 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Progress Toward Elimination of Haemophilus influenzae Type b Invasive Disease Among Infants and Children --- United States, 1998--2000Haemophilus influenzae type b (Hib) was the leading cause of bacterial meningitis and a major cause of other serious invasive diseases among children aged <5 years in the United States before Hib conjugate vaccines became available in 1988 (1,2). In 1991, all infants starting at age 2 months were recommended to receive Hib conjugate vaccines; by 1996, incidence of Hib invasive disease (i.e., illness clinically compatible with invasive disease, such as meningitis or sepsis, with isolation of the bacterium from a normally sterile site) among children aged <5 years had declined by >99% (1,3). This report presents 1998--2000 Haemophilus influenzae (Hi) surveillance data, which indicate that the incidence of reported Hib invasive disease remains low. Achieving the national health objective for 2010 of reducing to zero indigenous Hib invasive disease cases in children aged <5 years (4) will require improved age-appropriate vaccination of children, complete reporting of vaccination and relevant medical histories, standardization of the serotyping procedure, and complete ascertainment and reporting of serotype for all Hi invasive disease cases. In 1991, Hi invasive disease became a nationally notifiable disease. State health agencies, the District of Columbia, and New York City provide weekly reports of provisional cases of Hi invasive disease to CDC through the National Electronic Telecommunications System for Surveillance (NETSS) and the National Bacterial Meningitis and Bacteremia Reporting System (NBMBRS). Case reports include demographic data about persons with Hi invasive disease and supplemental information (e.g., the serotype that caused the illness, type of clinical illness, outcome, and Hib vaccination history). States were contacted to obtain and confirm supplemental data for cases of Hi invasive disease in children aged <5 years with onset in 1998, 1999, and 2000. Only Hib vaccine doses given >14 days before illness onset were considered valid. Annual population estimates for 1998 and 1999 from the U.S. Census Bureau were used to calculate incidence rates. CDC also coordinates the Active Bacterial Core surveillance (ABCs) system with sites in selected states. Illnesses identified as Hi invasive disease (i.e., isolation of H. influenzae from a normally sterile site in a resident of the surveillance area) are reported to CDC and the various state health departments (3). During 1998--2000, project personnel contacted all microbiology laboratories serving acute care hospitals in each surveillance area every 2--4 weeks; specimens were sent to CDC for serotype confirmation. The population of children aged <5 years in the surveillance areas increased from 750,534 in 1989 to 2,208,625 in 2000. In 1998, the surveillance area covered three counties in the San Francisco Bay Area, five counties in Tennessee, seven counties in New York, 20 counties in Georgia, and the entire states of Connecticut, Maryland, Minnesota, and Oregon. By January 2000, the surveillance area had increased to include 15 counties in New York, 11 in Tennessee, and all of Georgia. Rates were race-adjusted to the annual U.S. population estimates. During 1998--2000, a total of 824 Hi invasive disease cases was reported among children aged <5 years; rates were 1.4 per 100,000 children in 1998 and 1999 and 1.6 in 2000. Among children aged <5 years, serotype data were available for 219 (83%) of 265 cases in 1998, 214 (82%) of 262 cases in 1999 and 236 (79%) of 297 cases in 2000 (Figure 1). Of the 669 cases with known serotype, Hib accounted for 75 (34%) cases in 1998, 71 (33%) cases in 1999 and 51 (22%) cases in 2000; annual Hib invasive disease rates were 0.4, 0.4, and 0.3, respectively. Compared with the rate in 1990 (23 cases per 100,000), the average annual rate for 1998--2000 (0.3 cases per 100,000) represents a 99% decline. During the 3-year period, the annual average for reporting of serotype information was 81%, representing a 98% improvement from 1994 (Figure 1). By state, excluding Alaska, Hib invasive disease average annual incidence rates ranged from 0 to 2.1 per 100,000 children aged <5 years; in Alaska, the rate was 9.4 (Table 1). For nontype b Hi invasive disease, the average annual incidence rate by state ranged from 0 to 5.8 with a national average of 0.8 per 100,000 children aged <5 years (Table 1). For the 3-year period, the clinical outcome was known for 693 (84%) of the 824 Hi cases reported; 50 (7%) of the 693 patients died. Of 197 Hib cases reported, 169 (86%) had known outcome; 14 (8%) children died. By race/ethnicity, Hib invasive disease average annual incidence among children aged <5 years during 1998-2000 was 14.0 among American Indians/Alaska Natives, 1.0 among Hispanics, 0.9 among non-Hispanic whites, 0.6 among non-Hispanic blacks, and 0.4 among Asians/Pacific Islanders. Race/ethnicity data were missing for 10 (5%) Hib patients. During 1998--2000, of 197 Hib patients, 86 (44%) were aged <6 months and had not completed the 2- or 3-dose primary Hib vaccination series. Of the 111 (56%) children who were aged >6 months and eligible to have completed the primary series, 19 (17%) had unknown vaccination status, 31 (28%) were unvaccinated, 22 (20%) were undervaccinated, and 39 (35%) had completed a primary series, 21 of whom received a booster dose (given at 12--15 months). Among the 14 Hib invasive disease deaths reported, 11 (79%) patients aged <6 months were unvaccinated and three (21%) patients aged >6 months were undervaccinated. During 1998--2000, a total of 128 Hi invasive disease cases in children aged <5 years was reported from ABCs sites; 19 (15%) were caused by Hib, 95 (74%) by nontype b Hi, and 14 (11%) by unknown Hi serotypes. The annual race-adjusted incidence rates were 0.2, 0.6 and 0.2 per 100,000 children aged <5 years for Hib invasive disease compared with 1.8, 1.5 and 1.6 per 100,000 for nontype b Hi invasive disease in 1998, 1999, and 2000, respectively (Figure 2). Reported by: S Bath, MPH, K Bisgard, DVM, T Murphy, MD, Epidemiology and Surveillance Div, National Immunization Program; K Shutt, MPH, N Rosenstein, MD, Div of Bacterial and Mycotic Diseases, National Center for Infectious Diseases; C Ohuabunwo, MBBS, EIS Officer, CDC. Editorial Note:With widespread use of Hib conjugate vaccines beginning in 1990, the incidence of reported Hib invasive disease among children aged <5 years declined from an estimated 100 per 100,000 in the prevaccine era to a record low of 0.3 in 1996 (2,3). The findings in this report indicate that the incidence of invasive Hib disease remains low. During 1998--2000, although Hib remained an infrequent cause of invasive disease among children, illness and death occurred among infants aged <6 months who had not completed the 2- or 3-dose primary series of Hib vaccination and among unvaccinated or undervaccinated children; some of these cases might have been preventable. These data also suggest that primary or secondary vaccination failure occurs less frequently than failure to vaccinate. Understanding the reasons for Hib invasive disease among fully vaccinated children requires the reporting of full vaccination history (i.e., dates, dose, vaccine name, lot number, and manufacturer) and relevant medical histories (e.g., prematurity, immunosuppression, or other chronic diseases). Localized populations with low vaccination coverage contribute to the continued circulation of Hib despite sustained national Hib vaccination coverage of >90% (5). In Pennsylvania, during December 1999--February 2000, eight Hib invasive disease cases occurred in unvaccinated children aged <5 years, six of whom were from communities with lower Hib vaccination coverage and higher Hib carriage rates than other groups (6). As in the prevaccine era, Hib invasive disease rates among American Indian/Alaska Native children remain persistently higher than in the general U.S. population (7), which suggests that Hib elimination will require additional characterization of colonization and disease among these high-risk populations (7). Attaining and maintaining high Hib vaccination coverage at the community level should reduce the Hib carriage rate among young children by decreasing exposure of susceptible infants and interrupting Hib transmission (7). Because Hib vaccines protect against type b and not other Hi strains, serotyping of all Hi isolates from patients with invasive disease is necessary to monitor the vaccination program effectiveness and national progress towards Hib elimination. Serotype information is needed to measure the sensitivity of the surveillance system and to detect the emergence of invasive disease from nontype b Hi strains (8). The reporting of serotype information on Hi cases among children aged <5 years has improved; however, to ensure that all Hi isolates from children aged <5 years are serotyped and to minimize false-positive results (9), continued promotion and standardization of the serotyping procedure by states is essential. Because of inconsistencies in Hi serotyping (9), until December 2002, CDC requests that state health laboratories send all Hi isolates associated with invasive disease in children aged <5 years to CDC (telephone [404] 639-3158) for serotyping. The incidence of nontype b Hi invasive disease can be a useful indicator of the sensitivity of the surveillance system. Although Hib invasive disease in children aged <5 years declined to near-elimination levels during the last decade, the incidence of nontype b invasive disease from ABCs sites remained consistently >1 per 100,000 children aged <5 years. Adequate identification and reporting of nontype b Hi invasive disease might indicate sufficient sensitivity to readily identify cases of Hib invasive disease. States are encouraged to report invasive disease caused by all Hi strains as recommended by the Council of State and Territorial Epidemiologists and CDC (10). Public health efforts to achieve and document Hib invasive disease elimination in children aged <5 years will be advanced by 1) enhanced promotion of age-appropriate Hib vaccination at the community level, 2) complete reporting of vaccination and medical histories to characterize cases of Hib suspected to be vaccine failures, 3) standardization of the serotyping procedure, and 4) ascertainment and reporting of serotype for all Hi invasive disease cases in children. AcknowledgementsThis report is based on data contributed by state health departments to the National Notifiable Disease Surveillance System and by sites in the Active Bacterial Core surveillance (ABCs) system: L Gelling, MPH, P Daily, MPH, G Rothrock, MPH, A Reingold, MD, D Vugia, MD, State Epidemiologist, California Dept of Health Svcs. S Zansky, P Smith, MD, State Epidemiologist, New York State Health Dept. N Barrett, MS, JL Hadler, MD, State Epidemiologist, Connecticut State Dept of Health Svcs. W Baughman, MS, M Farley, MD, K McCombs, K Arnold, Georgia Dept of Human Resources, Div of Public Health. MA Pass, L Harrison, MD, J Roche, MD, State Epidemiologist, Maryland State Dept of Health and Mental Hygiene. J Rainbow, MPH, J Besser MS, R Lynfield, MD, R Danila PhD, H Hull MD, State Epidemiologist, Minnesota Dept of Health. KR Stefonek, MPH, PR Cieslak, MD, MA Kohn, MD, State Epidemiologist, Oregon Dept of Human Resources, State Health Div. W Schaffner, MD, B Barnes, Vanderbilt Univ, Nashville; A Craig, MD, State Epidemiologist, Tennessee Dept of Health. References
Table 1 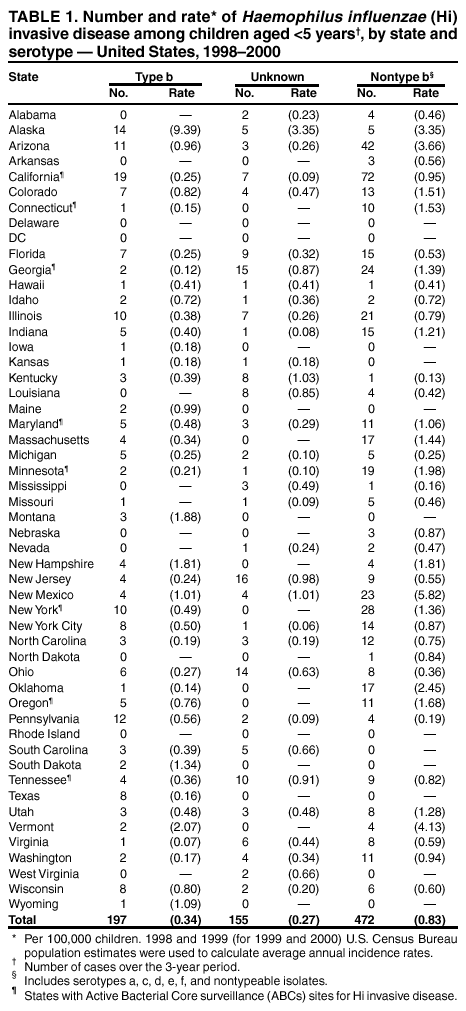 Return to top. Figure 1 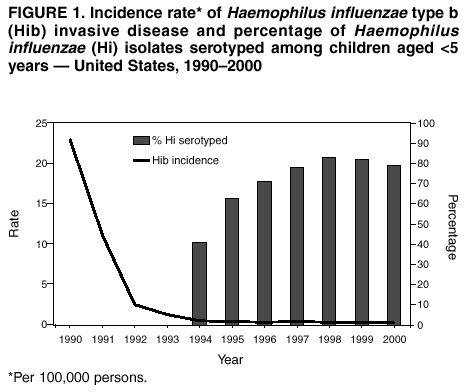 Return to top. Figure 2 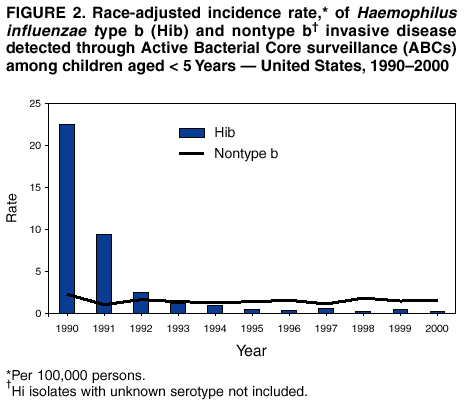 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 3/21/2002 |
|||||||||
This page last reviewed 3/21/2002
|