Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Reduction in Rotavirus After Vaccine Introduction --- United States, 2000--2009
Worldwide, rotavirus is the leading cause of severe acute diarrhea in children aged <5 years (1). In the United States, before introduction of a live, oral pentavalent rotavirus vaccine (RV5) in 2006, rotavirus caused an estimated 20 to 60 deaths, 55,000 to 70,000 hospitalizations, 205,000 to 272,000 emergency department visits, and 410,000 outpatient visits annually (2). Before 2000, rotavirus had a predictable winter-spring seasonality and geographic pattern in the United States, with activity beginning in the West census region during December--January, extending across the country, and ending in the Northeast region during May--June (3,4). A similar but less pronounced trend was observed during 2000--2006 (5). To characterize trends and compare the 2007--08 and 2008--09 rotavirus seasons with the prevaccine period 2000--2006, CDC analyzed data from the National Respiratory and Enteric Viruses Surveillance System (NREVSS). The results indicated that the 2007--08 and 2008--09 seasons were both shorter and later than the median during 2000--2006. The 2008--09 season had 15% more positive rotavirus test results than the 2007--08 season, but the number of positive test results during each season was substantially lower than the median observed during 2000--2006. Continued surveillance is needed to characterize the effect of routine childhood rotavirus vaccination on rotavirus disease in U.S. children.
NREVSS is a national, passive laboratory surveillance network that monitors temporal and geographic trends for various viral pathogens, including rotavirus. Participating laboratories provide weekly reports to CDC of the number of rotavirus antigen detection tests performed and positive results obtained using commercially available enzyme immunoassays, which have >90% sensitivity and specificity. A median of 67 laboratories (range: 62--72) contributed rotavirus testing data to NREVSS during July 2000--June 2009. Data were aggregated by surveillance week for three periods: the baseline prevaccine period July 2000--June 2006 and the 2007--08 and 2008--09 postvaccine rotavirus seasons. Using data from all NREVSS laboratories contributing testing results, these periods were examined to compare dates of rotavirus season onset and offset and peak incidence. Data from the transitional season (July 2006--June 2007) were excluded because rotavirus vaccine recommendations from the Advisory Committee on Immunization Practices (ACIP) were first published in August 2006. The onset of rotavirus season was defined as the first of 2 consecutive weeks during which the percentage of stool specimens testing positive for rotavirus was ≥10%, and offset was defined as the last of 2 consecutive weeks during which the percentage of stool specimens testing positive for rotavirus was ≥10%. To characterize national and regional changes in rotavirus test results, CDC examined the number of tests performed and the number of positive results obtained from 29 NREVSS laboratories that consistently reported ≥30 weeks of data per season during 2000--2009.
Compared with the median rotavirus season onset of December (surveillance week 50) during 2000--2006, onset of the 2007--08 rotavirus season in early March (week 9) was approximately 11 weeks later, and onset of the 2008--09 season in late January (week 4) was approximately 6 weeks later (Figure) (5,6). Durations of the 2007--08 and 2008--09 rotavirus seasons were 14 weeks and 17 weeks, respectively, compared with 26 weeks (range: 25--28) during 2000--2006. Peak activity was observed in late April (week 17) during the 2007--08 season and in March (week 11) during the 2008--09 season, compared with a median peak activity in early March (week 9) during 2000--2006. The peak percentage of positive rotavirus test results was 17% during the 2007--08 season and 25% during the 2008--09 season, compared with a median of 43% (range: 37--56%) during 2000--2006.
During the 2008--09 season, rotavirus activity onset in the Northeast, Midwest, and South regions occurred during weeks 48, 3, and 46, respectively, within 8 weeks of the median onsets noted for each region during 2000--2006 (weeks 4, 1, and 52, respectively). In contrast, the West region, which historically has had the earliest onset, had the latest onset during the 2008--09 season, beginning week 10, or 14 weeks later than the median onset for the West region (week 48) during 2000--2006.
Of the 29 laboratories included in the test result analyses, 10 were in the South region, nine in the West, eight in the Midwest, and two in the Northeast (Table). During 2000--2006, an annual median of 14,211 rotavirus tests (range: 11,844--17,060) were performed overall by these laboratories; similar total numbers of tests were performed during the 2007--08 season (14,532) and 2008--09 season (14,201). However, although the number of tests performed was similar, the number with positive results decreased 64%, from a median of 3,551 tests (range: 3,007--3,949) during 2000--2006 to 1,281 during 2007--08, and decreased 59% to 1,468 during 2008--09 (Table). Similarly, when compared with the median of 25% of total tests positive for rotavirus during 2000--2006, the percentage of rotavirus positive tests decreased 64% during the 2007--08 season to 9%, and decreased 60% during the 2008--09 season to 10%.
Among regions, laboratories in the South and Midwest had more positive test results during the 2008--09 season than during the 2007--08 season (529 versus 301 and 549 versus 380, respectively) (Table). In contrast, laboratories in the West had fewer positive test results during the 2008--09 season than during the 2007--08 season (356 versus 566) (Table). Laboratories in the Northeast had the same number of positive results (34) for both seasons. In all regions, the number of positive test results reported during the 2007--08 and 2008--09 seasons was lower than the median reported during 2000--2006, and the percentage of positive results was lower than the percentage reported during 2000--2006.
Reported by: National Respiratory and Enteric Virus Surveillance System. CA Panozzo, MPH, JE Tate, PhD, DC Payne, PhD, MM Cortese, MD, M Patel, MD, J Gentsch, PhD, U Parashar, MBBS, Div of Viral Diseases, National Center for Immunization and Respiratory Diseases. JE Cortes, MD, DH Esposito, MD, EIS officers, CDC.
Editorial Note:
Sentinel laboratory surveillance demonstrates a decline in U.S. rotavirus activity during the first two seasons after introduction of vaccination against rotavirus in 2006. During the 2007--08 season, onset of rotavirus activity was 11 weeks later than the median onset during 2000--2006, and the number of positive test results was 64% lower than the prevaccine period (5,6). Although the number of positive test results was somewhat greater and the rotavirus season was longer during 2008--09 compared with 2007--08, rotavirus activity during both seasons was substantially lower than that reported during 2000--2006. Regional variations in season onset, duration, and test results also were observed during 2008--09, particularly in the West, underscoring the importance of monitoring to assess trends after introduction of a vaccine.
This analysis suggests that RV5 vaccination might provide benefit to both vaccinated and unvaccinated persons by reducing overall rotavirus transmission (i.e., herd immunity). Although nationally representative data regarding rotavirus vaccination coverage are not available, CDC used data through December 2007 from six sentinel immunization sites to estimate 1-dose RV5 coverage at 58% for children aged 3 months and coverage with ≥1 doses at 31% for children aged <2 years (5). In contrast, the number of positive rotavirus test results was 64% less during 2007--08 than 2000--2006, more than double the estimated vaccination coverage of 31% for children aged <2 years (5). Other surveillance studies have described declines in rotavirus activity among older children not eligible for vaccination (7). Similar observations were reported recently from Australia, where rotavirus vaccination was introduced in 2007 (8). Indirect benefits for unvaccinated persons after introduction of vaccine also have been documented with other vaccine preventable diseases. For example, significant reductions of invasive pneumococcal disease in adults occurred after introduction in 2000 of a 7-valent pneumococcal conjugate vaccine for children (9).
Although an additional birth cohort was vaccinated during 2008--09, only laboratories in the West region reported a decrease in the number of positive rotavirus test results from 2007--08. Substantial season-to-season variations in incidence of rotavirus disease have been documented (10), and these variations should be considered in analyzing the differences in rotavirus activity between the 2007--08 and 2008--09 seasons. Rotavirus activity after vaccine introduction declined substantially during the 2007--08 season, compared with the prevaccine era 2000--2006, and this decline persisted for a second season after vaccine introduction. Further study is needed to characterize the contribution of routine rotavirus childhood vaccination, herd immunity, provider testing patterns, or other factors.
The findings in this report are subject to at least four limitations. First, NREVSS does not collect information on patient demographics or clinical data, precluding examination of trends in rotavirus detection by key factors such as age and vaccination status. Second, because patient-level information is unavailable, NREVSS might receive more than one result for a given patient. However, the likelihood of duplicate results was similar in the prevaccine and postvaccine periods and should not have affected the substantial decline observed in positive test results. Third, because the results come from a small, selected group of laboratories, they might not reflect national and regional trends in rotavirus detection. Finally, testing patterns of health-care providers for rotavirus were not assessed; possible changes in testing practices after vaccine introduction might have affected these findings.
The incidence of rotavirus disease has declined since introduction of rotavirus vaccination for children in the United States. Continued monitoring of rotavirus seasons and epidemiologic studies to examine patient age, vaccination status, and other characteristics of rotavirus cases, will enhance understanding of the effect of vaccination on rotavirus disease in U.S. children.
References
- Parashar UD, Gibson CJ, Bresse JS, Glass RI. Rotavirus and severe childhood diarrhea. Emerg Infect Dis 2006;12:304--6.
- CDC. Prevention of rotavirus gastroenteritis among infants and children: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 2009;58(No. RR-2).
- Turcios RM, Curns AT, Holman RC, et al. Temporal and geographic trends of rotavirus activity in the United States, 1997--2004. Pediatr Infect Dis J 2006;25:451--4.
- Torok TJ, Kilgore PE, Clarke MJ, Holman RC, Bresee JS, Glass RI. Visualizing geographic and temporal trends in rotavirus activity in the United States, 1991 to 1996. National Respiratory and Enteric Virus Surveillance System Collaborating Laboratories. Pediatr Infect Dis J 1997;16:941--6.
- Tate JE, Panozzo CA, Payne DC, et al. Decline and change in seasonality of U.S. rotavirus activity after the introduction of rotavirus vaccine. Pediatrics 2009;124:465--71.
- CDC. Delayed onset and diminished magnitude of rotavirus activity---United States, November 2007--May 2008. MMWR 2008;57:697--700.
- Payne DC, Edwards KM, Staat MA, et al. Decline in rotavirus hospitalizations in three U.S. counties after introduction of rotavirus vaccine. Presented at the Pediatric Academic Societies Conference, Baltimore, MD; May 2--5, 2009.
- Lambert SB, Faux CE, Hall L, et al. Early evidence for direct and indirect effects of the infant rotavirus vaccine program in Queensland. Med J Aust 2009;191:157--60.
- Whitney CG, Farley MM, Hadler J, et al. Decline in invasive pneumococcal disease after the introduction of protein-polysaccharide conjugate vaccine. N Engl J Med 2003;348:1737--46.
- Payne DC, Szilagyi P, Staat MA, et al. Secular variation in U.S. rotavirus disease rates and serotypes. Implications for assessing the rotavirus vaccination program. Pediatr Infect Dis J 2009. In press.
|
What is already known on this topic? Before introduction of a vaccine in 2006, rotavirus caused an estimated 20 to 60 deaths, 55,000 to 70,000 hospitalizations, and 205,000 to 272,000 emergency department visits in the United States each year. What is added by this report? The 2007--08 and 2008--09 rotavirus seasons were shorter, later, and characterized by substantially fewer positive rotavirus test results, compared with median data for 2000--2006 from a national network of sentinel laboratories. What are the implications for public health practice? Continued monitoring is needed to enhance understanding of the effect of vaccination on rotavirus disease among those who are vaccinated and unvaccinated. |
FIGURE. Percentage of rotavirus tests with positive results, by surveillance week --- participating laboratories, National Respiratory and Enteric Virus Surveillance System (NREVSS), United States, July 2000--June 2009*
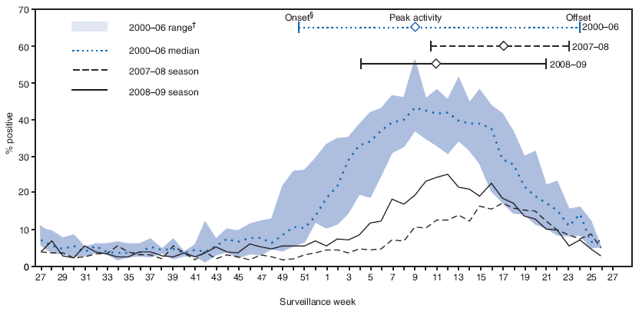
* A median of 67 laboratories (range: 62--72) contributed rotavirus testing data to NREVSS during July 2000--June 2009.
† Range created using the maximum or minimum percentage of rotavirus positive tests for each surveillance week during 2000--2006. Maximums and minimums for each week might have occurred during any of the six rotavirus seasons during 2000--2006.
§ The onset of rotavirus season was defined as the first of 2 consecutive weeks during which the percentage of stool specimens testing positive for rotavirus was ≥10%; offset was defined as the last of 2 consecutive weeks during which the percentage of stool specimens testing positive for rotavirus was ≥10%.
Alternative Text: The figure above shows the percentage of rotavirus tests with positive results, by surveillance week- from participating laboratories- derived from the National Respiratory and Enteric Virus Surveillance System for July 2000 through June 2009. Compared with the median rotavirus season onset of December (surveillance week 50) during 2000-2006, onset of the 2007-08 rotavirus season in early March (week 9) was approximately 11 weeks later, and onset of the 2008-09 season in late January (week 4) was approximately 6 weeks later.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services. |
All MMWR HTML versions of articles are electronic conversions from typeset documents. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 10/21/2009


