Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Progressive Vaccinia in a Military Smallpox Vaccinee --- United States, 2009
Progressive vaccinia (PV), previously known as vaccinia necrosum, vaccinia gangrenosum, or disseminated vaccinia, is a rare, often fatal adverse event after vaccination with smallpox vaccine, which is made from live vaccinia virus (1). During recent vaccination programs potential cases of PV were investigated, but none met standard case definitions (2). PV has not been confirmed to have occurred in the United States since 1987 (3). On March 2, 2009, a U.S. Navy Hospital contacted the Poxvirus Program at CDC to report a possible case of PV in a male military smallpox vaccinee. The service member had been newly diagnosed with acute mylegenous leukemia M0 (AML M0). During evaluation for a chemotherapy-induced neutropenic fever, he was found to have an expanding and nonhealing painless vaccination site 6.5 weeks after receipt of smallpox vaccine. Clinical and laboratory investigation confirmed that the vaccinee met the Brighton Collaboration and CDC adverse event surveillance guideline case definition for PV (4,5). This report summarizes the patient's protracted clinical course and the military and civilian interagency governmental, academic, and industry public health contributions to his complex medical management. The quantities of investigational and licensed therapeutics and diagnostics used were greater than anticipated based on existing smallpox preparedness plans. To support future public health needs adequately, the estimated national supply of therapeutics and diagnostic resources required to care for smallpox vaccine adverse events should be reevaluated.
Case Description
On January 13, 2009, a healthy service member aged 20 years received a primary smallpox vaccination (ACAM2000 [Acambis, Inc., Cambridge, Massachusetts]) in accordance with the U.S. Department of Defense smallpox vaccination policy*; no other vaccinations were administered that day. Twelve days later, the patient visited a local hospital with fever and headache of 1 day's duration and was admitted for workup of leukopenia after his white blood cell count was found to be 1,400 cells/mm3. On January 28, after transfer to a U.S. Navy tertiary-care facility, he was diagnosed with AML M0. On January 30 and February 13, the patient underwent two successive rounds of induction chemotherapy with cytarabine, idarubicin, and dexamethasone. Before initial chemotherapy, the vaccination site pustule had a central crust and measured approximately 1 cm in diameter with minimal surrounding erythema. During the patient's hospital stay from the end of January to the beginning of March, his vaccination site dressing was changed daily.
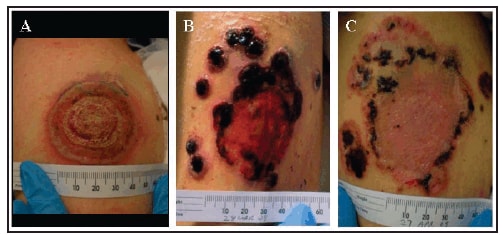
On March 2, during the evaluation of neutropenic fever, the failure of the patient's vaccination site to heal was described. An annular lesion with a deep bulla, raised violaceous leading edge, and a central crust that bled with pressure was noted. The size of the lesion had progressed to approximately 4 x 4 cm with minimal surrounding erythema or induration (Figure). The patient described no pain at the site, although he reported occasional pruritus. A swab of the lesion and serum were sent to CDC for viral and serologic analysis. Viral analysis of the swab by multiple real-time polymerase chain reaction (PCR) assays for orthopoxvirus and vaccinia yielded evidence of viral DNA; viral culture was positive for orthopoxvirus. Serum showed equivocal to absent levels of anti-orthopoxvirus immunoglobulin G (IgG) and immunoglobulin M (IgM) by enzyme-linked immunosorbent assay. The results of the diagnostic testing combined with the patient's medical history met the PV level 1 case definition as defined by the Brighton Collaboration and the confirmed case definition as described by CDC surveillance guidelines (4,5). The criteria met by both case definitions were 1) a documented clinical diagnosis of a disease that is known to be associated with cell-mediated immunodeficiency (in this case AML M0), 2) the primary vaccination site's failure to resolve (in this case >6 weeks post vaccination), and 3) the laboratory confirmation of vaccinia virus as the causative agent.
On March 3, imiquimod was applied directly to the lesion. Within 24 hours of confirmation of PV on March 4, the patient received licensed Vaccinia Immune Globulin Intravenous (Human) (VIGIV) (Cangene Corporation, Winnipeg, Canada). On March 5 and March 6, oral and topical ST-246 (SIGA Technologies, Corvallis, Oregon) were administered under an Emergency Investigational New Drug (E-IND) application. The patient remained stable until the evening of March 7, when he became septic with Pseudomonas aeruginosa, likely from a perirectal abscess. He required intubation, maximal vasopressor support, multiple antibiotics, and stress dose corticosteroids. He then developed multiorgan failure and began continuous venovenous hemodialysis. During the next 12 days, the patient slowly stabilized. As a consequence of the duration and amount of vasopressor support, the patient required a bilateral trans-tibial amputation because of dry gangrene of his feet.
During March 6--19, the patient received additional oral and topical ST-246 and VIGIV; his ST-246 levels were noted to be lower than those achieved both in healthy subjects in phase I clinical trials and in successful treatment of nonhuman primates with systemic orthopoxvirus disease. The lesion size remained unchanged, but the central crust of the vaccination site sloughed off, followed by most of the outer "ring" flattening, leaving a shallow ulcer with healthy-appearing granulation tissue. During his steroid taper, additional satellite lesions surrounding the vaccination site appeared on March 18, and viral DNA was detected again in the blood. These lesions became vesicular in nature, and on March 26, after a second E-IND was issued, CMX001 (Chimerix, Inc., Research Triangle Park, North Carolina), a lipid conjugate of cidofovir, was administered.
From March 24 onward, the satellite and main vaccination site lesions continued to crust, the scabs separated, and underlying tissue epithelialized (Figure). Blood viral DNA levels cleared on March 29. On April 10, the borders of lesions again appeared raised; a shave biopsy grew methicillin-resistant Staphylococcus aureus, which responded to antibiotic therapy. The patient received intermittent granulocyte colony-stimulating factor, and his absolute neutrophil and lymphocyte count increased over time. By May 1, significant portions of the scabs/eschars had fallen off or were removed manually, revealing healthy epidermis. Numerous therapeutics with different biologic mechanisms were used to treat PV in this patient (Table).
From February 21 onward, the patient had remained in contact isolation, first for a Clostridium difficile infection and then for his progressive vaccinia infection. On May 5, contact precautions were discontinued because of the lack of viable virus in lesion specimens from the previous 4 weeks. No cases of contact vaccinia were identified among this patient's health-care workers or close contacts.
During March 3--May 18, nearly 200 clinical specimens (lesion and satellite swabs/crusts, ethylenediaminetetraacetic acid [EDTA] blood, bone marrow, and serum) were collected and submitted to CDC to evaluate disease progression and guide therapeutic interventions. After April 23, swabs from satellite lesions or the main vaccination site showed significantly reduced or absent levels of viral DNA, and no viable virus was detected after April 2. Oropharyngeal sampling and bone marrow biopsies from early and late March, respectively, were negative for vaccinia virus. Orthopoxvirus DNA was detected in EDTA blood at intermittent times during the course of the patient's infection; however, no viable virus was cultured from blood. As of May 12, the patient had no demonstrable IgM response to orthopoxvirus; IgG levels appeared fully reliant on VIGIV infusion.
During March 3--May 18, a total of 20 conference calls to discuss patient status and treatment options were held between the Vaccine Healthcare Centers Network, Military Vaccine Agency (MILVAX), Bureau of Medicine and Surgery of the Navy, CDC, Food and Drug Administration (FDA), National Institutes of Health (NIH), SIGA Technologies, Chimerix, Inc., and academic and health-care professionals. As of May 18, MILVAX provided 22 and the Strategic National Stockpile (SNS) provided 254 vials of VIGIV used in treatment of this case.
Reported by: E Lederman, MD, H Groff, MD, T Warkentien, MD, A Reese, MD, US Naval Medical Center. D Hruby, PhD, T Bolken, D Grosenbach, PhD, S Yan, PhD, SIGA Technologies, Corvallis, Oregon. W Painter, MD, L Trost, MD, B Lampert, MD, Chimerix, Inc., Research Triangle Park, North Carolina. J Cohen, MD, National Institutes of Health; R Engler, MD, Walter Reed Vaccine Healthcare Center; W Davidson, MPH, S Smith, MS, K Wilkins, Z Braden, Y Li, PhD, I Damon, MD, Div of Viral and Rickettsial Diseases, National Center for Zoonotic, Vector-Borne, and Enteric Diseases, CDC.
Editorial Note:
Although PV is a rare adverse event (one case per million during routine vaccination during 1963--1968), its case fatality rate in primary U.S. vaccinees was 15% despite treatment with massive amounts of VIG (intramuscular) (6). Extensive surgical debridement was sometimes required, even necessitating disarticulation of the arm to "debulk" the amount of infectious material (7). Before smallpox vaccination, patients are screened for numerous contraindications (8). At the time of his vaccination, the patient described in this report did not have any obvious signs or symptoms that would meet any exclusion criteria for vaccination. Training in use of, and careful adherence to, screening tools can identify vaccine candidates at risk for PV and other adverse events (2). Despite this, vaccinees with occult immunodeficiencies might not be recognized, and therefore appropriately deferring vaccination in these persons is not always possible.
Lack of inflammation at the expanding vaccination site is the hallmark of PV. Any smallpox vaccinee who has an expanding, nonhealing, painless vaccination site without inflammation for more than 2 weeks should be evaluated for an underlying immunodeficiency, and diagnosis of and treatment for PV should be considered. Health-care providers should report suspected cases of PV or other adverse events to the Vaccine Adverse Event Reporting System (VAERS).† Suspected cases of PV also should be reported to state health officials and CDC for clinical consultation and to obtain select therapeutics available only through the SNS. State health departments should call the CDC Emergency Operations Center at 770-488-7100.
This patient's protracted clinical course is consistent with previously published cases reports and surveillance summaries. The development of progressive vaccinia, historically observed in patients with cellular immunodeficiencies, often leads to superinfection and subsequent sepsis (i.e., fungal, parasitic, and bacterial infections resulting in toxic or septicemic shock, then ultimately death). Past treatment typically included massive doses of VIG, administration of thiosemicarbazone, blood products, and supportive care for accompanying infections (7,9). The improvement of progressive vaccinia in this patient was associated with receipt of VIGIV (the only licensed product for treatment of vaccinia adverse events stockpiled by the SNS), ST-246, and CMX001, and an increase in lymphocyte count. The use of two antiviral agents with different mechanisms of action§ was enabled by the research and development of medical countermeasures for smallpox preparedness activities, as well as the use of the emergency IND process. As of May 18, the patient had shed nearly all of the scab material on and around the vaccination site.
The rapid mobilization of military, CDC, FDA, NIH, drug manufacturer, and academic and health-care human resources to review the case's status and to provide daily, then biweekly laboratory findings that guided treatment recommendations, was enabled by smallpox public health preparedness research and training efforts. Future cases of PV likely will require similar intensive and multidisciplinary clinical consultation. Experts with background in vaccine safety, PV treatment, clinical virology, infectious disease, and immunodeficiencies should be engaged.
Continuing medical education and reinforcement of training related to the prevention, early recognition, and treatment of smallpox vaccine--related adverse events should be part of smallpox vaccination programs.¶ The patient described in this report received VIGIV in the amount originally estimated to treat 30 persons. The extraordinary amounts of VIGIV used to treat this single case of PV underscore the need to reevaluate the adequacy of the national stockpiled supply of this or other medical countermeasures (treatment or prophylactic). Such reevaluation, with additional focus on immunocompromised hosts, will aid in the smallpox vaccination program planning and overall smallpox preparedness efforts.
Acknowledgments
This report is based, in part, on contributions by JM Lane and staff members from SIGA Technologies, Corvallis, Oregon; Chimerix, Inc., Research Triangle Park, North Carolina; Center for Biologics Evaluation and Research and Center for Drug Evaluation and Research, Food and Drug Admin; Military Vaccine Agency; Walter Reed Vaccine Healthcare Center; Bur of Medicine and Surgery of the Navy; Office of the Chief of Naval Operations; Chemical Biological Medical Systems of the US Dept of Defense; and the Emergency Operations Center and Strategic National Stockpile, CDC.
References
- CDC. Recommendations for using smallpox vaccine in a pre-event vaccination program. Supplemental recommendations of the Advisory Committee on Immunization Practices (ACIP) and the Healthcare Infection Control Practices Advisory Committee (HICPAC). MMWR 2003;52(No. RR-7).
- Vellozzi C, Lane JM, Averhoff F, et al. Generalized vaccinia, progressive vaccinia, and eczema vaccinatum are rare following smallpox (vaccinia) vaccination: United States surveillance, 2003. Clin Infect Dis 2005;41:689--97.
- Redfield RR, Wright DC, James WD, Jones TS, Brown C, Burke DS. Disseminated vaccinia in a military recruit with human immunodeficiency virus (HIV) disease. N Engl J Med 1987;316:673--6.
- Nell P, Kohl KS, Graham PL, et al. Progressive vaccinia as an adverse event following exposure to vaccinia virus: case definition and guidelines of data collection, analysis, and presentation of immunization safety data. Vaccine 2007;25:5735--44.
- CDC. Surveillance guidelines for smallpox vaccine (vaccinia) adverse reactions. MMWR 2006;55:(No. RR-1).
- Aragón TJ, Ulrich S, Fernyak S, Rutherford GW. Risks of serious complications and death from smallpox vaccination: a systematic review of the United States experience, 1963--1968. BMC Public Health 2003;3:26.
- Maurer DM, Harrington B, Lane JM. Smallpox vaccine: contraindications, administration, and adverse reactions. Am Fam Physician 2003;68:889--96.
- Fulginiti VA, Papier A, Lane JM, Neff JM, Henderson DA. Smallpox vaccination: a review, part II. Adverse events. Clin Infect Dis 2003;37:251--71.
- Bray M, Wright ME. Progressive vaccinia. Clin Infect Dis 2003;36:766--74.
- Quenelle, DC, Prichard MN, Keith KA, et al. Synergistic efficacy of the combination of ST-246 with CMX001 against orthopoxviruses. Antimicrob Agents Chemother 2007;51:4118--24.