 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Malaria Surveillance --- United States, 2001Scott Filler, M.D.1,2 AbstractProblem/Condition: Malaria is caused by any of four species of intraerythrocytic protozoa of the genus Plasmodium (i.e., P. falciparum, P. vivax, P. ovale, or P. malariae). These parasites are transmitted by the bite of an infective female Anopheles sp. mosquito. The majority of malaria infections in the United States occur among persons who have traveled to areas with ongoing transmission. In the United States, cases can occur through exposure to infected blood products, by congenital transmission, or by local mosquitoborne transmission. Malaria surveillance is conducted to identify episodes of local transmission and to guide prevention recommendations for travelers. Period Covered: This report covers cases with onset of illness in 2001. Description of System: Malaria cases confirmed by blood film are reported to local and state health departments by health-care providers or laboratory staff. Case investigations are conducted by local and state health departments, and reports are transmitted to CDC through the National Malaria Surveillance System (NMSS). Data from NMSS serve as the basis for this report. Results: CDC received reports of 1,383 cases of malaria with an onset of symptoms in 2001 among persons in the United States or one of its territories. This number represents a decrease of 1.4% from the 1,402 cases reported for 2000. P. falciparum, P. vivax, P. malariae, and P. ovale were identified in 50.1%, 27.8%, 4.5%, and 3.6% of cases, respectively. Fourteen patients (1.0% of total) were infected by >2 species. The infecting species was unreported or undetermined in 179 (12.9%) cases. Compared with 2000, the number of reported malaria cases acquired in Africa increased by 13.2% (n = 886), whereas the number of cases acquired in Asia (n = 163) and the Americas (n = 240) decreased by 31.5% and 11.4%, respectively. Of 891 U.S. civilians who acquired malaria abroad, 180 (20.2%) reported that they had followed a chemoprophylactic drug regimen recommended by CDC for the area to which they had traveled. Two patients became infected in the United States, one through congenital transmission and one whose infection cannot be linked epidemiologically to secondary cases. Eleven deaths were attributed to malaria, 10 caused by P. falciparum and one caused by P. ovale. Interpretation: The 1.4% decrease in malaria cases in 2001, compared with 2000, resulted primarily from a decrease in cases acquired in Asia and the Americas, but this decrease was offset by an increase in the number of cases acquired in Africa. This decrease probably represents year-to-year variation in malaria cases, but also could have resulted from local changes in disease transmission, decreased travel to malaria-endemic regions, fluctuation in reporting to state and local health departments, or an increased use of effective antimalarial chemoprophylaxis. In the majority of reported cases, U.S. civilians who acquired infection abroad were not on an appropriate chemoprophylaxis regimen for the country in which they acquired malaria. Public Health Actions: Additional information was obtained concerning the 11 fatal cases and the two infections acquired in the United States. Persons traveling to a malarious area should take one of the recommended chemoprophylaxis regimens appropriate for the region of travel, and travelers should use personal protection measures to prevent mosquito bites. Any person who has been to a malarious area and who subsequently develops a fever or influenza-like symptoms should seek medical care immediately and report their travel history to the clinician; investigation should include a blood-film test for malaria. Malaria infections can be fatal if not diagnosed and treated promptly. Recommendations concerning malaria prevention can be obtained from CDC by calling the Malaria Hotline at 770-488-7788 or by accessing CDC's Internet site at http://www.cdc.gov/travel. IntroductionMalaria is caused by infection with one or more of four species of Plasmodium (i.e., P. falciparum, P. vivax, P. ovale, and P. malariae) that can infect humans. The infection is transmitted by the bite of an infective female Anopheles sp. mosquito. Malaria infection remains a devastating global problem, with an estimated 300--500 million cases occurring annually (1). Forty-one percent of the world's population lives in areas where malaria is transmitted (e.g., parts of Africa, Asia, the Middle East, Central and South America, Hispaniola, and Oceania) (1), and 700,000--2.7 million persons die of malaria each year, 75% of them African children (2). In previous decades, malaria was endemic throughout the southeastern United States; an estimated 600,000 cases occurred in 1914 (3). During the late 1940s, a combination of improved socioeconomic conditions, water management, vector-control efforts, and case management was successful at interrupting malaria transmission in the United States. Since then, malaria case surveillance has been maintained to detect locally acquired cases that could indicate the reintroduction of transmission and to monitor patterns of antimalarial drug resistance. Anopheline mosquitos remain seasonally present in all states except Hawaii. Through 2001, the majority of cases of malaria diagnosed in the United States have been imported from regions of the world where malaria transmission is known to occur, although congenital infections and infections resulting from exposure to blood or blood products are also reported in the United States. In addition, a limited number of cases are reported that might have been acquired through local mosquitoborne transmission (4). State and local health departments and CDC investigate malaria cases acquired in the United States, and CDC analyzes data from imported cases to detect acquisition trends. This information is used to guide malaria prevention recommendations for travelers abroad. For example, an increase in P. falciparum malaria among U.S. travelers to Africa, an area with increasing chloroquine resistance, prompted CDC to change the recommended chemoprophylaxis regimen from chloroquine to mefloquine in 1990 (5). The signs and symptoms of malaria illness are varied, but the majority of patients experience fever. Other common symptoms include headache, back pain, chills, increased sweating, myalgia, nausea, vomiting, diarrhea, and cough. The diagnosis of malaria should be considered for persons who experience these symptoms and who have traveled to an area with known malaria transmission. Malaria should also be considered in the differential diagnoses of persons who experience fevers of unknown origin, regardless of their travel history. Untreated P. falciparum infections can rapidly progress to coma, renal failure, pulmonary edema, and death. Asymptomatic parasitemia can occur, most commonly among persons who have been long-term residents of malarious areas. This report summarizes malaria cases reported to CDC with onset of symptoms in 2001. MethodsData SourcesMalaria case data are reported to the National Malaria Surveillance System (NMSS) and the National Notifiable Diseases Surveillance System (NNDSS) (6). Although both systems rely on passive reporting, the numbers of reported cases might differ because of differences in collection and transmission of data. A substantial difference in the data collected in these two systems is that NMSS receives more detailed clinical and epidemiologic data regarding each case (e.g., information concerning the area to which the infected person has traveled). This report presents only data regarding cases reported to NMSS. Cases of blood-film--confirmed malaria among civilians and military personnel are identified by health-care providers or laboratories. Each slide-confirmed case is reported to local or state health departments and to CDC on a uniform case report form that contains clinical, laboratory, and epidemiologic information. CDC staff review all report forms when received and request additional information from the provider or the state, if necessary (e.g., when no recent travel to a malarious country is reported). Reports of other cases are telephoned directly by health-care providers to CDC, usually when assistance with diagnosis or treatment is requested. Cases reported directly to CDC are shared with the relevant state health department. All cases that have been acquired in the United States are investigated, including all induced and congenital cases and possible introduced or cryptic cases. Information derived from uniform case report forms is entered into a database and analyzed annually. DefinitionsThe following definitions are used in this report:
This report also uses terminology derived from the recommendations of the World Health Organization (7). Definitions of the following terms are included for reference:
Microscopic Diagnosis of MalariaThe early diagnosis of malaria requires that physicians consider malaria in the differential diagnosis of every patient who is experiencing fever; the evaluation of such a patient should include taking a comprehensive travel history. If malaria is suspected, a Giemsa-stained film of the patient's peripheral blood should be examined for parasites. Thick and thin blood films must be prepared correctly because diagnostic accuracy depends on blood-film quality and examination by experienced laboratory personnel* (Appendix). ResultsGeneral SurveillanceFor 2001, CDC received 1,383 malaria case reports occurring among persons in the United States and its territories, representing a 1.4% decrease from the 1,402 cases reported with a date of onset in 2000 (8). This incidence is the fifth highest number of reported cases since 1980 and represents the highest number of U.S. civilian cases reported in the previous 30 years (Table 1). In 2001, a total of 891 cases occurred among U.S. civilians, compared with 827 cases reported for 2000, whereas the number of cases among foreign civilians decreased from 354 cases to 316 (Figure 1). Cases among U.S. military personnel decreased from 46 to 18 in 2001. In 158 cases, information was insufficient to determine civilian or military status. Plasmodium SpeciesThe infecting species of Plasmodium was identified in 1,204 (87.1%) of the cases reported in 2001. P. falciparum and P. vivax were identified in blood films from 50.1% and 27.8% of infected persons, respectively (Table 2). The 693 P. falciparum cases reported for 2001 represented a 13.4% increase from the 611 cases in 2000, whereas the number of P. vivax infections decreased by 26.2% (from 522 in 2000 to 385 in 2001). Among 1,149 cases in which both the region of acquisition and the infecting species were known, 76.6% of infections acquired in Africa were attributed to P. falciparum; 11.0% were attributed to P. vivax. The converse was true of infections acquired in the Americas and Asia: 70.0% and 79.7% were attributed to P. vivax, and only 23.8% and 11.9% were attributed to P. falciparum, respectively. Region of Acquisition and DiagnosisAll but two of reported cases (n = 1,381) were imported. Of 1,309 imported cases in which the region of acquisition was known, the majority (67.7%; n = 886) were acquired in Africa; 18.3% (n = 240) and 12.5% (n = 164) were acquired in the Americas and Asia, respectively (Table 3). A limited number of imported cases were acquired in Oceania (1.5%; n = 19). The highest concentration of cases acquired in Africa came from countries in West Africa (65.0%; n = 576); a substantial percentage of cases acquired in Asia came from the Indian subcontinent (49.4%; n = 81). From within the Americas, the majority of cases were acquired in Central America and the Caribbean (73.3%; n = 176), followed by South America (17.9%; n = 43). Information regarding region of acquisition was missing for 72 (5.2%) of the imported cases. Compared with 2000, the number of reported malaria cases acquired in Africa increased by 13.2%, and the number of cases acquired in Asia and the Americas decreased by 31.1% and 11.4%, respectively. In the United States, the five health departments reporting the highest number of malaria cases were New York City (n = 239), California (n = 173), New York State (n = 79), New Jersey (n = 69), and Florida (n = 65) (Figure 2). Whereas the majority of these health departments reported an increase in cases compared with 2000, an overall decrease in cases occurred nationwide. This decrease probably represents year-to-year variation in malaria cases rather than a trend but could also have resulted from local changes in disease transmission, decreased travel to malaria-endemic regions, fluctuation in reporting to state and local health departments, or an increased use of effective antimalarial chemoprophylaxis. Interval Between Arrival and IllnessThe interval between date of arrival in the United States and onset of illness and the infecting Plasmodium species were known for 678 (49.0%) of the imported cases of malaria (Table 4). Symptoms began before arrival in the United States for 98 (14.5%) persons, whereas symptoms began after arrival in the United States for 580 (85.5%) of these patients. Clinical malaria developed within 1 month after arrival in 351 (76.7%) of the 452 P. falciparum cases and in 55 (33.3%) of the 165 P. vivax cases (Table 4). Only 10 (1.5%) of the 678 persons became ill >1 year after returning to the United States. Imported Malaria CasesImported Malaria Among U.S. Military Personnel In 2001, a total of 18 cases of imported malaria was reported among U.S. military personnel. Of the 14 cases for whom information regarding chemoprophylaxis use was available, three patients were not using any prophylaxis. Imported Malaria Among Civilians A total of 1,207 imported malaria cases were reported among civilians. Of these, 891 (73.8%) cases occurred among U.S. residents, and 316 (26.2%) cases occurred among residents of other countries (Table 5). Of the 891 imported malaria cases among U.S. civilians, 634 (71.2%) had been acquired in Africa, an increase of 14.2% from cases reported in 2000. Asia accounted for 103 (11.6%) cases of imported malaria among U.S. civilians, and travel to the Central American and Caribbean regions accounted for an additional 92 (10.3%) cases. Of the 316 imported cases among foreign civilians, the majority of cases were acquired in Africa (n = 188; 59.5%). Antimalarial Chemoprophylaxis UseChemoprophylaxis Use Among U.S. Civilians Information concerning chemoprophylaxis use and travel area was known for 815 (91.5%) of the 891 U.S. civilians who had imported malaria. Of these 815 persons, 487 (59.8%) had not taken any chemoprophylaxis, and 122 (15.0%) had not taken a CDC-recommended drug for the area visited (9). Only 180 (22.1%) U.S. civilians had taken a CDC-recommended medication (9). Data for the specific drug taken were missing for the remaining 26 (3.2%) travelers. A total of 124 (68.9%) patients on CDC-recommended prophylaxis had taken mefloquine weekly; 28 (15.6%) had taken doxycycline daily; and 12 (6.7%) who had traveled only in areas where chloroquine-resistant malaria has not been documented had taken chloroquine weekly. Sixteen patients (8.9%) had taken combinations of drugs that included >1 CDC-recommended drug for the travel region. Of the 122 patients taking a nonrecommended drug, 61 (50.0%) reported taking chloroquine either alone or in combination with another ineffective drug during travel to an area where chloroquine resistance has been documented. Malaria Infection After Recommended Prophylaxis Use A total of 200 patients (i.e., 180 U.S. civilians, eight persons in the U.S. military, five foreign civilians, and seven persons whose information regarding their status was missing) experienced malaria after taking a recommended antimalarial drug for chemoprophylaxis. Information regarding infecting species was available for 176 (88.0%) patients taking a recommended antimalarial drug; the infecting species was undetermined for the remaining 24. Cases of P. vivax or P. ovale After Recommended Prophylaxis Use. Of the 200 patients who experienced malaria after recommended chemoprophylaxis use, 74 cases (37.0%) were caused by P. vivax and 14 (7.0%) by P. ovale. Notes on the malaria case surveillance reports indicated that 21 (23.9%) of these 88 patients were noncompliant with antimalarial prophylaxis. A total of 32 (33.7%) cases of P. vivax or P. ovale occurred >45 days after arrival in the United States. These cases were consistent with relapsing infections and, thus, do not indicate primary prophylaxis failures. Information was insufficient, because of missing data regarding symptom onset or return date, to assess whether 40 cases were relapsing infections. Sixteen cases, 15 by P. vivax and one by P. ovale, occurred <45 days after the patient returned (n = 13) or before return (n = 3) to the United States. Of these 16 patients, four were known to be noncompliant with their antimalarial chemoprophylaxis. Region of acquisition varied for the 12 remaining case-patients who were not known to be noncompliant (three from West Africa, one from Central Africa, three from Central America, four from Asia, and one from Papua New Guinea). Serum drug levels were available for the patient who had traveled to Papua New Guinea. The patient had reported taking mefloquine for prophylaxis, but serum drug levels were undetectable, thus indicating either noncompliance with the recommended regimen or malabsorption of the drug. Blood samples were not available for the remaining 11 cases; serum drug levels were not measured for any of these patients. The probable explanations for these cases are either inappropriate dosing or noncompliance that was not reported. Evidence is lacking that would indicate any new area of chloroquine-resistant P. vivax. Cases of P. falciparum and P. malariae after Recommended Prophylaxis Use. The remaining 112 cases of malaria reported among persons who had taken a recommended antimalarial drug for chemoprophylaxis include 74 cases of P. falciparum, nine cases of P. malariae, five cases of mixed infection, and 24 cases in which the infecting species was unidentified. A total of 64 of the 74 P. falciparum cases among those who reported taking a recommended antimalarial drug was acquired in Africa, six in the Caribbean, two in Asia, and two in Oceania. In 27 (36.5%) of these 74 cases, noncompliance with antimalarials was reported. Of the remaining 47 cases of P. falciparum for which patient compliance was unknown, the majority was acquired in Africa (n = 38): 25 in West Africa, four in southern Africa, four in East Africa, two in Central Africa, and three in an unspecified African region. Nine cases were acquired outside Africa: six in the Caribbean (Haiti) and three others (Indonesia, Papua New Guinea, and an unspecified region). Serum drug levels were not available for any of these 47 patients. Eight of the nine P. malariae cases among those who reported taking a recommended antimalarial drug were acquired in Africa. In two (22.2%) of these nine cases, noncompliance with antimalarials was reported. In the seven remaining cases, whether the patient complied with prophylaxis was unknown; six had traveled in Africa, and one in Central America. Purpose of TravelPurpose of travel to malaria-endemic areas was reported for 678 (76.1%) of the 891 U.S. civilians with imported malaria (Table 6). Of the U.S. civilians with malaria, the largest percentage (37.4%) were persons who had visited friends or relatives in malarious areas; the second and third highest percentages, 10.6% and 9.2%, had traveled for tourism and to do missionary work, respectively. Malaria During PregnancyA total of 22 cases of malaria was reported among pregnant women in 2001, representing 5.0% of cases among women. Seven of the 22 (31.8%) were among U.S. civilians. Four of these seven women had traveled to visit friends and relatives; six had traveled in Africa, and one in Asia. Only four pregnant women of 22 (18.1%) reported taking prophylaxis, compared with 31.3% of nonpregnant women. Malaria Acquired in the United StatesCongenital Malaria One case of congenital malaria was reported in 2001 and is described in the following case report:
Cryptic Malaria One case of cryptic malaria was reported in 2001 and is described in the following case report:
Deaths Attributed to MalariaEleven deaths attributable to malaria were reported in 2001 and are described in the following case reports:
DiscussionA total of 1,383 cases of malaria were reported to CDC for 2001, representing a 1.4% decrease from the 1,402 cases reported for 2000. This change primarily resulted from a decrease in cases acquired in Asia and the Americas, which was largely offset by an increase in the number of cases acquired in Africa. Since 2000, CDC has routinely contacted state health departments to ask for outstanding malaria case reports from the previous reporting year or for a statement that reporting is complete. The decrease in cases in 2001, compared with 2000, most likely is as result of expected variation in the number of cases, although other possibilities include decreased international travel, changing patterns of travel (e.g., decreased immigration from malarious areas), or an increased use of effective antimalarial chemoprophylaxis. One reason for conducting malaria surveillance is to monitor for prophylaxis failures that might indicate emergence of drug resistance; however, ~75% of imported malaria among U.S. civilians occurred among persons who were either not taking prophylaxis or were taking nonrecommended prophylaxis for the region to which they were traveling. Of the 99 cases where appropriate prophylaxis was reported and for whom adequate information was available regarding species and onset of symptoms to indicate that the infection was a primary one rather than a relapse, 66 (i.e., 47 P. falciparum, 11 P. vivax, 7 P. malariae, and 1 P. ovale) had insufficient information to determine whether these cases represented problems with adherence while using correct antimalarial chemoprophylaxis, malabsorption of the antimalarial drug, or emerging drug resistance. No conclusive evidence existed to indicate a single national or regional source of infection among this group of patients or the failure of a particular chemoprophylactic regimen. Health-care providers are encouraged to contact CDC rapidly whenever they suspect chemoprophylaxis failure, thus enabling measurement of serum drug levels of the antimalarial drugs in question. In 2001, to be better able to evaluate chemoprophylaxis failures, CDC revised the NMSS case report form to facilitate collection of more thorough data regarding chemoprophylaxis. The current form solicits more detailed information regarding the prescribed regimen, the degree of compliance with the regimen, and the reasons for noncompliance, if any. Data gathered from the responses will be useful in generating public health messages to improve use of antimalarial chemoprophylaxis and therefore decrease malaria-associated morbidity and mortality among U.S. civilians. The importance of taking correct precautions and chemoprophylaxis is underscored by the 11 fatal cases of malaria that occurred in the United States in 2001. An earlier review of deaths attributed to malaria in the United States identified certain risk factors for fatal malaria, including failure to take recommended antimalarial chemoprophylaxis, refusal of or delay in seeking medical care, and misdiagnosis (11). The occurrence of 22 cases of malaria among pregnant U.S. civilians is also cause for concern. Malaria during pregnancy among nonimmune women is more likely to result in severe disease or contribute to an adverse outcome than malaria in nonpregnant women (12); the fetus might be adversely affected as well (13). Pregnant travelers should be counseled to avoid travel to malarious areas, if possible. If deferral of travel is impossible, pregnant women should be informed that the risks for malaria outweigh those associated with prophylaxis and that safe chemoprophylaxis regimens are available. Specific guidance for pregnant travelers is available from CDC's website at http://www.cdc.gov/travel/mal_preg_pub.htm. Signs and symptoms of malaria are often nonspecific, but fever is usually present. Other symptoms include headache, chills, increased sweating, back pain, myalgia, diarrhea, nausea, vomiting, and cough. Prompt diagnosis requires that malaria be included in the differential diagnosis of illness in a febrile person with a history of travel to a malarious area. Clinicians should ask all febrile patients for a travel history, including when evaluating febrile illnesses among international visitors, immigrants, refugees, migrant laborers, and international travelers. Prompt treatment of suspected malaria is essential, because persons with P. falciparum infection are at risk for experiencing life-threatening complications. Ideally, therapy for malaria should be initiated immediately after the diagnosis has been confirmed by a positive blood film. Treatment should be determined on the basis of the infecting Plasmodium species, the probable geographic origin of the parasite, the parasite density, and the patient's clinical status (14). If the diagnosis of malaria is suspected and cannot be confirmed, or if a diagnosis of malaria is confirmed but species determination is not possible, antimalarial treatment should be initiated that is effective against P. falciparum. Resistance of P. falciparum to chloroquine is worldwide, with the exception of a limited number of geographic regions (e.g., Central America). Therefore, therapy for presumed P. falciparum malaria should usually entail the use of a drug effective against such resistant strains. Health-care workers should be familiar with prevention, recognition, and treatment of malaria and are encouraged to consult appropriate sources (Table 7) for malaria treatment recommendations or call CDC's National Center for Infectious Diseases, Division of Parasitic Diseases, at 770-488-7788. Detailed recommendations for preventing malaria are available 24 hours/day from CDC at telephone 877-394-8747 (toll-free voice information system) or 888-232-3299 (toll-free facsimile request line), or on the Internet at http://www.cdc.gov/travel/diseases.htm/malaria. In addition, CDC biannually publishes recommendations in Health Information for International Travel (commonly referred to as The Yellow Book) (9), which is available for purchase from the Public Health Foundation at 877-252-1200 or 301-645-7773; it is also available and updated more frequently on CDC's Internet site at http://www.cdc.gov/travel. CDC provides support for the diagnosis of malaria through DPDx, a program that enhances diagnosis of parasitic diseases throughout the world. It includes an Internet site, http://www.dpd.cdc.gov/dpdx, that contains information regarding laboratory diagnosis, geographic distribution, clinical features, treatment, and life cycles of >100 parasite species. The DPDx Internet site is also a portal for diagnostic assistance for health-care providers through telediagnosis. Digital images captured from diagnostic specimens are submitted for diagnostic consultation through electronic mail. Because laboratories can transmit images to CDC and rapidly obtain answers to their inquiries, this system allows more efficient diagnosis of difficult cases and more rapid dissemination of information. Approximately 41 public health laboratories in 38 states and Puerto Rico have, or are in the process of acquiring, the hardware to perform telediagnosis. Acknowledgments The authors acknowledge the state, territorial, and local health departments; health-care providers; and laboratories for reporting this information to CDC. References
* To obtain confirmation diagnosis of blood films from questionable cases and to obtain appropriate treatment recommendations, contact either your state or local health department or CDC's National Center for Infectious Diseases, Division of Parasitic Diseases, Malaria Epidemiology Branch at 770-488-7788. Table 1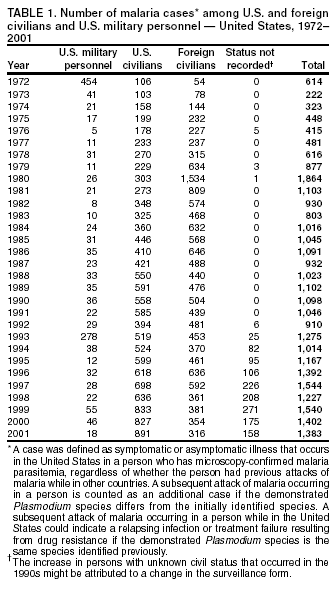 Return to top. Figure 1 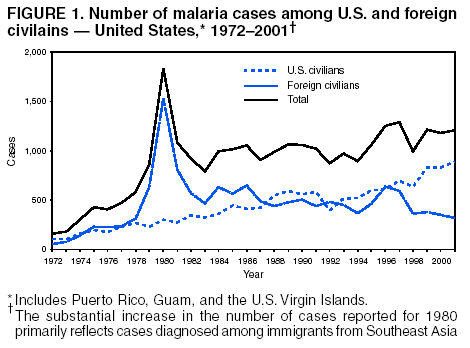 Return to top. Table 2 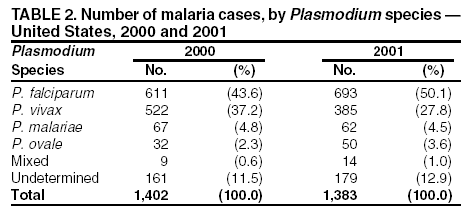 Return to top. Figure 2 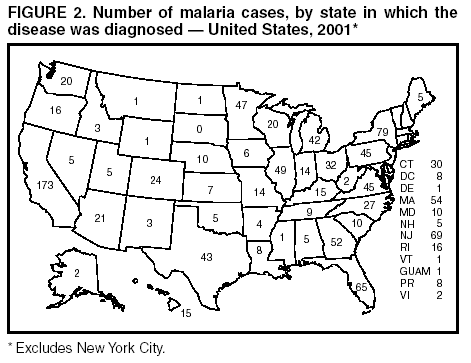 Return to top. Table 3 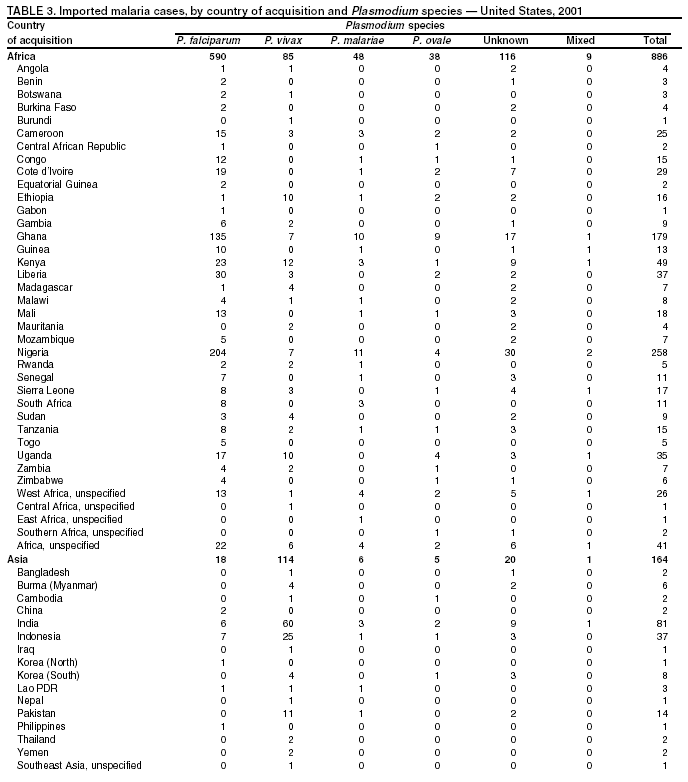 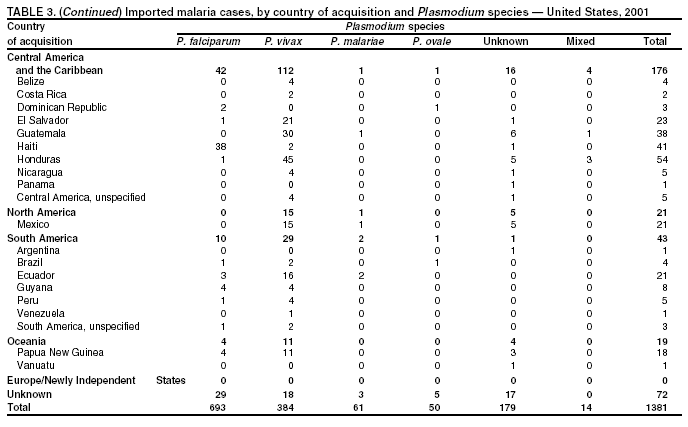 Return to top. Table 4 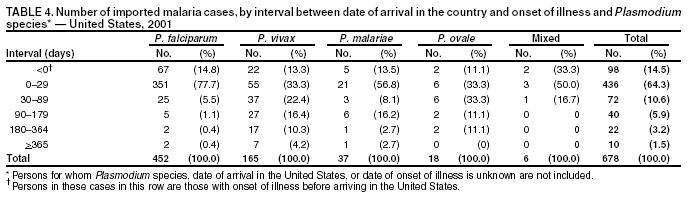 Return to top. Table 5  Return to top. Table 6 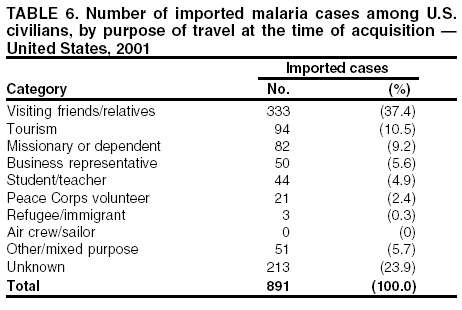 Return to top. Table 7  Return to top.
All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 7/7/2003 |
|||||||||
This page last reviewed 7/7/2003
|