 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Hantavirus Pulmonary Syndrome --- United States: Updated Recommendations for Risk ReductionPrepared by The material in this report was prepared for publication by the National Center for Infectious Diseases, James M. Hughes, M.D., Director; Office of Bioterrorism Preparedness and Response Activity, Scott Lillibridge, M.D., Director; and the National Immunization Program, Walter A. Orenstein, M.D., Director. Summary This report provides updated recommendations for prevention and control of hantavirus infections associated with rodents in the United States. It supersedes the previous report (CDC. Hantavirus infection---southwestern United States: interim recommendations for risk reduction. MMWR 1993;42[No. RR-11]:1--13). These recommendations are based on principles of rodent and infection control, and accumulating evidence that most infections result from exposure, in closed spaces, to active infestations of infected rodents. The recommendations contain updated specific measures and precautions for limiting household, recreational, and occupational exposure to rodents, eliminating rodent infestations, rodent-proofing human dwellings, cleaning up rodent-contaminated areas and dead rodents, and working in homes of persons with confirmed hantavirus infection or buildings with heavy rodent infestations. IntroductionBackgroundIn 1993, a previously unknown disease, hantavirus pulmonary syndrome (HPS), was identified among residents of the southwestern United States (1--3). HPS was subsequently recognized throughout the contiguous United States and the Americas. As of June 6, 2002, a total of 318 cases of HPS have been identified in 31 states, with a case fatality of 37%.* The association of hantaviruses with rodent reservoirs warrants recommendations to minimize exposure to wild rodents. These recommendations are based on current understanding of the epidemiologic features of hantavirus infections in the United States. Rodent Reservoirs of Viruses Causing HPSAll hantaviruses known to cause HPS are carried by the New World rats and mice, family Muridae, subfamily Sigmodontinae. The subfamily Sigmodontinae contains at least 430 species of mice and rats, which are widespread in North and South America. These wild rodents are not generally associated with urban environments as are house mice and the black and Norway rats (all of which are in the murid subfamily Murinae). However, some species (e.g., deer mouse and white-footed mouse) will enter human habitation in rural and suburban areas. A third group of rodents, the voles and lemmings (family Muridae, subfamily Arvicolinae), is associated with a group of hantaviruses distinct from those that cause HPS. None of the numerous arvicoline viruses has been associated with human disease in the United States (4). Several hantaviruses that are pathogenic for humans have been identified in the United States. In general, each virus has a single primary rodent host. Other small mammals can be infected as well, but are much less likely to transmit the virus to other animals or humans (5--7). The deer mouse (Peromyscus maniculatus) (Figure 1) is the host for Sin Nombre virus (SNV), the primary causative agent of HPS in the United States. The deer mouse is common and widespread in rural areas throughout much of the United States (Figure 2). Although prevalence varies temporally and geographically, on average approximately 10% of deer mice tested throughout the range of the species show evidence of infection with SNV (5). Other hantaviruses associated with sigmodontine rodents and known to cause HPS include New York virus (8), hosted by the white-footed mouse, Peromyscus leucopus (Figures 3,4); Black Creek Canal virus (9), hosted by the cotton rat, Sigmodon hispidus (Figures 5,6); and Bayou virus (10), hosted by the rice rat, Oryzomys palustris (Figures 7,8). Nearly all of the continental United States falls within the range of one or more of these host species. Several other sigmodontine rodent species in the United States are associated with additional hantaviruses that have yet to be implicated in human disease. These species include the brush mouse, Peromyscus boylii (11); and the Western harvest mouse, Reithrodontomys megalotis (12). Only the deer mouse and the white-footed mouse are commonly associated with peridomestic environments. Identifying characteristics and natural history of all these host species are available from other sources (13,14). Numerous species of sigmodontine rodents also are associated with HPS in South America (4). Several new sigmodontine hantavirus hosts have been discovered each year and more probably await discovery. Until the extent of hantavirus infection throughout the subfamily Sigmodontinae becomes known, as does the pathogenicity of hantaviruses hosted by sigmodontine species, treating all sigmodontines as potential hosts of HPS-causing hantaviruses, and each sigmodontine rodent as though it were infected and infectious is recommended. For the general public, this recommendation applies to all wild mice and rats encountered in rural areas throughout the United States. Other Diseases Associated with Hantavirus InfectionBecause the sigmodontine rodents are restricted to the Americas, HPS is restricted to the Americas. Another group of hantaviruses associated with murine and arvicoline rodents causes a group of diseases of varying severity referred to as hemorrhagic fever with renal syndrome (HFRS) in Europe and Asia. Hantaan and Dobrava viruses, hosted by the murine field mice (Apodemus agrarius and Apodemus flavicollis, respectively), cause thousands of cases of severe HFRS each year in Asia and Eastern Europe. Fatality associated with these infections can be as high as 10% (15). The cosmopolitan Norway rat (Rattus norvegicus) is host for Seoul virus, which causes a mild form of HFRS in Asia. Although evidence of infection with Seoul virus has been found in Norway rats throughout much of the world, including the United States, human disease caused by Seoul virus is largely restricted to Asia. Only three suspected cases have been reported in the United States (16). Overall mortality associated with Seoul virus infection is probably <1% (15). Puumala virus, carried by an arvicoline rodent, the bank vole (Clethrionomys glareolus), causes a mild form of HFRS, referred to as nephropathia epidemica (NE). NE, which is very common in northern Europe, has a case fatality of <1%. Several other species of arvicoline rodents host hantaviruses in the northern hemisphere, including the United States; none of these have been associated with any human disease. Infection in the HostHantaviruses do not cause overt illness in their reservoir hosts (17). Although infected rodents shed virus in saliva, urine, and feces for many weeks, months, or for life, the quantity of virus shed can be much greater approximately 3--8 weeks after infection (18). The demonstrated presence of infectious virus in saliva of infected rodents and the marked sensitivity of these animals to hantaviruses following intramuscular inoculation suggest that biting might be an important mode of transmission from rodent to rodent (18,19). Field data suggest that transmission in host populations occurs horizontally, more frequently among male rodents, and might be associated with fighting, particularly, but not exclusively, among males (7,20). Occasional evidence of infection (antibody) is found in numerous other species of rodents and their predators (e.g., dogs, cats, and coyotes), indicating that many (perhaps any) mammal species coming into contact with an infected host might become infected (21). No evidence supports the transmission of infection to other animals or to humans from these "dead-end" hosts. However, domestic animals (e.g., cats and dogs) might bring infected rodents into contact with humans. Arthropod vectors are not known to have a role in the transmission of hantaviruses (17,22). The reservoir hosts of the hantaviruses in the western United States also act as hosts for the bacterium Yersinia pestis, the etiologic agent of plague. Although no evidence exists that fleas and other ectoparasites play a role in hantavirus epidemiology, rodent fleas transmit plague. Species of Peromyscus are susceptible to Y. pestis infection and can act as hosts for infected fleas. Control of rodents without concurrent control of fleas might therefore increase the risk of human plague as the rodent fleas seek an alternative food source. Transmission to HumansThe Old World hantaviruses causing HFRS, and the New World agents of HPS are believed to be transmitted by the same mechanisms. Human infection occurs most commonly through the inhalation of infectious, aerosolized saliva or excreta. Persons visiting laboratories where infected rodents were housed have been infected after only a few minutes of exposure to animal holding areas (22). Transmission can occur when dried materials contaminated by rodent excreta are disturbed and inhaled, directly introduced into broken skin or conjunctivae, or possibly, when ingested in contaminated food or water. Persons have also acquired HFRS and HPS after being bitten by rodents (23,24). High risk of exposure has been associated with entering or cleaning rodent-infested structures (25). Person-to-person transmission has not been associated with any of the Old World hantaviruses (26) or with HPS cases in the United States (27). However, person-to-person transmission, including nosocomial transmission of Andes virus, was well documented for a single outbreak in southern Argentina (28,29) and suspected to have occurred much less extensively in another outbreak in Chile associated with the same virus (30). EpidemiologyHantavirus infections are associated with domestic, occupational, or recreational activities that bring humans into contact with infected rodents, usually in rural settings. Known hantavirus infections of humans occur primarily in adults. HPS cases in the United States occur throughout the year, but greater numbers are reported in spring and summer. Hantavirus infection (resulting in HPS or HFRS) has been epidemiologically associated with the following situations (25,31--36):
However, in North America, the absolute risk of hantavirus infection to the general public is low; only 20--50 cases of HPS have been confirmed annually in the United States since the disease was described in 1993 (Figure 2). Physical Properties of HantavirusesHantaviruses have lipid envelopes that are susceptible to most disinfectants (e.g., dilute chlorine solutions, detergents, or most general-purpose household disinfectants) (37). Depending on environmental conditions, these viruses probably survive <1 week in indoor environments and much shorter periods (perhaps hours) when exposed to sunlight outdoors (38). PreventionEradicating the reservoir hosts of hantaviruses is neither feasible nor desirable because of the wide distribution of sigmodontine rodents in North America and their importance in the function of natural ecosystems. The best currently available approach for disease control and prevention is risk reduction through environmental modification and hygiene practices that deter rodents from colonizing the home and work environment, as well as safe cleanup of rodent waste and nesting materials. Controlled experiments have demonstrated that simple and inexpensive methods are effective in preventing rodents from entering rural dwellings (39). These recommendations emphasize the prevention of HPS associated with sigmodontine rodents in the Americas. Although the risk of acquiring hantavirus disease from contact with native arvicoline rodents in North America or introduced murine rodents throughout the Americas is low, the true pathogenicity for humans of all hantaviruses carried by these groups of rodents has not been established. Therefore, we recommend that persons avoid contact with all wild and peridomestic rats and mice. The precautions described in this report are broadly applicable to all groups of rats and mice. Precautions To Limit Household Exposure to RodentsRodent control in and around the home remains the primary strategy in preventing hantavirus infection. Rodent infestation can be determined by direct observation of animals, or inferred by observation of their nests or feces on floors or in protected areas (e.g., closets, kitchen cabinets, drawers, wall voids, furnace and hot water heating cabinets, and behind ventilation screens), or from evidence that rodents have been gnawing on food or other objects. The interior and exterior of the home should be carefully inspected at least twice per year for any openings where rodents could enter the home and for conditions that could support rodent activity. If any evidence of rodent infestation is detected inside the home or in outbuildings, precautions should be taken. The guidelines in the section Special Precautions for Homes of Persons with Confirmed Hantavirus Infection or Buildings with Heavy Rodent Infestations should be followed if a structure is associated with a confirmed case of hantavirus disease or if evidence of heavy rodent infestation is present (e.g., piles of feces or numerous nests or dead rodents). Recommendations are listed below for 1) reducing rodent shelter and food sources inside and outside the home and 2) preventing rodents from entering the home by rodent-proofing (40--42). Reduction of Rodent Shelter and Food Sources Inside and Outside the HomePrecautions for Inside the Home
In the western United States (west of the 100th meridian, a line from mid-Texas through mid-North Dakota), a risk of plague transmission to humans from fleas exists. Use insect repellent (containing N,N-diethyl-m-toluamide [DEET]) on clothing, shoes, and hands to reduce the risk of fleabites when picking up dead rodents and traps. In cases of heavy rodent infestation in indoor spaces in the western United States, use an insecticide before trapping. Contact your local or state health department to find out if plague is a danger in the area and for additional advice on appropriate flea-control methods.
When resident mice are removed from rural buildings without measures to prevent reentry, they are replaced almost immediately by other mice from the outside. Therefore, indoor rodent-trapping could be unsuccessful in reducing rodent infestations without simultaneous efforts to rodent-proof permeable dwellings. Precautions for Outside the Home
Preventing Rodents from Entering the Home by Rodent-Proofing
Precautions To Limit Occupational and Recreational Exposure to RodentsPrecautions for Workers Frequently Exposed to RodentsPersons who frequently handle or are exposed to wild rodents are probably at higher risk for hantavirus infection than the general public because of the frequency of their exposures. Such persons include, but are not limited to, mammalogists, pest-control workers, some farm and domestic workers, and building and fire inspectors. Therefore, enhanced precautions are warranted to protect them against hantavirus infection, as described below.
Precautions for Other Occupational Groups Having Potential Contact with RodentsInsufficient information is available to provide general recommendations regarding risks and precautions for persons who work in occupations with unpredictable or incidental contact with rodents or their nesting sites. Examples of such occupations include telephone installers, maintenance workers, plumbers, electricians, and certain construction workers. Workers in these jobs might have to enter buildings, crawl spaces, or other sites that are potentially rodent-infested, and HPS has been reported among these workers. Recommendations for such circumstances must be made on a case-by-case basis after the specific working environment has been assessed and state or local health and labor officials or trade unions and management, as appropriate, have been consulted. Determining the level of risk present and implementing appropriate protective measures is the employer's responsibility.*** Precautions for Campers and HikersNo evidence exists to suggest that travel should be restricted in areas where HPS cases have occurred. The majority of typical tourist activities are associated with limited or no risk that travelers will be exposed to rodents or their excreta. However, persons engaged in outdoor activities (e.g., camping or hiking) should take precautions to reduce the likelihood of exposure to potentially infectious materials by following these recommendations.
Precautions for Cleanup of Rodent-Contaminated Areas and Dead RodentsAreas with evidence of rodent activity (e.g., dead rodents and rodent excreta) should be thoroughly cleaned to reduce the likelihood of exposure to hantavirus-infected materials. Cleanup procedures must be performed in a manner that limits the potential for dirt or dust from contaminated surfaces to become airborne. Recommendations are listed in this report for cleaning up 1) rodent urine and droppings, and surfaces potentially contaminated by rodents and 2) dead rodents and rodent nests. Cleanup of Rodent Urine and Droppings and Contaminated Surfaces
Cleanup of Dead Rodents and Rodent Nests
Disinfecting Solutions Two types of disinfecting solutions are recommended to clean up rodent materials. 1. General-Purpose Household Disinfectant --- Prepare according to the label, if not prediluted. Almost any agent commercially available in the United States is sufficient as long as the label states that it is a disinfectant. Effective agents include those based on phenols, quaternary ammonium compounds, and hypochlorite. 2. Hypochlorite Solution --- A chlorine solution, freshly prepared by mixing 1½ cups of household bleach in 1 gallon of water (or a 1:10 solution) can be used in place of a commercial disinfectant. When using chlorine solution, avoid spilling the mixture on clothing or other items that might be damaged by bleach. Wear rubber, latex, vinyl, or nitrile gloves when preparing and using chlorine solutions. Chlorine solutions should be prepared fresh daily. Cleaning Sheds and Other Outbuildings Before cleaning closed sheds and other outbuildings, ventilate the building by opening doors and windows for at least 30 minutes. Use cross ventilation if possible. Leave the area during the airing-out period. This airing helps to remove infectious primary aerosols that might be created when hantavirus-infected rodents urinate. In substantially dirty or dusty environments, additional protective clothing or equipment may be worn. Such equipment includes coveralls (disposable when possible) and safety glasses or goggles, in addition to rubber, latex, vinyl, or nitrile gloves. For recommendations regarding precautions for cleanup of outbuildings with heavy rodent infestations, see Special Precautions for Homes of Persons with Confirmed Hantavirus Infection or Building with Heavy Rodent Infestations. Special Precautions for Homes of Persons with Confirmed Hantavirus Infection or Buildings with Heavy Rodent InfestationsSpecial precautions are indicated for cleaning homes or buildings with heavy rodent infestations. A rodent infestation is considered heavy if piles of feces or numerous nests or dead rodents are observed. Persons cleaning these homes or buildings should contact the local or state public health agency or CDC for guidance. These precautions also can apply to vacant dwellings that have attracted rodents while unoccupied and to dwellings and other structures that have been occupied by persons with confirmed hantavirus infection. Workers who are either hired specifically to perform the cleanup or asked to do so as part of their work activities should receive a thorough orientation from the responsible health agency or employer about hantavirus transmission and disease symptoms and should be trained to perform the required activities safely. Recommendations for Cleaning Homes or Buildings with Heavy Rodent Infestations
Applicability and UpdatesThe control and prevention recommendations in this report represent general measures to minimize the likelihood of human exposure to hantavirus-infected rodents in the Americas. Although different geographic areas might have varying housing types and rodent populations, the precautions should be the same. The effect and utility of the recommendations will be continually reviewed by CDC and the involved state and local health agencies as additional epidemiologic, field, and laboratory data become available. These recommendations might be supplemented or modified in the future. These recommendations and additional information concerning hantaviruses are periodically updated and made available on CDC's website, All About Hantaviruses (http://www.cdc.gov/ncidod/disease/hanta/hps/index.htm). Additional information can be obtained by contacting CDC, National Center for Infectious Diseases (NCID), Special Pathogens Branch, Mailstop A-26, 1600 Clifton Road, N.E., Atlanta, GA 30333; e-mail dvd1spath@cdc.gov; fax 404-639-1509; or by telephone 404-639-1510. Acknowledgments This update is based on interim recommendations from James E. Childs, Sc.D.; Arnold F. Kaufmann, D.V.M.; and Clarence J. Peters, M.D., NCID; and Richard L. Ehrenberg, M.D., National Institute for Occupational Safety and Health. Graphics were produced by Amera R. Khan, M.P.H.; and Kent D. Wagoner, Ph.D., NCID. Jacob H. Creswell, M.P.H.; Kimberly C. Dills, M.P.H.; and Emily S. Jentes, NCID, provided technical assistance and careful reviews of the manuscript. Kathleen F. Cavallaro, M.S., NCID, helped coordinate numerous aspects of the manuscript preparation. The preparation of this document was supported in part by an appointment to the Research Participation Program at CDC's NCID, Division of Viral and Rickettsial Diseases, administered by the Oak Ridge Institute for Science and Education, Oak Ridge, TN, through an interagency agreement between the U.S. Department of Energy and CDC. References
* Updated HPS information is available at http://www.cdc.gov/ncidod/diseases/hanta/hps/noframes/caseinfo.htm. † A rodent-proof container is made of thick plastic, glass, or metal and has a tight-fitting lid. § Only spring-loaded traps that kill rodents should be used. Live and sticky traps that do not kill rodents are not recommended. Infectious aerosols might be produced when live rodents urinate or struggle to free themselves. Disposal of live rodents also introduces the risk of rodent bite, which also can result in infection. ¶ See Precautions for Cleanup of Rodent-Contaminated Areas and Dead Rodents for detailed information regarding making a chlorine solution. ** Follow the recommendations specified in the section Precautions for Cleanup of Rodent-Contaminated Areas and Dead Rodents, if rodent urine, droppings, nests, or dead rodents are encountered while these measures are being carried out. †† Lath screen or metal is a light-gauge metal mesh and is commonly installed over wooden walls before plaster is applied. A galvanized product is preferable. Lath screen is malleable and can be folded and pushed into larger holes. These materials can be found in the masonry or building materials section at hardware or building supply stores. §§ STUF-FIT is a soft copper-mesh material that might be preferable to steel wool because it does not rust and is not easily pulled apart by rodents. It can be obtained from pest control retail stores or from Allen Special Products (telephone 800-848-6805). ¶¶ Illustrated, complete instructions for rodent-proofing are available 1) in the National Park Service's manual, Mechanical Rodent Proofing Techniques; 2) on CDC's website, All About Hantaviruses (http://www.cdc.gov/ncidod/diseases/hanta/hps/index.htm); and 3) from CDC's Ramah Home Seal-up protocol, Special Pathogens Branch (e-mail dvd1spath@cdc.gov). *** General Duty Clause, Occupational Safety and Health Act of 1970, Section 5(a)(1); http://www.osha.gov/OshAct_data/OSHACT.htm#5. Consultants The following persons provided consultation in the development of the previous or current versions of these recommendations: Michael A. Bogan, Ph.D., U.S. Fish and Wildlife Service, Fort Collins, CO; Patrick O. Bohan, M.S., Indian Health Service, Navajo Regional Office, Window Rock, AZ; Ted L. Brown, M.S., New Mexico Department of the Environment, Sante Fe, NM; James E. Cheek, M.D., Indian Health Service, Albuquerque, NM; Richard M. Davis, D.Sc., California Department of Health Services, Ventura, CA; John Doll, Ph.D., Arizona Department of Health Services, Phoenix, AZ; Richard L. Ehrenberg, M.D., National Institute for Occupational Safety and Health, Atlanta, GA; Eric Esswein, M.S.P.H., National Institute for Occupational Safety and Health, Denver, CO; Kathleen A. Fagerstone, Ph.D., U.S. Department of Agriculture, Denver, CO; William Ferraro, Philadelphia Department of Public Health, Philadelphia, PA; Curtis L. Fritz, D.V.M., Ph.D., California Department of Health Services, Sacramento, CA; Gregory Glass, Ph.D., Department of Molecular Microbiology and Immunology, Johns Hopkins School of Hygiene and Public Health, Baltimore, MD; Marcia Goldoft, M.D., Washington State Department of Health, Seattle, WA; Jerry M. Hershovitz, Division of Emergency and Environmental Health Services, National Center for Environmental Health, Atlanta, GA; Randall B. Hirschhorn, M.S., Philadelphia Department of Public Health, Philadelphia, PA; Gerry Hoddenbach, M.S., Hoddenbach Consulting, Torrey, UT; William Jackson, Ph.D., Bowling Green State University, Bowling Green, OH; Karl Johnson, M.D., University of New Mexico, Albuquerque, NM; James W. LeDuc, Ph.D., Division of Viral and Rickettsial Diseases, National Center for Infectious Diseases, Atlanta, GA; Craig R. Nichols, M.P.H., Utah Department of Health, Salt Lake City, UT; Mark G. Novak, Ph.D., California Department of Health Services, Sacramento, CA; Robert Parmenter, Ph.D., University of New Mexico, Albuquerque, NM; Timothy M. Radtke, M.S., Office of Occupational Safety and Health, U.S. Department of the Interior, Denver, CO; Robert N. Reynolds, M.S., U.S. Department of Agriculture, Lakewood, CO; Luis Ruedas, Ph.D., Division of Viral and Rickettsial Diseases, National Center for Infectious Diseases, Atlanta, GA; Connie S. Schmaljohn, Ph.D., U.S. Army Medical Research Institute for Infectious Diseases, Fort Detrick, MD; Herman F. Shorty, Navajo Nation Office of Environmental Health, Window Rock, AZ; Thomas M. Smylie, U.S. Department of Agriculture, Albuquerque, NM; Rosemary Sokas, M.D., National Institute for Occupational Safety and Health, Washington, D.C.; Dale T. Tanda, Colorado Department of Health, Denver, CO; Ronald E. Vorhees, M.D., New Mexico Department of Health, Sante Fe, NM; Richard Yanagihara, M.D., National Institutes of Health, Bethesda, MD. Figure 1  Return to top. Figure 2 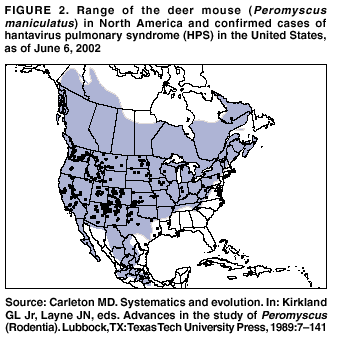 Return to top. Figure 3  Return to top. Figure 4 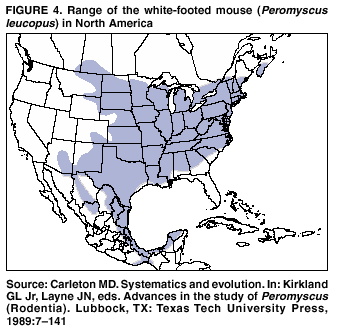 Return to top. Figure 5  Return to top. Figure 6 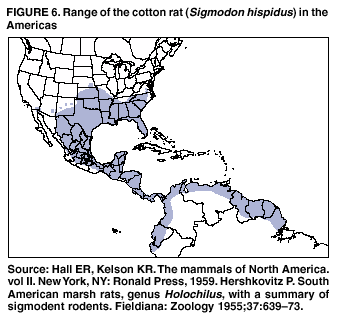 Return to top. Figure 7  Return to top. Figure 8 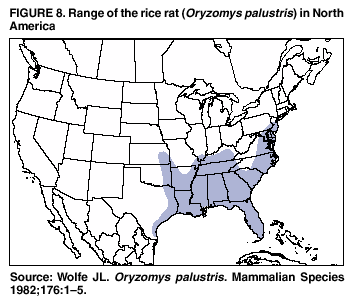 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 7/12/2002 |
|||||||||
This page last reviewed 7/12/2002
|