 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Gonorrhea Among Men Who Have Sex with Men -- Selected Sexually Transmitted Diseases Clinics, 1993-1996Among men who have sex with men (MSM), gonorrhea trends may reflect changes in sexual behaviors that also influence risk for human immunodeficiency virus (HIV) infection (1). Data from the Gonococcal Isolate Surveillance Project (GISP) were used to assess trends in gonococcal infection (GC) among MSM. For the subset of GISP sites where a substantial proportion of GC cases were in MSM, a special survey of the local areas was conducted to describe factors associated with GC in MSM. This report summarizes the results of that survey, which indicate that the number and proportion of MSM diagnosed with GC has increased in the sexually transmitted diseases (STD) clinics of several large cities in the United States. GISP is a sentinel surveillance project begun in 1987 to monitor antimicrobial resistance in Neisseria gonorrhoeae. Through GISP, STD clinics in 26 U.S. cities collect gonococcal isolates and clinical information, including sexual orientation, from the first 20 male patients with urethral GC examined each month. The sexual orientation of GISP participants examined from 1993 through 1996 was analyzed, and surveys were conducted of the clinics where greater than 5% of GISP isolates over the 4-year period were from MSM (n=8) *. The survey collected information from locally available data about absolute numbers of GC cases in MSM, in contrast to the proportions that were available from GISP data. The survey also addressed overall gonorrhea and other STD (i.e., nongonococcal urethritis {NGU}) trends and factors that could be associated with GC trends in MSM (i.e., changes in clinic practices, geographic clustering of cases, sexual risk behaviors, illicit drug use, and HIV serostatus). MSM comprised 5.0% of all cases in the GISP sample in 1993, a proportion similar to preceding years, and 8.7% in 1996 (p less than 0.001). Among the subset of eight GISP clinics where greater than 5% of isolates were obtained from MSM, the proportion increased from 12.0% (range: 4.2%-35.1%) in 1993 to 23.5% (range: 7.2%-57.1%) in 1996 (p less than 0.001) (Figure_1). The median age of MSM reported from all 26 clinics was 30 years and remained stable from 1993 through 1996. Changes in the race/ethnicity distribution were not statistically significant: from 1993 to 1996, the percentage of MSM who were white increased from 58.6% to 64.9%, and the percentage who were black decreased from 23.0% to 17.4%. Clinics in San Francisco; Seattle; and Portland, Oregon, experienced increases in both the absolute number and proportion of gonorrhea cases among MSM. From 1994 to 1996, GC among MSM at the Seattle clinic increased 125% (from 51 cases to 115 cases), while clinic visits by MSM increased by 17%. During the same period, the proportion of positive rectal and pharyngeal GC cultures among MSM at the Seattle clinic increased from 5.0% to 8.0% and from 1.5% to 6.7%, respectively. From 1994 to 1995 at the San Francisco clinic, the absolute number of MSM with GC increased 24% (from 271 to 336); similar increases occurred in the number of GC cases identified from a neighborhood known to have a high concentration of MSM and in the number of rectal GC cases. From 1995 to 1996 at the Portland clinic, the number of MSM with GC increased 124% (from 33 cases to 74 cases). Clinic visits by MSM during the same period increased, but the number of MSM seen by the clinic decreased, indicating that the increase in GC cases did not result from an increase in the number of MSM served by the clinic. Enhanced GC screening efforts targeted to MSM were initiated in San Francisco in early 1996, potentially contributing to an increase in reported cases in 1996; however, the absolute number of cases of GC in 1996 were not available at the time of this report. Changes in screening or outreach to MSM were not identified at other clinics in this survey. In addition, none of the clinics surveyed were aware of any changes in the availability of clinical services that might have prompted MSM to shift STD care-seeking from other venues to public STD clinics. From 1994 to 1996, NGU increased 27% among MSM at the Seattle clinic and 40% among MSM at the San Francisco clinic from 1993 to 1995. Approximately one fourth of MSM with GC identified at the Seattle and Portland clinics and one fourth of all MSM examined at the San Francisco clinic were HIV positive. Of the remaining five clinics surveyed, those in Honolulu and San Diego reported substantial increases in the number of MSM with GC. In San Diego, from 1996 to the first quarter of 1997, the proportion of rectal specimens that were positive for N. gonorrhoeae increased from 3.9% (seven of 180) to 14.6% (six of 41), and the proportion of male pharyngeal cultures that were positive increased from 2.9% (15 of 522) to 4.0% (five of 125). Based on five interviews of MSM with GC, four also were HIV infected. At the Denver clinic, the absolute number of MSM with GC decreased despite an increase in the proportion of GC cases identified in MSM from 1995 to 1996. In comparison with other clinics surveyed, the Denver clinic observed an increase in the number of black MSM examined from 1995 to 1996. The Long Beach and Orange County, California, clinics are investigating local trends in GC among MSM. Reported by: W Whittington, Univ of Washington; S Desmon, Seattle-King County Dept of Public Health, Seattle. C Kent, R Kohn, San Francisco Dept of Public Health, San Francisco; T Brazell, MD, San Diego Dept of Health Svcs, San Diego; JM Montes, STD Control Br, California Dept of Health Svcs. D Harger, V Fox, Oregon Health Div. R Ohye, Hawaii Dept of Health. FN Judson, MD, Denver Dept of Health, Denver. Gonorrhea, Chlamydia, and Chancroid Br, Div of AIDS, STD, and TB Laboratory Research, National Center for Infectious Diseases; Epidemiology and Surveillance Br, Div of STD Prevention, National Center for HIV, STD, and TB Prevention, CDC. Editorial NoteEditorial Note: The incidence of gonorrhea among MSM declined substantially in the United States during the early 1980s (2) as the HIV epidemic led to substantial reductions in sexual risk behaviors (3). However, findings in both the United States and Europe indicate a possible reversal in GC trends among MSM (4,5). Several studies have documented relapses in high-risk behavior among MSM, including unprotected anogenital intercourse (1,6). A recent multicity survey of MSM aged 15-22 years indicated that HIV was highly prevalent (5%-10%) in this population, coincident with a high rate of unprotected anal sex (7). The findings in this report indicate that, despite a continuing decline in overall rates of GC in the United States (8), the incidence of GC in MSM may be increasing in several U.S. cities. This increase cannot be explained by such factors as improved case ascertainment or increased screening in this population. This report also documented increases in rectal GC in several clinics, an indicator of unprotected anal intercourse. Although complete data were unavailable, preliminary observations from Portland, San Francisco, and Seattle linked GC cases in MSM with attendance at certain local clubs and other places frequented by MSM. Observations from Seattle further implicated sexual activities with anonymous partners and the use of illicit drugs and alcohol in the increase in GC cases among MSM. An increase in high-risk encounters among MSM could explain the increase in GC cases and could enhance HIV transmission in this population. Among clinics with information about HIV status, approximately one fourth of MSM with GC also were HIV infected. The presence of urethritis in persons with HIV increases the quantity of HIV in their semen (9) and presumably the likelihood of HIV transmission, while the presence of urethritis in persons without HIV has been associated with an increased likelihood of HIV acquisition (10). Because information about sexual orientation and behaviors is not part of routine GC case reporting, data about GC trends among MSM must be obtained through special surveillance efforts. This report demonstrates how a sentinel surveillance system designed for tracking antimicrobial resistance can be useful for following epidemiologic trends. However, proportional changes noted in GISP data may require further investigation using local data. STD clinics and other facilities that serve substantial numbers of MSM are encouraged to collect and analyze local data to follow trends in STDs and sexual behaviors that may increase the risk for acquisition or transmission of HIV infection among MSM. The increase in GC among MSM underscores the need for innovative approaches to achieving and maintaining safer-sex practices among MSM. In addition, the relation between GC and the spread of HIV (9,10) necessitates specific attention to GC control, including routine screening for GC among MSM served by both public and private providers, prompt treatment of infection, and appropriate partner management. Public health agencies and other organizations serving MSM must recognize the importance of GC as a public health problem that is linked, through behavior and biology, to spread of HIV. References
* Clinics were located in Denver; Honolulu; Long Beach, Orange County, San Diego, and San Francisco, California; Portland, Oregon; and Seattle, Washington. Figure_1 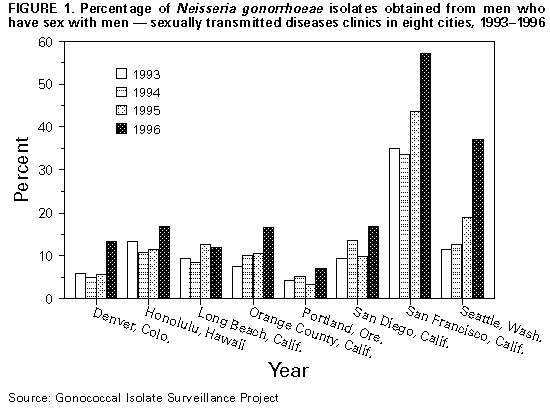 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 09/19/98 |
|||||||||
This page last reviewed 5/2/01
|