 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Current Trends Update: Influenza Activity -- United States and Worldwide, 1993-94 Season, and Composition of the 1994-95 Influenza VaccineIn collaboration with the World Health Organization (WHO) and its network of international collaborating laboratories and with state and local health departments in the United States, CDC conducts surveillance to monitor influenza activity and to detect antigenic changes in the circulating strains of influenza viruses. This report summarizes surveillance for influenza in the United States and worldwide during the 1993-94 season and describes the composition of the 1994-95 influenza vaccine. United States During August and early September 1993, three outbreaks of influenza type A(H3N2) associated with high attack rates occurred in Louisiana (1). Virologic or serologic evidence indicated that all three outbreaks were caused by viruses similar to the A/Beijing/32/92 strain, which was first isolated in the United States during the 1992-93 influenza season and was included in the influenza vaccine formulated for the 1993-94 season. Regional * influenza activity associated with laboratory-confirmed outbreaks of influenza type A(H3N2) was first reported in early November 1993 in Wyoming and Montana and in mid-November in Idaho. In all three states, outbreaks were first recognized among schoolchildren (2). Influenza activity increased from mid-November 1993 through early January 1994. Although the timing and intensity of influenza activity varied by region, influenza activity peaked nationally during the last week of 1993 and the first week of 1994. The proportion of patient visits for influenza-like illness to family practitioners participating in the CDC sentinel physician surveillance system peaked at 8% during the week ending January 1, 1994. Reports from state and territorial epidemiologists and from the WHO collaborating laboratories peaked during the week ending January 8, when state and territorial epidemiologists reported either widespread or regional influenza activity in 35 states, and WHO collaborating laboratories in the United States reported 709 influenza virus isolates. Although most reported outbreaks occurred in schools, outbreaks were reported among persons in all age groups; reports of high absenteeism in the workplace were common during peak influenza activity. Outbreaks also occurred among residents of nursing homes. Of total deaths reported through CDC's 121-city mortality surveillance system, the proportion attributed to pneumonia and influenza (P&I) exceeded the epidemic threshold ** for 10 consecutive weeks from December 19, 1993, through February 26, 1994 (Figure_1). The highest proportion of P&I deaths (9.2% of total deaths) was reported the week ending January 22. Of the 3963 influenza virus isolates reported to CDC from WHO collaborating laboratories in the United States through March 5, 99.9% were influenza type A; only four of the isolated viruses were influenza type B. Of the 1899 influenza type A viruses that have been subtyped, 99% have been influenza type A(H3N2). Worldwide Influenza activity worldwide has occurred at moderate to moderately severe levels. Influenza viruses have been isolated in association with sporadic activity, outbreaks, or epidemic activity in Asia, Europe, and North America. Although most activity has been associated with influenza type A(H3N2), influenza type B viruses were isolated during periods of sporadic activity or outbreaks in some countries. Isolation of influenza type A(H1N1) viruses has been rare. Influenza type A(H3N2) viruses were first detected during localized outbreaks that occurred during August and September in the United States and in Scotland. An epidemic caused by type A(H3N2) occurred in the United Kingdom during November and December. In western and northern continental Europe (Austria, Belgium, Denmark, Finland, France, the Netherlands, Norway, Sweden, and Switzerland), influenza type A(H3N2) epidemics occurred during November and December. From October through February, sporadic cases or outbreaks caused by influenza type A(H3N2) also were reported in Bulgaria, Croatia, the Czech Republic, Germany, Greece, Iceland, Ireland, Italy, Japan, People's Republic of China, Romania, the Russian Federation, Spain, Yugoslavia, and Zambia. When compared with type A influenza, type B viruses have been isolated less frequently worldwide; influenza type B isolates were first reported in association with sporadic activity in China, Hong Kong, and Thailand during October and November. Outbreaks caused by type B viruses subsequently occurred in China during December and January. Influenza type B viruses also were reported during outbreaks in Slovakia and in association with sporadic activity in Canada, Finland, Japan, the Netherlands, Portugal, the Russian Federation, Spain, Sweden, Switzerland, the United Kingdom, and the United States. Influenza type A(H1N1) viruses have been reported in association with sporadic activity from Hungary, Hong Kong, the Netherlands, the Russian Federation, and the United States. Composition of the 1994-95 Vaccine The Food and Drug Administration Vaccines and Related Biologicals Advisory Committee (VRBAC) has recommended that the 1994-95 trivalent influenza vaccine for the United States contain A/Texas/36/91-like (H1N1), A/Shangdong/9/93-like (H3N2), and B/Panama/45/90-like viruses. This recommendation was based on the antigenic analysis of recently isolated influenza viruses and the antibody response of persons vaccinated with the 1993-94 vaccine. Although many of the influenza type A(H3N2) viruses that have been antigenically characterized are similar to the A/Beijing/32/92 strain included in the 1993-94 vaccine, some recently isolated strains from Asia, Europe, and North America are more similar to the antigenic variant A/Shangdong/9/93 (Table_1). Vaccines containing the A/Beijing/32/92 virus induced a good antibody response to the vaccine strain but induced lower and less frequent antibody responses to recent type A(H3N2) strains such as A/Shangdong/9/93 (3). Therefore, VRBAC recommended changing the influenza type A(H3N2) vaccine component to an A/Shangdong/9/93-like strain for the 1994-95 season. Influenza B viruses that have been antigenically characterized, including the most recent isolates from China, are similar to B/Panama/45/90 and the closely related variant B/Qingdao/102/91 (4). Vaccines containing B/Panama/45/90 virus induced antibodies at a similar frequency and titer to the vaccine virus and to representative recent isolates. VRBAC therefore recommended retaining a B/Panama/45/90-like vaccine strain in the 1994-95 vaccine. Because isolation of influenza type A(H1N1) virus has been rare worldwide during the 1993-94 season, no type A(H1N1) viruses isolated since October 1993 have been characterized. However, viruses characterized during the 1992-93 season were closely related to the reference strains A/Taiwan/1/86 or A/Texas/36/91. Vaccines containing the A/Texas/36/91 strain induced antibodies with similar frequency and titer to the vaccine virus and to type A(H1N1) strains isolated during the 1992-93 influenza season. Therefore, VRBAC recommended retaining an A/Texas/36/91-like strain in the 1994-95 vaccine. Reported by: Participating state and territorial health dept epidemiologists and state public health laboratory directors. M Chakraverty, PhD, Central Public Health Laboratory, J Skehel, PhD, A Hay, PhD, National Institute for Medical Research, London; G Schild, PhD, J Wood, PhD, National Institute for Biological Standards and Control, Hertfordshire, England. I Gust, MD, A Hampson, Commonwealth Serum Laboratories, Parkville, Australia. World Health Organization National Influenza Centers, Microbiology and Immunology Support Svcs, Geneva. Div of Virology, Center for Biologics Evaluation and Research, Food and Drug Administration. Epidemiology Activity and World Health Organization Collaborating Center for Surveillance, Epidemiology, and Control of Influenza, Div of Viral and Rickettsial Diseases, National Center for Infectious Diseases, CDC. Editorial NoteEditorial Note: The outbreaks of influenza in Louisiana in August and September 1993 were unusual because they occurred during the summer and were characterized by high attack rates. Influenza virus infections during the summer or fall in the United States usually occur as sporadic cases rather than as outbreaks. Outbreaks of influenza during the summer have been associated with earlier than usual epidemic influenza activity (5-7). The 1993-94 influenza season began and peaked earlier than usual in the United Kingdom and in the United States. In the United States, reports of sustained regional and widespread activity began and peaked 1-6 weeks (mean: 5 weeks) earlier than in 10 of the previous 11 influenza seasons; sustained excess mortality attributable to P&I began earlier than in any of the previous 11 seasons. Compared with seasons of predominant influenza type A(H1N1) or type B activity, seasons in which influenza type A(H3N2) viruses predominate are associated with higher morbidity and mortality among the elderly. During the 1993-94 season -- which has been characterized by predominant type A(H3N2) activity -- all age groups have been affected, and influenza-related mortality has been high, especially among the elderly. Strains to be included in the next season's influenza vaccine are selected usually during the preceding late January through February because of scheduling requirements for production, quality control, packaging, and distribution of vaccine for administration before onset of the next influenza season. Recommendations of the Advisory Committee on Immunization Practices for the use of vaccine and antiviral agents for prevention and control of influenza are published annually in the MMWR Recommendations and Reports, usually during May. References
* Levels of activity are 1) sporadic -- sporadically occurring influenza-like illness (ILI) or culture-confirmed influenza with no outbreaks detected; 2) regional -- outbreaks of ILI or culture- confirmed influenza in counties with a combined population of less than 50% of the state's total population; and 3) widespread -- outbreaks of ILI or culture-confirmed influenza in counties having a combined population of 50% or more of the state's total population. *
* The epidemic threshold is 1.645 standard deviations above the
seasonal baseline. The expected seasonal baseline is projected
using a robust regression procedure in which a periodic regression
model is applied to observed percentages of deaths from P&I since
1983.
TABLE 1. Hemagglutination-inhibition titers of influenza A(H3N2) viruses with
serum specimens from infected ferrets *
================================================================================
Ferret antiserum
Viral antigen A/Beijing/32/92 A/Hong Kong/23/92 A/Shangdong/9/93
---------------------------------------------------------------------------
Reference antigen
A/Beijing/32/92 640 160 320
A/Hong Kong/23/92 160 640 320
A/Shangdong/9/93 160 320 320
Recent isolates
A/Georgia/3/93 80 80 320
A/Canada/251/94 80 160 160
A/Lyon/1983/93 160 160 320
A/Nanchang/58/93 160 160 320
A/Netherlands/261/93 80 320 160
---------------------------------------------------------------------------
* A fourfold difference in hemagglutination-inhibition titers with two viruses
is usually indicative of antigenic variation between viruses.
================================================================================
Return to top. Figure_1 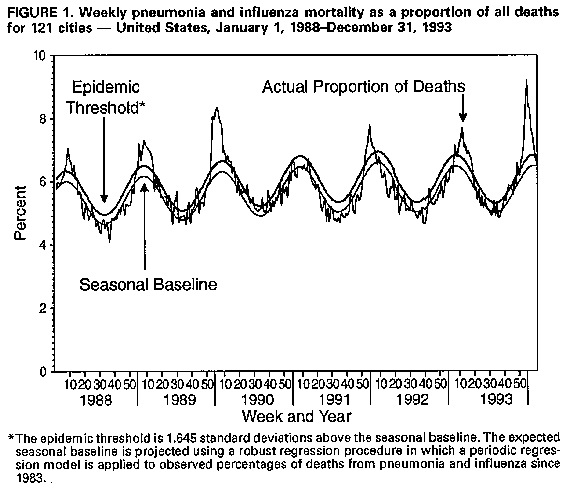 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 09/19/98 |
|||||||||
This page last reviewed 5/2/01
|