 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Progress in Measles Control --- Nepal, 2000--2006In 2002, the United Nations General Assembly Special Session on Children set a goal to reduce global measles deaths by half (compared with 1999) by 2005 (1). Nepal, a southeast Asian country with an estimated population of 27 million, adopted the measles mortality reduction strategies of the World Health Organization (WHO) (2) in 2003, with a goal of reducing measles deaths by half (compared with 2003) by 2005. The strategies consisted of strengthening routine childhood immunization programs, providing a second opportunity for measles vaccination through supplementary immunization activities (SIAs),* improving surveillance, and improving measles case management. This report describes routine immunization activities in Nepal, the implementation of measles SIAs, and measles surveillance data for the period 2000--2006. The findings demonstrate a substantial decrease in reported measles incidence. Assuming a reduction in measles deaths that paralleled the decrease in incidence, the findings also suggest progress toward the goal of measles mortality reduction. Background and Routine VaccinationNepal is divided into 75 districts in five regions. The Expanded Programme on Immunization (EPI) was initiated in 1979 in Nepal in three districts; by 1988, the program had been expanded to all 75 districts (3). The program aims to achieve and maintain coverage of >90% fully immunized† children nationwide by 2010 (4) and targets children aged >9 months with measles vaccine. According to WHO/UNICEF estimates, measles vaccination coverage among children aged <1 year increased from 58% in 1988 to 71% in 2000; coverage further increased from 75% in 2003 to 85% in 2006 (5). Despite high national coverage in 2006, six of 75 districts (representing 4% of the population aged <5 years) were unable to reach >70% coverage because of lack of security resulting from civil unrest, limited access to certain areas, or lack of human resources. SurveillanceMeasles in Nepal is reported as part of the Health Management Information System (HMIS), which covers all 4,102 government health facilities in Nepal. However, HMIS does not provide detailed geographic and age group data, and reports often are incomplete and not timely; moreover, HMIS reports only clinically suspected measles and does not report laboratory testing. Information on measles-related deaths is not reported systematically. In March 2003, the government of Nepal and WHO initiated a more comprehensive measles surveillance system to supplement HMIS with more detailed information on cases in clusters of suspected measles. The new measles surveillance system, which includes field investigations and laboratory testing of blood specimens, is supported by surveillance medical officers (SMOs), who have conducted health facility visits for active acute flaccid paralysis (AFP) surveillance since 1998. This integrated surveillance network provides timely and detailed data on AFP, Japanese encephalitis, and measles cases though weekly reports from 413 major health-care centers and hospitals throughout all 75 districts of the country (i.e., approximately 10% of all government health facilities), including all inpatient facilities. In addition, SMOs conduct weekly visits to 84 active surveillance sites within this network. If five or more cases of suspected measles are detected during a 2-week period from one geographic area, an outbreak investigation is undertaken in which epidemiologic information is collected on all suspected measles cases§ in the area, and blood samples are drawn for at least five cases for laboratory confirmation of measles (i.e., via identification of immunoglobulin M [IgM] measles antibodies). An outbreak is considered a confirmed measles outbreak if at least one case is laboratory confirmed in a person who had not received measles vaccination 1 month before. All untested suspected cases in a laboratory-confirmed outbreak are considered epidemiologically confirmed. Since January 2004, all samples that test negative for measles IgM have been tested for rubella IgM. With rubella IgM testing, similar criteria allow an outbreak to be considered a confirmed rubella outbreak or a confirmed mixed measles and rubella outbreak. Approximately 90% of cases associated with confirmed measles outbreaks in 2003 were in children aged <15 years; this finding supported the decision to conduct a "catch-up" SIA¶ targeting children aged 9 months--15 years. Measles Vaccination Campaign, 2004--2005Nepal public health authorities conducted a nationwide measles SIA in three phases during September 2004--April 2005, targeting an estimated 9.4 million children aged 9 months--15 years. Oral poliovirus vaccine also was administered to all children aged <5 years. The overall reported measles vaccination coverage was 105% of the population target; in one district the coverage was as low as 64%. The population targets were obtained from administrative lists. Measles IncidenceIn 2003, a total of 67 suspected measles outbreaks were investigated using the integrated system; in 2004, a total of 196 outbreaks were investigated. Nearly 70% of these outbreaks were confirmed measles outbreaks (Table). After the start of the SIAs, the number of suspected measles outbreaks detected decreased to 46 in 2005 and to 31 in 2006. In 2005, only one (2%) of the 46 investigated outbreaks was a laboratory-confirmed measles outbreak, whereas 36 (78%) were laboratory-confirmed rubella outbreaks. Similarly, in 2006, two (6%) of 31 outbreaks were laboratory-confirmed measles outbreaks, and 24 (77%) were laboratory-confirmed rubella outbreaks. During 2005 and 2006, three mixed measles and rubella outbreaks were detected: two (4%) in 2005 and one (3%) in 2006. The number of measles cases associated with outbreaks decreased from approximately 1,000 in 2003 to approximately 50 in 2006. During 2005 and 2006, a total of 1,119 suspected measles cases that were not part of any recognized outbreak were reported to SMOs. Serum specimens were collected for 84 of these cases; three (4%) were laboratory confirmed as measles cases. The average annual number of measles cases reported through HMIS during the 4 years (2000--2003) before the start of the SIA was 10,425. After the SIA, the number of reported cases decreased to 3,931 in 2005 and to 1,935 in 2006, decreases of 62% and 81%, respectively, from the 2000--2003 average (Table, Figure). Reported by: YV Pradhan, MD, Dept of Health Svcs, Nepal Ministry of Health and Population; P Bangdel, MSc, UNICEF Country Office, Kathmandu; T Sedai, MA, B Lamichhane, MBBS, MPH, J Partridge, PhD, World Health Organization Country Office, Kathmandu, Nepal. J Liyanage, MBBS, MPH, World Health Organization Regional Office for Southeast Asia, New Delhi, India. P Strebel, MBChB, A Dabbagh, PhD, Dept of Immunization, Vaccines, and Biologicals, World Health Organization, Geneva, Switzerland. O Mach, MD, V Dietz, MD, Global Immunization Div, National Center for Immunization and Respiratory Diseases, CDC. Editorial Note:Because information on measles-related deaths is not routinely collected in Nepal, no direct measurement of reduction in deaths associated with improved measles control is possible. However, reports from other countries have assumed that a reduction in measles deaths occurred in the same proportion as a reduction in reported measles cases (6,7). A concomitant decrease in suspected cases and measles deaths has been observed in other countries that monitored measles deaths before and after SIAs (8,9). By making this same assumption for Nepal, the findings in this report suggest that, by the end of 2005, Nepal had achieved its goal of reducing measles mortality by at least 50% from 2003 levels. The reduction in measles incidence in Nepal during 2003--2006 indicated by HMIS data might underestimate the actual relative reduction in measles deaths because, compared with pre-SIA years, a more pronounced decrease occurred in the number of confirmed outbreaks and in the proportion of confirmed measles cases in outbreaks during post-SIA years. In addition, treatment of measles patients** has been emphasized since 2003. On the basis of progress to date, the government of Nepal has decided to set its measles program objective toward elimination. In the Ministry of Health and Population's Multi-Year Plan of Action for immunization, the measles elimination phase will begin in 2010 (4). Major components of the elimination strategy include high routine immunization coverage (>90% in >80% of districts), provision of a second opportunity for measles vaccination through routine vaccination or SIAs, and case-based surveillance with laboratory confirmation. The integration of measles surveillance and AFP surveillance since 2003 has made use of the extensive surveillance infrastructure in Nepal, which was developed for AFP surveillance and, since 2004, has included investigation and laboratory testing of suspected encephalitis cases for Japanese B encephalitis. WHO formally accredited the Nepal national measles reference laboratory in 2006. The first steps toward further strengthening surveillance began in January 2007; a case-based measles surveillance system, in which all suspected measles cases are investigated and laboratory tested for IgM, was started in 12 active surveillance sites in the Kathmandu Valley and in two active surveillance sites in the Far West Development Region. In addition to continuing outbreak investigations, this case-based surveillance system will expand to include the entire country by 2010 and will use measles virus genotyping to determine the origin of virus isolates; however, data on measles-related mortality are not available through this system. Additional measures to increase routine vaccination coverage, particularly in remote areas and those with low coverage, will be critical for preventing outbreaks and moving toward the goal of measles elimination. Despite advances in delivering routine vaccination, the proportion of children susceptible to measles started to increase after the 2004--2005 SIA, increasing the likelihood of measles outbreaks. A nationwide follow-up measles vaccination campaign†† targeting children aged 9 months to 4 years 11 months is planned for 2008. Given the difficulties with access to certain areas of Nepal, providing a second measles vaccination opportunity through routine vaccination is not likely to reach high coverage levels with both doses. Because SIAs have been effective throughout Nepal, including in areas that are difficult to access, repeated SIAs likely will be the long-term strategy for regularly providing a second measles vaccination opportunity. Nepal has achieved a substantial reduction in reported measles incidence and in the number of confirmed measles outbreaks. This experience provides useful lessons for other countries in southeast Asia as they progress toward measles mortality reduction. References
* Mass campaigns conducted during a short period (days to weeks) in which a dose of measles-containing vaccine is administered to all children in a targeted age group (e.g., 9 months--15 years), regardless of previous vaccination history. Campaigns can be conducted nationally or in portions of the country. † A fully immunized child is a child who, by his or her first birthday, has received 1 dose of bacille Calmette-Guérin vaccine; 3 doses of diphtheria, tetanus, and pertussis vaccine; 3 doses of oral poliovirus vaccine; and 1 dose of measles-containing vaccine. § For surveillance purposes, a suspected case of measles is defined as generalized maculopapular rash and fever plus one of the following: cough, coryza (i.e., runny nose), or conjunctivitis (i.e., red eyes). Additional information available at http://www.afro.who.int/measles/guidelines/index.html. ¶ Catch-up campaigns are one-time events targeting all children in a particular age group. The goal is to vaccinate all children who might not have been previously vaccinated for the first time against measles and provide a second opportunity for measles vaccination in addition to routine vaccination. During a catch-up campaign, all children in the targeted age group receive a supplementary dose of measles vaccine, regardless of previous disease or vaccination history. Additional information is available at http://www.measlesinitiative.org/vaccination.asp. ** Treatment includes administration of vitamin A and, if complications are noted, antibiotics. Additional information available at http://www.who.int/mediacentre/factsheets/fs286/en. †† Follow-up SIAs are conducted periodically (i.e., every 3--5 years) to maintain low levels of susceptibility. A follow-up campaign provides children with a second opportunity for measles vaccination and aims to reach all children aged >9 months who were born after the previous catch-up campaign. Additional information available at http://www.measlesinitiative.org/vaccination.asp. Figure 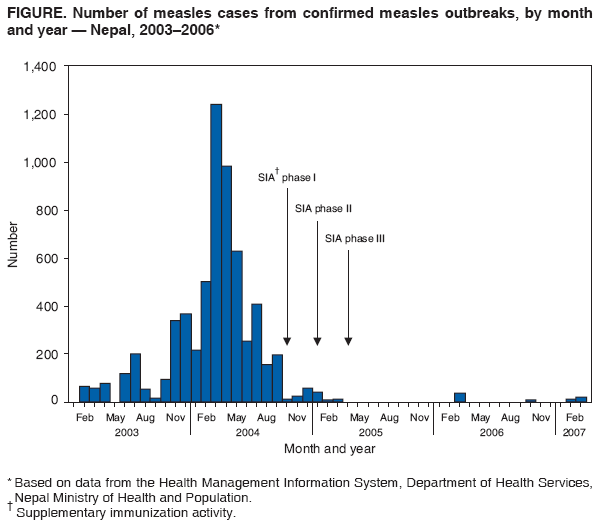 Return to top. Table 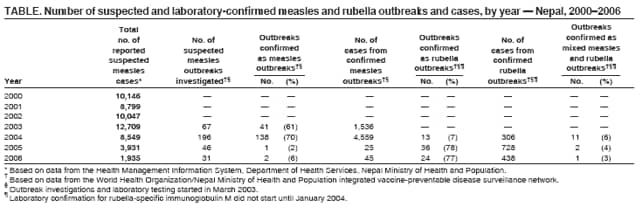 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 10/3/2007 |
|||||||||
|