 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Influenza Vaccination of Health-Care PersonnelRecommendations of the Healthcare Infection Control Practices Advisory Committee (HICPAC) and the Advisory Committee on Immunization Practices (ACIP)Please note: An erratum has been published for this article. To view the erratum, please click here. Prepared by
The material in this report originated in the National Center for Infectious Diseases, Rima F. Khabbaz, MD, Director; Division of Healthcare Quality Promotion, Denise M. Cardo, MD, Director; Division of Viral and Rickettsial Diseases, Steve Monroe, PhD, Acting Director; and National Immunization Program, Anne Schuchat, MD, Director; Epidemiology and Surveillance Division, Alison Mawle, PhD, Acting Director. Corresponding preparer: Michele L. Pearson, MD, Division of Healthcare Quality Promotion, National Center for Infectious Diseases, 1600 Clifton Road, NE, MS A-31, Atlanta, GA 30333. Telephone: 404-639-4251; Fax: 404-639-4046; E-mail: mpearson@cdc.gov. SummaryThis report summarizes recommendations of the Healthcare Infection Control Practices Advisory Committee (HICPAC) and the Advisory Committee on Immunization Practices (ACIP) concerning influenza vaccination of health-care personnel (HCP) in the United States. These recommendations apply to HCP in acute care hospitals, nursing homes, skilled nursing facilities, physician's offices, urgent care centers, and outpatient clinics, and to persons who provide home health care and emergency medical services. The recommendations are targeted at health-care facility administrators, infection-control professionals, and occupational health professionals responsible for influenza vaccination programs and influenza infection-control programs in their institutions. HICPAC and ACIP recommend that all HCP be vaccinated annually against influenza. Facilities that employ HCP are strongly encouraged to provide vaccine to their staff by using evidence-based approaches that maximize vaccination rates. IntroductionInfluenza transmission and outbreaks in hospitals (1--8) and nursing homes (9--13) are well documented. HCP can acquire influenza from patients or transmit influenza to patients and other staff. Despite the documented benefits of HCP influenza vaccination on patient outcomes (14,15) and HCP absenteeism (16) and on reducing influenza infection among staff (16,17), vaccination coverage among HCP remain low (i.e., <50%) (18). Because HCP provide care to patients at high risk for complications of influenza, HCP should be considered a high priority for expanding influenza vaccine use. In addition, older HCP (i.e., aged >65 years) and those who have underlying chronic medical conditions or who might be pregnant are at increased risk for influenza-related complications. Achieving and sustaining high vaccination coverage among HCP will protect staff and their patients, and reduce disease burden and health-care costs. This report summarizes recommendations of the Healthcare Infection Control Practices Advisory Committee (HICPAC) and the Advisory Committee on Immunization Practices (ACIP) concerning influenza vaccination of health-care personnel (HCP)* in the United States. These recommendations are targeted at health-care facility administrators, infection control professionals, and occupational health professionals responsible for influenza vaccination programs and influenza infection control programs in their institutions. HICPAC and ACIP recommend that all HCP be vaccinated annually against influenza. Facilities that employ HCP are strongly encouraged to provide vaccine to their staff by using evidence-based approaches that maximize vaccination rates. This report supplements ACIP's previous statement regarding use of influenza vaccine and antiviral agents (1), which provides details regarding the epidemiology of influenza transmission in nonhealth-care settings, influenza vaccination of nonhealth-care personnel, composition of influenza vaccines, and use of antiviral medications. Summary RecommendationsThe summary recommendations contained in this report are categorized by using the HICPAC evidence ranking system (Table 1). The recommendations were drafted after review of peer-reviewed scientific articles, and whenever possible are based on well-designed studies; certain recommendations are based on strong theoretic rationale and expert opinion. All recommendations have been approved by HICPAC and ACIP. The committees involved in drafting and reviewing these recommendations included persons with expertise in infectious diseases, infection control, pediatrics, vaccinology, internal medicine, and public health. The recommendations are as follows:
BackgroundInfluenza Among HCPA limited number of prospective and cross-sectional studies provide estimates of incidence of influenza and influenza-like illness (ILI) among HCP (17,19,20). In one serosurvey of HCP, 23% of HCP had documented serologic evidence of influenza infection after a mild influenza season; however, of these, 59% could not recall having influenza, and 28% could not recall any respiratory infection, suggesting a high proportion of asymptomatic illness (17). In a randomized trial of influenza vaccine among HCP, 13% of placebo recipients subsequently had influenza infection (18). In a cross-sectional survey of house staff, 37% reported ILI during an 8-month period (September--April); 9% reported more than one illness. Length of illness varied (range: 1--10 days; mean: 7 days), as did the number of days of work missed (range: 0--10 days; mean: 0.7 days) (20). Efficacy and Effectiveness of Influenza Vaccines Among AdultsTrivalent inactivated influenza vaccine prevents influenza illness among approximately 70%--90% of healthy adults aged <65 years when the vaccine and circulating viruses are antigenically similar (17,21--23). The effectiveness of inactivated influenza vaccine in preventing influenza illness might be lower when vaccine and circulating viruses are not well matched or among adults aged >65 years and persons with certain chronic conditions (e.g., diabetes, human immunodeficiency virus (HIV), or chronic obstructive pulmonary disease) (24--28). Vaccination of healthy adults also decreases work absenteeism and use of health-care resources, including antibiotics, when the vaccine and circulating viruses are well matched (17,21, 23,29--31). In addition, influenza vaccine prevents secondary complications and reduces the risk for influenza-related hospitalization and death among adults aged >65 years with and without high-risk medical conditions (e.g., heart disease and diabetes) (32--36). LAIV has demonstrated similar benefits in randomized controlled trials among healthy working adults aged 18--64 years. In one study, vaccination with LAIV reduced severe febrile illnesses 19% and upper respiratory tract illnesses 24%; LAIV use also was associated with fewer days of illness and of work lost, fewer health-care provider visits, and reduced use of prescription antibiotics and over-the-counter medications (37). These results were recorded during a season in which the vaccine and circulating influenza A (H3N2) strains were not well matched. In the same study, LAIV vaccination yielded similar benefits among a subset of healthy adults aged 18--49 years, and antibiotic use in this age group decreased 41%--51% (37). In one study, overall efficacy of LAIV and inactivated influenza vaccine in preventing laboratory-documented influenza was 85% and 71%, respectively (38). Impact of HCP Vaccination on Influenza in Health-Care SettingsVaccination of HCP is an important component of influenza prevention programs in the United States (18). Vaccination of HCP reduces transmission of influenza in health-care settings, staff illness and absenteeism, and influenza-related morbidity and mortality among persons at increased risk for severe influenza illness (14--17). Use of antiviral drugs used for chemoprophylaxis or treatment of influenza is an adjunct to (but not a substitute for) vaccination (18). Transmission of Influenza in Health-Care SettingsInfluenza outbreaks in hospitals (4,39) and long-term--care facilities (40) have been associated with low vaccination rates among HCP. In addition, higher vaccination levels among staff have been associated with a lower incidence of nosocomial influenza cases (14,15,39). In one tertiary care facility in which routine surveillance for influenza was conducted, the relation between staff vaccination coverage and annual incidence of nosocomial influenza was assessed for 12 influenza seasons during 1987--2000. During this period, staff vaccination coverage increased from 4% during 1987--1988 to 67% during 1999--2000 (p<0.0001), and the proportion of laboratory-confirmed cases of influenza that occurred among HCP decreased from 42% during 1990--1993 to 9% during 1997--2000 (p<0.0001). The proportion of nosocomial cases among hospitalized patients decreased 32% to 0 (p<0.0001). After controlling for potential confounders by using logistic regression, a significant and inverse relationship was demonstrated between vaccination rates among HCP and the rate of nosocomial influenza among patients, suggesting that staff vaccination contributed to the observed decline in the number of nosocomial influenza cases (39). Staff Illness and AbsenteeismDuring an influenza season, HCP might acquire influenza from infected patients with resulting morbidity and absenteeism. The impact of influenza vaccination on staff illness and absenteeism has been evaluated in two randomized, placebo-controlled, double-blind trials. In one trial, HCP who received vaccine had 28% fewer documented lost work days attributable to respiratory infections (1.0 and 1.4, respectively; p = 0.02) and 28% fewer days on which they felt unable to work, whether they were on or off duty (2.5 and 3.5, respectively; p = 0.02). Vaccination did not reduce either the number of episodes (1.8 and 2.0, respectively) or the total number of days (13.5 and 14.6, respectively) of respiratory infection (16). In a second trial conducted in two large teaching hospitals for 3 consecutive years that measured serologically confirmed influenza, days of febrile respiratory illness, and days absent from work, HCP who received influenza vaccine had a substantially lower incidence of influenza than controls (1.7% and 13.4%, respectively) with an estimated vaccine efficacy against serologically defined influenza A and influenza B infection of 88% and 89%, respectively. HCP who received influenza vaccine also tended to have fewer total respiratory illnesses (28.7 and 40.6 per 100 persons, respectively; p = 0.57) and days of lost work (9.9 and 21.1 per 100 persons, respectively; p = 0.41) than did controls (17). In a cross-sectional survey, similar reductions in staff illness episodes and days of illness were reported (20). Overall, compared with unvaccinated coworkers, vaccinated house staff reported 23% fewer ILIs (42 and 54 per 100 persons, respectively; p = 0.03), 27% fewer days of illness (80 and 115 per 100 persons, respectively; p = 0.02), and a 59% reduction in illness during vacation time (1.7% and 4.0% of persons, respectively; p = 0.08). The two groups had a similar number of lost work days attributable to ILI (18 and 21 per 100 subjects, respectively; p = 0.69). During influenza season, vaccination was associated with reductions of 30% in ILI (p = 0.05), 43% in the proportion of house staff reporting illnesses associated with fever and cough (p = 0.05), and 63% in illnesses associated with fever and cough (p = 0.03). The inability to consistently demonstrate statistically significant decreases in absenteeism among staff who received vaccination is likely attributable to the finding that HCP tend to work despite illness (17,41). Patient OutcomesHCP who are clinically or subclinically infected can transmit influenza virus to other persons. Decreasing transmission of influenza from caregivers to persons at high risk might reduce influenza-related deaths among persons at high risk for complications from influenza. Residents of long-term--care facilities are particularly vulnerable to influenza and influenza-related complications. In1999, an estimated 1.6 million persons resided in nursing homes in the United States (42). During influenza outbreaks in long-term--care facilities, attack rates among residents have ranged as high as 25%--60%, with case-fatality rates of 10%--20% (13,43--45). When vaccine and epidemic strains are well matched, achieving increased vaccination rates among persons living in closed settings (e.g., nursing homes and other chronic-care facilities) and among staff can reduce the risk for outbreaks by inducing herd immunity (32). Two randomized controlled trials have evaluated the impact of influenza vaccination of HCP on the outcomes of residents in nursing homes. In one study, staff vaccination was associated with a 43% decrease in incidence of ILI (odds ratio [OR] = 0.6; 95% confidence interval [CI] = 0.3--0.9) and a 44% decrease in overall mortality among facility residents, from 17% to 10% (OR = 0.6; 95% CI = 0.4--0.8) (15). No virologic data were provided in this study. In a second study, 20 long-term--care facilities were randomized to have vaccine routinely offered (intervention facilities) or not offered (control facilities) to their staff (14). Facilities were paired by number of beds and patient vaccination policies. Staff vaccination coverage was higher in intervention facilities than in control facilities (50.9% and 4.9%, respectively). Crude mortality rates were 42% lower among residents in facilities with higher staff vaccination coverage than those in control facilities (13.6% and 22.4%, respectively; OR = 0.6; 95% CI = 0.4--0.8; p = 0.014). Incidence of laboratory-confirmed influenza did not differ between the two groups (5.4% and 6.7%, respectively), but postmortem samples from patients in control facilities were more likely to be positive for influenza by a polymerase chain reaction test than samples from patients in intervention facilities (six [20%] of 30 and none of 17, respectively; p = 0.055), suggesting that in this study population, HCP vaccination reduced influenza-related mortality in patients despite not reducing the incidence of non-fatal influenza infection. In neither study was a significant association demonstrated between patient vaccination and mortality. Randomized trials assessing the impact of staff vaccination on patient outcomes in acute care facilities have not been conducted, but low staff vaccination coverage has been correlated with influenza outbreaks in hospitals (4,39). Cost-Effectiveness of Influenza VaccineCost-effectiveness studies of adults aged <65 years indicate that vaccination can reduce both direct medical costs and indirect costs from work absenteeism (21,23,29,30,46,47), resulting in 13%--44% fewer health-care provider visits, 18%--45% fewer lost workdays, 18%--28% fewer days working with reduced effectiveness, and a 25% decrease in antibiotic use for ILI (21,29,48,49). Among healthy persons aged 18--64 years, vaccination can save an estimated $60--$4,000 per illness, depending on the cost of vaccination, the influenza attack rate, and vaccine effectiveness against ILI (23). In another economic analysis, vaccination resulted in an average annual cost savings of $13.66 per person vaccinated (50); however, other analyses have not demonstrated cost savings (21). Among studies of healthy young adults, >70% of the costs prevented were associated with reductions in lost work productivity. Vaccination Coverage Levels Among HCPDuring 1989--2003, HCP vaccination coverage levels in the United States increased substantially, from 10% to 40%; however, coverage levels have remained relatively constant since 1997 (18). One of the national health objectives for 2010 is to achieve HCP vaccination coverage levels of 60% (objective no. 14-29g) (51). Substantially lower vaccination rates have been reported among HCP who have contact with certain populations at high risk (12,52--54). In addition, HCP vaccination coverage varies by level and years of training, age, occupational group, and facility type (20,55,56). Barriers to HCP VaccinationReported barriers to HCP receipt or acceptance of influenza vaccination include fear of vaccine side effects (particularly ILI symptoms) (20,55,57--61), insufficient time or inconvenience (20), perceived ineffectiveness of the vaccine (20,55,58,59), medical contraindication (55), perceived low likelihood of contracting influenza (55,60,62), reliance on treatment with homeopathic medications (55,62), avoidance of medications (57), and fear of needles (57,59). Factors facilitating vaccine acceptance include a desire for self-protection (20,58,61), previous receipt of influenza vaccine (57,58,63--65), a desire to protect patients (61), and perceived effectiveness of vaccine (20). Strategies for Improving HCP Vaccination RatesFacilities that employ HCP are strongly encouraged to provide vaccine to staff by using evidence-based approaches that maximize vaccination rates. Successful HCP vaccination programs are multifaceted and combine publicity and education to combat fears and misconceptions about influenza and influenza vaccines, use of reminder recall systems, efforts to remove administrative and financial barriers, role modeling, and monitoring and feedback on vaccination coverage (66). In contrast, single-component interventions will likely have minimal effectiveness in achieving desired vaccination coverage levels (66,67). Education and CampaignsHCP knowledge, perceptions, and attitudes regarding influenza and influenza vaccination vary (20). Basic knowledge about influenza and influenza vaccination has been associated with vaccine receipt (57,68,69), and participation in structured in-service education or conferences has been associated with improved vaccination rates (62,65). Educational programs should emphasize the benefits of HCP vaccination for staff and patients (70). Organized campaigns that promote and make vaccine accessible can improve vaccination rates among HCP (52,71). Role ModelsVaccination of senior medical staff or opinion leaders has been associated with higher vaccination acceptance among staff members under their leadership (55,69,72,73). For example, medical students who have contact with infectious disease specialists are more likely to be vaccinated (69). Improved AccessRemoving administrative barriers (e.g., costs) (71) and providing vaccine in locations and at times easily accessible by HCP can substantially improve vaccine acceptance (40,52,55,72,74,75). In one survey, 33% of HCP reported that they would reject vaccination if they were required to pay for the vaccine (76). Making vaccine readily accessible at congregate areas (e.g., clinics), during conferences, or by use of mobile carts (40,52,55,72) has been demonstrated to improve vaccination coverage rates. Use of mobile carts has been associated with increased vaccine acceptance during outbreaks and nonoutbreak situations (75,76) . In a 3-year prospective study in a 630-bed acute care hospital, a sustained four- to fivefold increase in vaccination rates was associated with using mobile carts to deliver vaccine to staff rather than requiring HCP to visit an employee health center to receive vaccine. Provision of modest incentives also has been associated with improved vaccine acceptance among HCP (77). However, the benefits of vaccine deputies or peer-vaccinators have not been consistently associated with improved HCP vaccination (52). Measurement and FeedbackHCP influenza vaccination coverage should be regularly measured and reported. Posting of vaccination coverage levels in different areas of the hospital is a component of successful vaccination programs (6). Monitoring vaccination coverage by facility area (e.g., ward or unit) or occupational group allows facilities to identify where vaccination levels are low and interventions should be targeted. In addition, HICPAC has recommended that HCP influenza vaccination coverage be used as a health-care quality measure in those states that mandate public reporting of health-care--associated infections (78). The independent contribution of signed declination statements to improving HCP vaccination has not been studied. However, obtaining declination statements from HCP who refuse vaccination for reasons other than medical contraindications can assist facilities in identifying personnel who might require targeted education or other interventions to overcome barriers to vaccine acceptance. In addition, collection of such information will allow health-care facilities to determine what proportion of their staff are reached and offered vaccine. Legislation and RegulationLegislative and regulatory efforts have favorably affected hepatitis B vaccination rates among HCP (79,80). As of January 2005, a total of 13 states (Alabama, Arkansas, Kentucky, Maine, Maryland, New Hampshire, New York, Oklahoma, Oregon, Pennsylvania, Rhode Island, Texas, and Utah) and the District of Columbia were reported to have enacted regulations regarding influenza vaccination of staff in long-term--care facilities (67,81). However, because only one state (Pennsylvania) has monitored the impact of its laws on nursing home staff vaccination rates, data are insufficient to assess the overall impact of these legislative efforts on HCP influenza vaccination coverage (CDC, unpublished data, 2005). Recommendations for Using Inactivated Influenza Vaccine and LAIV Among HCPAll HCP should be vaccinated annually against influenza. Either inactivated influenza vaccine or LAIV can be used to reduce the risk for influenza among HCP (Table 2). LAIV is approved for use only among nonpregnant healthy persons aged 5--49 years. HCP who work with severely immunocompromised patients who require a protected environment should not receive LAIV. Inactivated influenza vaccine is approved for all persons aged >6 months who lack vaccine contraindications, including those with high-risk conditions (see Recommendations for Prioritization of Influenza Vaccine During the 2005--06 Influenza Season). Four influenza vaccines have been approved for use in the United States during the 2005--06 season (Table 3). Inactivated Influenza Vaccine RecommendationsDosage and Route Because immunity declines during the year after vaccination, HCP eligible to receive inactivated influenza vaccine should be administered 1 dose of the current year's vaccine each year (82,83). The intramuscular route is recommended for inactivated influenza vaccine. Adults should be vaccinated in the deltoid muscle, ideally by using a needle of length >1 inch because needles of length <1 inch might not penetrate muscle tissue in certain adults (84). Persons Who Should Not Be Vaccinated with Inactivated Influenza Vaccine Inactivated influenza vaccine should not be administered to persons known to have anaphylactic hypersensitivity to eggs or to other components of the influenza vaccine without first consulting a physician (see Side Effects and Adverse Reactions Associated with Vaccination). Prophylactic use of antiviral agents is an option for preventing influenza among such persons. However, persons who have a history of anaphylactic hypersensitivity to vaccine components but who are also at high risk for complications from influenza can benefit from vaccine after appropriate allergy evaluation and desensitization (18). Information regarding vaccine components is located in package inserts from each manufacturer. Persons with acute febrile illness typically should not be vaccinated until their symptoms have abated. However, minor illnesses with or without fever do not contraindicate use of influenza vaccine. LAIV RecommendationsLAIV licensed for use in the United States (FluMist™ manufactured by MedImmune, Inc., Gaithersburg, Maryland [http//www.medimmune.com]) is a live, trivalent, intranasally administered vaccine that is
The immunogenicity of the approved LAIV has been assessed in multiple studies (85--91). LAIV virus strains replicate primarily in nasopharyngeal epithelial cells. The protective mechanisms induced by vaccination with LAIV are not completely understood but appear to involve both serum and nasal secretory antibodies. No single laboratory measurement closely correlates with protective immunity induced by LAIV. Shedding and Transmission of Vaccine Viruses One concern regarding use of LAIV among HCP has been the potential for transmitting vaccine virus from persons receiving vaccine to nonimmune patients at high risk. Available data indicate that children and adults vaccinated with LAIV can shed vaccine viruses for >2 days after vaccination, although in lower titers than typically occur with shedding of wild-type influenza viruses. Shedding should not be equated with person-to-person transmission of vaccine viruses, although transmission of shed vaccine viruses from vaccinated persons to nonvaccinated persons has been documented in rare instances among children in a day care center (92). In one study of 20 healthy vaccinated adults aged 18--49 years, the majority of vaccine virus shedding occurred within the first 3 days after vaccination, although in one vaccinated person, viral shedding was detected on day 7 after vaccination (93). No vaccine viruses were shed >10 days after vaccination, and duration or type of symptoms associated with receipt of LAIV did not correlate with duration of shedding of vaccine viruses (93). In another study of 14 healthy adults aged 18--49 years, 50% of vaccinated persons had viral antigen detected by direct immunofluorescence or rapid antigen tests within 7 days of vaccination; the majority of viral shedding was detected on day 2 or 3 (94). Person-to-person transmission of vaccine viruses was not assessed in either of these studies. One study conducted in a child care center assessed transmissibility of vaccine viruses from 98 vaccinated persons to 99 unvaccinated controls aged 8--36 months; 80% of vaccine recipients shed one or more virus strains (mean duration: 7.6 days). One influenza type B isolate was recovered from a placebo recipient and confirmed to be vaccine-type virus; the isolate retained the cold-adapted, temperature-sensitive, attenuated phenotype and possessed the same genetic sequence as a virus shed from a vaccine recipient in the same children's play group. The placebo recipient from whom the influenza type B vaccine virus was isolated exhibited symptoms that were similar to those experienced by vaccine recipients. The estimated probability of acquiring vaccine virus after close contact with a single LAIV recipient in this child care population was 0.6%--2.4% (92). Using LAIV for HCP LAIV may be used for vaccination of healthy, nonpregnant persons aged 5--49 years, including HCP. When feasible, use of LAIV for vaccination of eligible HCP is especially encouraged during periods of limited supply of inactivated influenza vaccine because use of LAIV for HCP might increase availability of inactivated influenza vaccine for persons at high risk. Use of LAIV also provides an alternative vaccine strategy for HCP who avoid influenza vaccination because of an aversion to intramuscular injections. Persons Who Should Not Receive LAIV The following populations should not receive LAIV:
LAIV Dosage and Administration Eligible HCP should receive 1 dose of LAIV. LAIV is intended only for intranasal administration and should not be administered by the intramuscular, intradermal, or intravenous route. Administration can be accomplished by holding an individual sprayer in the palm of the hand until thawed, with subsequent immediate administration. Alternatively, the vaccine can be thawed in a refrigerator and stored at 35.6° F--46.4° F (2° C--8° C) for <60 hours before use. Vaccine should not be refrozen after thawing. LAIV is supplied in a prefilled single-use sprayer containing 0.5 mL of vaccine. Approximately 0.25 mL is sprayed into the first nostril while the recipient is in the upright position. An attached dose-divider clip is removed from the sprayer to administer the second half of the dose into the other nostril. If the vaccine recipient sneezes after administration, the dose should not be repeated. LAIV may be administered to persons with minor acute illnesses (e.g., diarrhea or mild upper respiratory tract infection, with or without fever). However, if clinical judgment indicates the presence of nasal congestion that might impede delivery of vaccine to the nasopharyngeal mucosa, deferral of administration should be considered until resolution of the illness. Whether concurrent administration of LAIV with other vaccines affects the safety or efficacy of either LAIV or the simultaneously administered vaccine is unknown. In the absence of specific data indicating interference, adherence to ACIP's general recommendations for vaccination is prudent (95). Inactivated vaccines do not interfere with the immune response to other inactivated vaccines or to live vaccines. An inactivated vaccine can be administered either simultaneously or at any time before or after LAIV. Whenever possible, two live vaccines not administered on the same day should be administered >4 weeks apart. Recommended Vaccines for HCP Who Have Close Contact with Severely Immunosuppressed PersonsInactivated influenza vaccine is the preferred vaccine for use among HCP who have close contact with severely immunosuppressed persons (e.g., patients with hematopoietic stem cell transplants) during those periods in which the immunosuppressed person requires care in a protective environment. The rationale for not using LAIV among HCP caring for such patients is the theoretic risk that a live, attenuated vaccine virus could be transmitted to the severely immunosuppressed person. HCP who receive LAIV should refrain from contact with severely immunosuppressed patients for 7 days after vaccine receipt. In addition, visitors who have received LAIV should refrain from contact with severely immunosuppressed persons for 7 days after vaccination; however, such persons need not be excluded from visitation of patients who are not severely immunosuppressed. Either inactivated influenza vaccine or LAIV can be used to vaccinate HCP who have close contact with persons with lesser degrees of immunosuppression (e.g., persons with diabetes, persons with asthma taking corticosteroids, or persons infected with human immunodeficiency virus) or who are in close contact with all other persons at high risk. Personnel Who May Administer LAIV The risk of acquiring vaccine viruses from the environment is unknown but likely small. Nevertheless, severely immunosuppressed persons should not administer LAIV because introduction of low levels of vaccine virus into the environment probably cannot be avoided when administering LAIV. However, other persons with conditions placing them at high risk for influenza complications (e.g., pregnant women, persons with asthma, and persons aged >50 years) may administer LAIV. LAIV and Use of Influenza Antiviral Medications How LAIV coadministration with influenza antiviral medications affects safety and efficacy has not been studied. However, because influenza antivirals reduce replication of influenza viruses, LAIV should not be administered until 48 hours after cessation of influenza antiviral therapy, and influenza antiviral medications should not be administered for 2 weeks after receipt of LAIV. LAIV Storage LAIV must be stored at -59° F (-15° C) or colder. LAIV may be stored in frost-free freezers without using a freezer-box. LAIV can be thawed in a refrigerator and stored at 35.6° F--46.4° F (2º C--8º C) for <60 hours before use. It should not be refrozen after thawing. Additional information regarding LAIV storage is available at http://www.FluMist.com. Vaccination of Specific HCP PopulationsPregnant Women Pregnant women are at increased risk for influenza-related complications (96--103) and hospitalizations (104). Therefore, all HCP who are pregnant during the influenza season should be vaccinated against influenza. However, pregnant women should receive only inactivated influenza vaccine; LAIV is not recommended for use during pregnancy. Inactivated influenza vaccine may be administered in any trimester. One study of influenza vaccination of approximately 2,000 pregnant women demonstrated no adverse fetal effects associated with receipt of inactivated influenza vaccine (105). Breastfeeding Mothers Influenza vaccine does not affect the safety of mothers who are breastfeeding or their infants. Breastfeeding does not adversely affect the immune response and is not a contraindication for vaccination. Persons Infected with HIV Detailed information on the use of influenza vaccine among persons infected with HIV has been published previously (18). Because influenza can result in serious illness and influenza vaccination can result in the production of protective antibody titers, vaccination with inactivated vaccine will benefit HIV-infected persons, including those that are pregnant. Timing of Annual Influenza Vaccination of HCPTiming of Organized Vaccination Campaigns Planning for influenza campaigns should begin as early as February or March (106). The optimal time to vaccinate HCP is during October--November. Beginning in October each year, health-care facilities should offer influenza vaccinations to all full- and part-time staff. Particular emphasis should be placed on vaccinating HCP who care for persons at high risk. Vaccination programs should educate HCP regarding the benefits of vaccination and the potential health consequences of influenza illness for themselves and their patients. As part of employee health programs, all HCP should be provided convenient access to free influenza vaccine at the work site (107). Vaccination in December and Later To improve vaccine coverage among HCP, influenza vaccine should continue to be offered in December and throughout the influenza season as long as vaccine supplies are available, even after influenza activity has been documented in the community. In the United States, seasonal influenza activity can increase as early as October or November, but influenza activity has not reached peak levels in the majority of recent seasons until late December--early March. Therefore, although the timing of influenza activity can vary by region, vaccine administered after November is likely to be beneficial in the majority of influenza seasons. Adults achieve peak antibody protection against influenza infection 2 weeks after vaccination (108,109). Recommendations for Prioritization of Influenza Vaccination During the 2005--06 Influenza SeasonAs a result of influenza vaccine distribution delays or supply shortages in the United States during recent influenza seasons (110,111), in September 2005, CDC issued recommendations for prioritizing the use of inactivated vaccine during the 2005--06 influenza season to ensure that early vaccine is available for those at the highest risk for complications from influenza (112). On the basis of uncertainties in doses and distribution, CDC recommended that the following groups receive priority for inactivated influenza vaccine until October 24, 2005:
These groups correspond to tiers 1A--1C in the table of inactivated influenza vaccine priority groups in the event of vaccination supply disruption that was published previously (113). After October 24, 2005, all persons were eligible for vaccination. Tiered use of prioritization was not recommended for LAIV administration. LAIV may be administered at any time for vaccination of nonpregnant healthy persons aged 5--49 years, including the majority of HCP, other persons in close contact with persons at high risk for influenza-related complications, and others desiring protection against influenza (18). Side Effects and Adverse Reactions Associated with VaccinationInactivated Influenza VaccineWhen educating HCP regarding potential side effects, providers should emphasize that 1) inactivated influenza vaccine contains noninfectious killed viruses and cannot cause influenza; and 2) coincidental respiratory disease unrelated to influenza vaccination can occur after vaccination. The occurrence of vaccine-related side effects has had limited to no impact on rates of absenteeism among HCP (16,17). Local ReactionsThe most frequent side effect of vaccination (affecting 10%--64% of patients) is soreness at the vaccination site, typically lasting <2 days (21,114--116). Local reactions typically are mild and rarely interfere with a person's ability to conduct everyday activities. In a controlled trial, only body aches (25.1%) were reported more frequently after inactivated influenza vaccine than placebo-injection (20.8%) (117). Systemic ReactionsFever, malaise, myalgia, and other systemic symptoms can occur after vaccination with inactivated vaccine and most often affect persons (e.g., infants) with no previous exposure to the influenza virus antigens in the vaccine (118,119). Such reactions typically begin 6--12 hours after vaccination and can persist for 1--2 days. Recent placebo-controlled trials demonstrate that among older persons and healthy young adults, administration of split-virus (i.e., detergent-disrupted virion) influenza vaccine is not associated with higher rates of systemic symptoms (e.g., fever, malaise, myalgia, and headache) compared with placebo injections (21,114--116). No increase in asthma exacerbations has been documented in association with receipt of influenza vaccine (117). Severe Adverse EventsImmediate and presumably allergic reactions (e.g., hives, angioedema, allergic asthma, and systemic anaphylaxis) rarely occur after influenza vaccination (120). These reactions probably result from hypersensitivity to certain vaccine components; the majority of reactions probably are caused by residual egg protein. Although current influenza vaccines contain only a limited quantity of egg protein, this protein can induce immediate hypersensitivity reactions among persons who have severe egg allergy. Persons who have had hives or swelling of the lips or tongue, or who have experienced acute respiratory distress or collapse after eating eggs should consult a physician for appropriate evaluation to help determine if vaccine should be administered. Persons who have documented immunoglobulin E (IgE)-mediated hypersensitivity to eggs, including those who have had occupational asthma or other allergic responses to egg protein, might also be at increased risk for allergic reactions to influenza vaccine, and consultation with a physician should be considered. Protocols have been published for administering influenza vaccine safely to persons with egg allergies (121--123). Hypersensitivity reactions to any vaccine component can occur. Although exposure to vaccines containing thimerosal can lead to induction of hypersensitivity, the majority of patients do not have reactions to thimerosal when it is administered as a component of vaccines, even when patch or intradermal tests for thimerosal allergy indicate hypersensitivity (124,125). When reported, hypersensitivity to thimerosal typically has consisted of local, delayed hypersensitivity reactions (124). GBSInvestigations to date indicate no substantial increase in GBS associated with influenza vaccines (other than the 1976 swine influenza vaccine) (126--130). If current influenza vaccines pose a risk for GBS, the estimated risk is approximately one additional case per million persons vaccinated, with the total combined number of GBS cases peaking 2 weeks after vaccination (131). This estimated risk for GBS is substantially less than the risk for severe influenza, which can be prevented by vaccination among all age groups, especially persons aged >65 years and those who have medical indications for influenza vaccination. The potential benefits of influenza vaccination in preventing serious illness, hospitalization, and death substantially outweigh the possible risks for experiencing vaccine-associated GBS. The average case-fatality ratio for GBS is 6% and increases with age (132,133). No evidence indicates that the case-fatality ratio for GBS differs among vaccinated persons and those not vaccinated. Incidence of GBS among the general population is low, but persons with a history of GBS have a substantially greater likelihood of subsequently experiencing GBS than persons without such a history (128,134). Whether influenza vaccination might increase the risk for recurrence of GBS is unknown; for this reason, persons who are not at high risk for severe influenza complications and who are known to have experienced GBS within 6 weeks after a previous influenza vaccination should not receive vaccine. Chemoprophylaxis using influenza antivirals might be an alternative for such persons. Although data are limited, for the majority of persons who have a history of GBS and who are at high risk for severe complications from influenza, the established benefits of influenza vaccination justify yearly vaccination. Health-care professionals should promptly report all clinically significant adverse events after influenza vaccination to the Vaccine Adverse Event Reporting System (VAERS), even if evidence is lacking that the vaccine caused the event. LAIVUntil additional data are available, persons at high risk for experiencing complications from influenza infection (e.g., immunocompromised patients; patients with asthma, cystic fibrosis, or chronic obstructive pulmonary disease; or persons aged >65 years) should not be vaccinated with LAIV. Protection from influenza among these groups should be accomplished by using inactivated influenza vaccine. Among adults, runny nose or nasal congestion (28%--78%), headache (16%--44%), and sore throat (15%--27%) have been reported more often among vaccine recipients than placebo recipients (37,135,136). In one clinical trial among a subset of healthy adults aged 18--49 years, signs and symptoms reported more frequently among LAIV recipients (n = 2,548) than placebo recipients (n = 1,290) within 7 days after each dose included cough (13.9% and 10.8%, respectively); runny nose (44.5% and 27.1%, respectively); sore throat (27.8% and 17.1%, respectively); chills (8.6% and 6.0%, respectively); and tiredness or weakness (25.7% and 21.6%, respectively) (37). Pneumonia, bronchitis, bronchiolitis, or central nervous system events have not been observed more frequently among LAIV than among placebo recipients. Severe Adverse Events Serious adverse events associated with receipt of LAIV among healthy adults aged 18--49 years occur at a rate of <1% (137). However, surveillance should continue for adverse events that might not have been detected in previous studies. Health-care professionals should promptly report to VAERS all clinically significant adverse events after LAIV administration, even if evidence is lacking that the vaccine caused the event. Additional Information Regarding Influenza Infection Control in Health-Care SettingsAdditional information on controlling and preventing influenza in health-care settings is available in the following publications:
References
* In this report, the term HCP refers to all paid and unpaid persons working in health-care settings who have the potential for exposure to infectious materials, including body substances, contaminated medical supplies and equipment, contaminated environmental surfaces, or contaminated air. HCP might include (but are not limited to) physicians, nurses, nursing assistants, therapists, technicians, emergency medical service personnel, dental personnel, pharmacists, laboratory personnel, autopsy personnel, students and trainees, contractual staff not employed by the health-care facility, and persons (e.g., clerical, dietary, housekeeping, maintenance, and volunteers) not directly involved in patient care but potentially exposed to infectious agents that can be transmitted to and from HCP. The recommendations in this report apply to HCP in acute care hospitals, nursing homes, skilled nursing facilities, physician's offices, urgent care centers, and outpatient clinics, and to persons who provide home health care and emergency medical services. † These persons should receive inactivated influenza vaccine. Healthcare Infection Control Practices Committee
Chairman: Patrick J. Brennan, MD, University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania. Executive Secretary: Michele L. Pearson, MD, CDC, Atlanta, Georgia. Members: Vicki L. Brinsko, Vanderbilt University Medical Center, Nashville, Tennessee; Raymond Y. W. Chinn, MD, Sharp Memorial Hospital, San Diego, California; E. Patchen Dellinger, MD, University of Washington School of Medicine, Seattle, Washington; Nancy E. Foster, American Hospital Association, District of Columbia; Steven M. Gordon, MD, Cleveland Clinic Foundation, Cleveland, Ohio; Lizzie J. Harrell, PhD, Duke University Medical Center, Durham, North Carolina; Carol O'Boyle, PhD, University of Minnesota, Minneapolis, Minnesota; Dennis M. Perrotta, PhD, Texas Department of Health, Austin, Texas; Harriett M. Pitt, MS, Long Beach Memorial Medical Center, Long Beach, California; Robert J. Sherertz, MD, Wake Forest University School of Medicine, Wake Forest, North Carolina; Nalini Singh, MD, Children's National Medical Center, District of Columbia; Kurt B. Stevenson, MD, Qualis Health, Boise, Idaho; Philip W. Smith, MD, University of Nebraska Medical Center, Omaha, Nebraska. Liaison Representatives: William Baine, MD, Agency for Healthcare Research and Quality, District of Columbia; Joan Blanchard, MSS, Association of periOperative Registered Nurses, Denver, Colorado; Georgia Dash, MS, Association for Professionals of Infection Control and Epidemiology, Inc., Philadelphia, Pennsylvania; Sandra L. Fitzler, American Healthcare Association, District of Columbia; David Henderson, MD, National Institutes of Health, Bethesda, Maryland; Lorine Jay, Health Services Resources Administration, Atlanta, Georgia; Stephen F. Jencks, MD, Center for Medicare and Medicaid Services, Baltimore, Maryland; Chiu S. Lin, PhD, Food and Drug Administration, Rockville, Maryland; Mark Russi, MD, American College of Occupational and Environmental Medicine, New Haven, Connecticut; Rachel Stricoff, MPH, Advisory Committee for the Elimination of Tuberculosis, New York, New York; Michael Tapper, MD, Society for Healthcare Epidemiology of America, Inc., New York, New York; and Robert Wise, MD, Joint Commisssion on the Accreditation of Healthcare Organizations, Oakbrooke, Illinois.
Advisory Committee on Immunization Practices
Chairman: Myron J. Levin, MD, Professor of Pediatrics and Medicine, University of Colorado Health Sciences Center, Denver, Colorado. Executive Secretary: Larry Pickering, MD, National Immunization Program, CDC, Atlanta, Georgia. Members: Jon S. Abramson, MD, Wake Forest University School of Medicine, Winston-Salem, North Carolina; Ban Mishu Allos, MD, Vanderbilt University School of Medicine, Nashville, Tennessee; Guthrie S. Birkhead, MD, New York State Department of Health, Albany, New York; Judith Campbell, MD, Baylor College of Medicine, Houston, Texas; Reginald Finger, MD, Focus on the Family, Colorado Springs, Colorado; Janet Gildsdorf, MD, University of Michigan, Ann Arbor, Michigan; Tracy Lieu, MD, Harvard Pilgrim Health Care and Harvard Medical School, Boston, Massachusetts; Edgar Marcuse, MD, Children's Hospital and Regional Medical Center, Seattle, Washington; Julia Morita, MD, Chicago Department of Health, Chicago, Illinois; Gregory Poland, MD, Mayo Clinic College of Medicine, Rochester, Minnesota; John B. Salamone, National Italian American Foundation, District of Columbia; Patricia Stinchfield, Children's Hospital and Clinics, St. Paul, Minnesota; John J. Treanor, MD, University of Rochester School of Medicine and Dentistry, Rochester, New York; Robin Womeodu, MD, University of Tennessee Health Sciences Center, Memphis, Tennessee. Ex-Officio Members: James E. Cheek, MD, Indian Health Service, Albuquerque, New Mexico; Stephen Phillips, DO, Department of Defense, Falls Church, Virginia; Geoffrey S. Evans, MD, Health Resources and Services Administration, Rockville, Maryland; Bruce Gellin, MD, National Vaccine Program Office, District of Columbia; Linda Murphy, Centers for Medicare and Medicaid Services, Baltimore, Maryland; George T. Curlin, MD, National Institutes of Health, Bethesda, Maryland; Norman Baylor, MD, Food and Drug Administration, Bethesda, Maryland; Kristin Lee Nichol, MD, Department of Veterans Affairs, Minneapolis, Minnesota. Liaison Representatives: American Academy of Family Physicians, Jonathan Temte, MD, Clarence, New York, and Richard Clover, MD, Louisville, Kentucky; American Academy of Pediatrics, Margaret Rennels, MD, Baltimore, Maryland, and Carol Baker, MD, Houston, Texas; American Association of Health Plans, Robert Scalettar, MD, North Haven, Connecticut; American College Health Association, James C. Turner, MD, Charlottesville, Virginia; American College of Obstetricians and Gynecologists, Stanley Gall, MD, Louisville, Kentucky; American College of Physicians, Kathleen Neuzil, MD, Seattle, Washington; American Medical Association, Litjen Tan, PhD, Chicago, Illinois; American Pharmacists Association, Stephan L. Foster, PharmD, Memphis, Tennessee; Association of Teachers of Preventive Medicine, W. Paul McKinney, MD, Louisville, Kentucky; Biotechnology Industry Organization, Clement Lewin, PhD, Cambridge, Massachusetts; Canadian National Advisory Committee on Immunization, Monica Naus, MD, Vancouver, British Columbia; Healthcare Infection Control Practices Advisory Committee, Steve Gordon, MD, Cleveland, Ohio; Infectious Diseases Society of America, Samuel L. Katz, MD, Durham, North Carolina, and William Schaffner, MD, Nashville, Tennessee; London Department of Health, David M. Salisbury, MD, London, United Kingdom; National Association of County and City Health Officials, Nancy Bennett, MD, Rochester, New York; National Coalition for Adult Immunization, David A. Neumann, PhD, Bethesda, Maryland; National Immunization Council and Child Health Program, Mexico, Romeo Rodriguez, Mexico City, Mexico; National Medical Association, Dennis A. Brooks, MD, Baltimore, Maryland; National Vaccine Advisory Committee, Charles Helms, MD, PhD, Iowa City, Iowa; Pharmaceutical Research and Manufacturers of America, Damian A. Braga, Swiftwater, Pennsylvania, Peter Paradiso, PhD, Collegeville, Pennsylvania; and Society for Adolescent Medicine, Amy Middleman, MD, Houston, Texas. Table 1  Return to top. Table 2  Return to top. Table 3 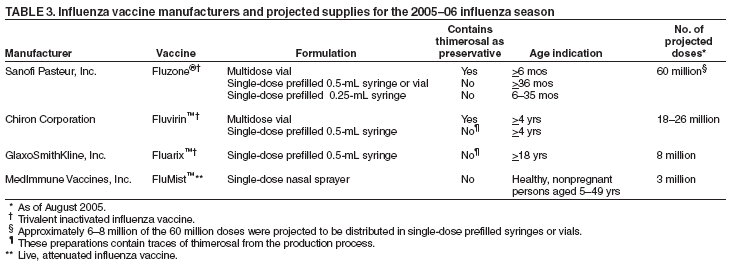 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 2/14/2006 |
|||||||||
|