 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Use of Mass Tdap Vaccination to Control an Outbreak of Pertussis in a High School --- Cook County, Illinois, September 2006--January 2007On September 6, 2006, the Cook County Department of Public Health (CCDPH) was notified that a local high school student aged 17 years had pertussis. During September 2006--January 2007, 36 pertussis cases directly linked to the high school were identified. Because Bordetella pertussis immunity from childhood vaccinations wanes over time, outbreaks of pertussis can periodically occur among students and staff at middle and high schools. School settings facilitate transmission of pertussis, disrupting school and community activities and putting vulnerable populations, such as unvaccinated infants, at risk (1--4). A pertussis booster vaccine suitable for adolescents and adults became available in the United States in 2005, when two new tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis (Tdap) vaccines were licensed for persons aged 10--18 years and 11--64 years, respectively. In 2006, the Advisory Committee on Immunization Practices (ACIP) recommended that all adolescents and adults receive a one-time Tdap booster vaccination (5,6). This report summarizes strategies used to control the pertussis outbreak in Cook County, Illinois, including efforts to increase Tdap vaccination coverage. Despite multiple communications recommending Tdap vaccination and implementation of a cough exclusion policy during the pertussis outbreak, student vaccination rates did not increase substantially until a school-based Tdap vaccination clinic was implemented. Because persons at risk for pertussis might not seek vaccination from their usual health-care provider, even during an outbreak, local health departments might consider early implementation of a cough exclusion policy and on-site Tdap vaccination clinic as control measures. At the time of the pertussis outbreak, the high school in Cook County had 4,154 students and 651 staff members on two campuses. The index patient at the school was a symptomatic student epidemiologically linked to the primary patient, involving a younger sibling who had cough onset August 10 and was confirmed to have pertussis by polymerase chain reaction (PCR). Both cases were reported to the school by the siblings' physician on September 6. On the day the index case was reported, CCDPH responded by sending a letter to parents of 12th-grade students and to teachers at the high school, urging them to seek medical care for any cough illness consistent with pertussis. The letters also recommended that eligible persons receive Tdap vaccination. An informational letter and a copy of the parent letter were faxed to 31 physician practices identified by school nurses as providing medical care for students at the school. The physician letter reviewed the recent ACIP recommendations for Tdap vaccine administration to adolescents and adults, and included guidelines for diagnosis, treatment, and chemoprophylaxis. A separate letter with similar information was given to staff members to take to their physicians. For this outbreak, all four probable cases met standard CDC clinical criteria (i.e., a cough illness lasting at least 2 weeks with one of the following: paroxysms of coughing, inspiratory "whoop," or post-tussive vomiting, without other apparent cause, as reported by a health professional). The 32 confirmed cases had either 1) laboratory confirmation by a positive PCR test result for B. pertussis from a nasopharyngeal specimen, or 2) an epidemiologic link to a laboratory-confirmed case (4). At the time of this outbreak, the Illinois Department of Public Health laboratory used a single-tier PCR test for laboratory confirmation of pertussis cases; culture was not performed. By October 31, approximately 6 weeks into the outbreak, 10 cases of pertussis had been diagnosed at the high school. At that point, active surveillance for cough illness was begun. On November 1, the 31 physician offices were telephoned by CCDPH to ensure physicians had Tdap vaccine on hand, were aware of plans to exclude students for cough illness, and that those students would need a note from a physician for clearance to return to school. An update letter regarding the outbreak also was faxed to the physician offices. A notice was sent to all parents and faculty on Friday, November 3, stating that students and staff with "persistent cough in the absence of an apparent cause" would be excluded from school and extracurricular activities until they could be evaluated by a physician. This notification emphasized the importance of all eligible students and staff members receiving Tdap vaccination. Teachers were responsible for identifying students exhibiting symptoms and sending them to school nurses to determine whether further medical assessment and exclusion were warranted. Students were given a form to be completed by their physician and then submitted to the school nurses as documentation. During the first week the policy was in force (November 6--10), 159 students (3.8% of the student body) were excluded from school for cough illness. The number of students with cough illness arriving at school in subsequent weeks declined substantially. Several of the larger physician practices sent direct mailings to the parents of their patients who were students at the school, urging that those children be brought in for Tdap vaccination. Over time, however, these practices and others reported that few students from the school had come to their offices for vaccination. In addition, a national shortage of the adult formulation of Tdap proved to be a substantial barrier to school faculty seeking vaccination. On November 16, CCDPH asked that school administrators anonymously survey 11th- and 12th-grade students and school staff members via e-mail to obtain a rough estimate of Tdap vaccination coverage. The overall response rate was 63.3%. The survey indicated that approximately 30% of students and 17% of staff members had been vaccinated. Sixteen additional pertussis cases (three probable and 13 laboratory confirmed) at the school were diagnosed during November 6--December 1. During September 6--November 22, CCDPH and school administrators sent a series of 11 letters* to parents urging Tdap vaccination, but many persons at risk for exposure failed to obtain Tdap vaccinations. Faced with ongoing transmission within the school, CCDPH elected to hold a voluntary Tdap vaccination clinic at the school. The clinic was held December 5--8, immediately before a 2-week winter break. Students and staff members were eligible to receive Tdap vaccination if they had not received a Td-containing vaccination (i.e., tetanus and diphtheria toxoids) in the preceding 2 years. Students were required to present a signed parental consent form. Over the 4-day period, 1,084 students (26.1% of the student body) and 416 staff members (63.9% of all staff members) received Tdap. Cook County government incurred all costs of the student vaccination clinic. CCDPH staff vaccinated the students, and local medical practices sent nurses and donated supplies to vaccinate the high school staff on-site, using Tdap vaccine provided by CCDPH. During December 5--8, all students were required to submit documentation of their Tdap immunization status, including date of vaccination. However, Tdap vaccination was not required for school attendance, and students were not excluded from school if they did not receive vaccination. School nurses entered the vaccination information into an electronic database managed by the school. CCDPH then reviewed the data to evaluate the effect of public health recommendations on vaccination rates. The overall pre-outbreak Tdap vaccination rate among students was 16.4%. Tdap coverage after the mass vaccination clinic ranged from 65.0% among 10th-grade students to 71.0% among 9th-grade students (Table, Figure). At the end of the vaccination campaign, 1,331 students (32% of the student body) had not received Tdap vaccination. Of students who did not receive vaccination, 558 (42%) were not eligible because they had received Td-containing vaccine within the preceding 2 years. The majority (81%) of those students were in the 9th- or 10th-grade classes. An additional 66 students were exempted from vaccination for various reasons. Ultimately, 707 (20%) of eligible students did not receive vaccination. The final two cases of pertussis were diagnosed on December 12 and December 19 in students who received Tdap at the school clinic. Both students had onset of illness 5 days after vaccination, which likely indicated that the infections occurred before immunity had developed. In all, 36 cases were identified in 33 students, one teacher, and two family members. None of the persons with pertussis required hospitalization. Of the 36 cases, four (11.1%) were probable, 29 (80.6%) were confirmed by PCR, and three (8.3%) were confirmed by epidemiologic link. Among confirmed cases, mean time to diagnosis after cough onset was 18.3 days (range: 1--58 days) before the cough exclusion policy was implemented, and 4.6 days (range: 1--14 days) after the policy was implemented (p<0.001, unpaired t-test). Overall, the 36 persons who became ill included four of 1,050 9th-grade students (attack rate [AR] = 0.4%), 12 of 1,030 10th-grade students (AR = 1.2%), 12 of 1,055 11th-grade students (AR = 1.1%), seven of 1,018 12th-grade students (AR = 0.7%), and one of 651 staff members (AR = 0.2%). Reported by: C Counard, MD, D Nimke, MPH, M Vernon, DrPH, Cook County Dept of Public Health, Oak Park, Illinois. A Cohn, MD, Div of Bacterial Diseases, National Center for Immunization and Respiratory Diseases, CDC. Editorial Note:Because a pertussis vaccine suitable for adolescents and adults was not available until 2005, pertussis outbreak control measures historically relied on rapid identification of cases for treatment and chemoprophylaxis of close contacts (4). In the Cook County outbreak, pertussis spread quickly to all grades within the high school, making this control strategy difficult to implement; only seven (19.4%) of the 36 cases had a clear epidemiologic link to another case. Strict enforcement of exclusion for cough illness was likely an important factor in controlling the outbreak. This measure limited exposure to persons with respiratory illness within the school, encouraged timely medical evaluation and treatment of cases, and promoted prompt administration of chemoprophylactics to close contacts. The time between cough onset and diagnosis for cases was reduced significantly after implementation of the policy. The exclusion measure began on November 6, after 10 cases had been reported. Additional study is needed to evaluate the point when application of more aggressive control measures, such as exclusion for cough illness or mass Tdap vaccination clinics, might be warranted to control an outbreak. Public health messages alone, particularly regarding the need for vaccination during the outbreak, had some effect on student vaccination rates. During the first 13 weeks after the first notices to parents and area physicians from CCDPH, Tdap vaccination coverage increased 5.8%. Before the on-site clinic at the school, Tdap vaccination coverage of students overall did not exceed 50%, even after the strict cough exclusion policy was adopted. After the on-site vaccination clinic, coverage increased another 30.8%. Which barriers prevented an earlier, more substantial increase in Tdap vaccination rates is unclear; however, the convenience of an on-site school clinic versus scheduling an appointment in a private physician's office might have played a role. Another barrier was the limited supply of Tdap vaccine for adults. Additionally, physician concern about the 5- and 10-year intervals recommended between Td-containing vaccines might have contributed to less compliance with vaccination early on in the outbreak. Tdap is recommended 5 years after Td vaccination in adolescents and after 10 years for adults. Shorter intervals between administration of vaccine doses containing tetanus and diphtheria toxoids have been associated with moderate to severe local reactions. However, clinicians may administer Tdap at an interval as short as 2 years from the last Td vaccination during outbreaks or other instances when risk for infection is a concern (5,6). CCDPH initially received many calls from area physicians requesting a reference for administering Tdap within a shorter interval and outside of typical prescribing practices. In response, CCDPH faxed portions of relevant reports (5,6) to those physicians. Although the effect of the Tdap vaccination clinic in shortening the duration of the outbreak is unclear, this experience shows that school-based Tdap vaccination clinics can quickly achieve high coverage during a pertussis outbreak. More experience with large Tdap vaccination clinics as part of the response to school pertussis outbreaks is needed to develop new recommendations for outbreak control. Preventing outbreaks of pertussis by increasing routine Tdap vaccination rates remains an important public health goal. As an initial step to prevent pertussis outbreaks, health-care providers, public health officials, and schools should promote routine Tdap vaccination before outbreaks occur. Acknowledgments This report is based, in part, on contributions from S Martin, PhD, S Martell, K Loewy, Cook County Dept of Public Health; C Conover, MD, K McMahon, Illinois Dept of Public Health; the high school administration and nursing staff; and the Illinois Dept of Public Health Laboratories. References
* All letters to parents urged vaccination and contained an update about the outbreak. Later letters discussed the need for the cough exclusion policy, and the results of the survey showing that few persons at risk were receiving vaccination. The letters were faxed and e-mailed. Parents quickly responded to the letters with details about the vaccination clinics once those were distributed. A separate survey conducted by CDC after the outbreak indicated that parents thought they had received sufficient information. Figure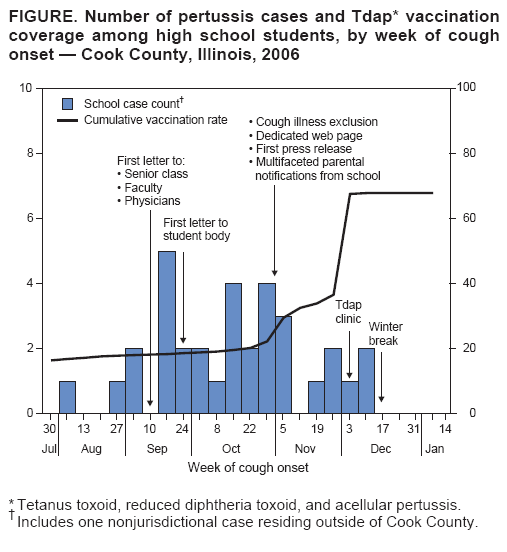
Return to top. Table 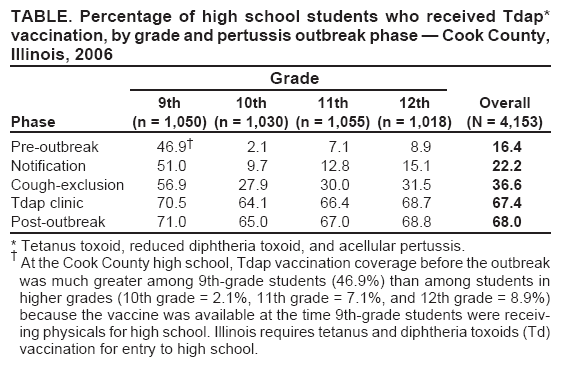 Return to top.
All MMWR HTML versions of articles are electronic conversions from typeset documents. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 7/23/2008 |
|||||||||
|