strains isolated in Oregon during 1993-1994, a total of 64 (56%) has been characterized at CDC by multilocus enzyme electrophoresis. Of these, 55 (86%) belong to the enzyme type-5 (ET-5) complex, a group of genetically related serogroup B meningococcal strains associated with epidemic meningococcal disease in other countries (2). Twelve of these isolates also have been serotyped, serosubtyped, and immunotyped; all are serotype 4 or 15, serosubtype P1.7,16, and immunotype L3,7,9,8,10. Reported by: K Hedberg, MD, F Hoesly, MD, D Fleming, MD, State Epidemiologist, State Health Div, Oregon Dept of Human Resources. K Steingart, MD, Southwest Washington Health District; M Goldoft, MD, P Stehr-Green, DrPH, State Epidemiologist, Washington Dept of Health. Div of Field Epidemiology, Epidemiology Program Office; Childhood and Respiratory Diseases Br, Div of Bacterial and Mycotic Diseases, National Center for Infectious Diseases, CDC. Editorial NoteEditorial Note: The recent increased occurrence of serogroup B meningococcal disease in Oregon has been associated with a group of closely related strains belonging to the ET-5 clonal complex. These strains were first identified as the cause of a serogroup B meningococcal epidemic in Norway that began in 1974 and persisted through 1991 (3). After its identification in 1974, serogroup B meningococci belonging to the ET-5 complex subsequently caused epidemics in Europe, Cuba, and South America (2). Endemic meningococcal disease typically is caused by a heterogeneous mix of strains. In comparison, the predominance of closely related strains, or clones, in Oregon is characteristic of epidemic disease, as is the disproportionate increase in age-specific incidence among young adults. The latter pattern has been suggested as a reliable predictor of the transition from endemic to epidemic meningococcal disease (4,5). Although serogroup C meningococcal outbreaks have occurred with increasing frequency nationwide since 1991 (6), Oregon is the only state to report substantially increased rates of serogroup B meningococcal disease. While Washington state had only a slight increase in the overall rate of meningococcal disease in 1994, rates of serogroup B meningococcal disease in Clark County, Washington (1994 population: 280,800) (across the Columbia River from metropolitan Portland, Oregon), have increased almost fivefold (from 1.5 per 100,000 in 1987 to 7.1 per 100,000 in 1994). These increasing rates underscore the need for determining the serogroup of all isolates to assist in assessing trends in the occurrence of meningococcal disease and serogroup distribution of invasive N. meningitidis in other states. The primary means for the control and prevention of serogroup B meningococcal disease is chemoprophylaxis of close contacts. The meningococcal vaccine licensed in the United States provides protection against serogroups A, C, Y, and W-135 but does not provide protection against serogroup B meningococcal disease. Meningococcal capsular polysaccharides determine serogroup and are used in purified form to produce the A/C/Y/W-135 vaccine. Unlike the other major meningococcal serogroups, however, serogroup B capsular polysaccharide is poorly immunogenic in humans. Alternate approaches to the development of a serogroup B meningococcal vaccine have focused on use of outer-membrane proteins from specific epidemic serogroup B meningococcal strains. Three outer-membrane protein-based serogroup B meningococcal vaccines employing two-dose regimens have been effective among older children and young adults in large clinical trials outside the United States (7-9); estimated efficacies ranged from 57% to 83%. The only vaccine available commercially is not licensed for use in the United States but has been used in some South American countries to control serogroup B meningococcal epidemics. In Sao Paulo, Brazil, approximately 2.4 million children aged 3 months to 6 years were vaccinated during 1989 and 1990, and the vaccine was estimated to be 74% effective in children aged 4-6 years (10). Efforts to initiate studies in the United States to evaluate available vaccines under an investigational new drug application are in progress. Oregon and three other states (California, Connecticut, and Minnesota) are participating in a cooperative agreement with CDC to study emerging infectious diseases. One focus of this program in Oregon is disease caused by N. meningitidis serogroup B; a study of potentially modifiable risk factors for meningococcal disease is under way. References
Figure_1 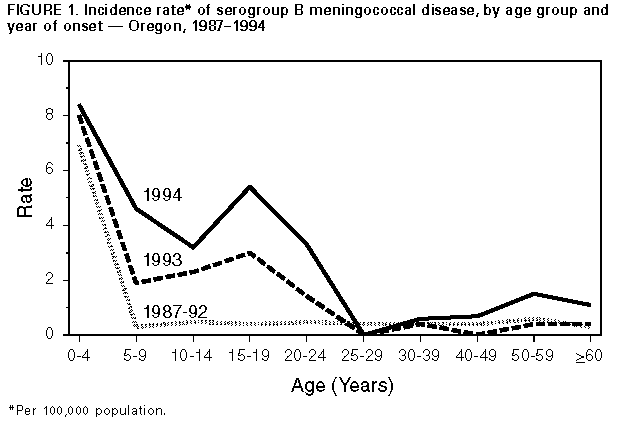 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 09/19/98 |
|||||||||
This page last reviewed 5/2/01
|