Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Human Vaccinia Infection After Contact with a Raccoon Rabies Vaccine Bait --- Pennsylvania, 2009
Since 2003, the U.S. Department of Agriculture's Wildlife Services has coordinated a multistate oral rabies vaccination (ORV) program for wildlife in a 15-state zone extending from Maine to Alabama and in Texas. The program seeks to enhance local control and prevent the spread of epizootic rabies among raccoons and, in Texas, among gray foxes and coyotes. The program uses baits containing liquid vaccinia-rabies glycoprotein (V-RG) recombinant virus vaccine. Because contact with ruptured baits can produce vaccinia virus infection in certain persons, surveillance for human and domestic animal contact with the baits is conducted, relying largely on reports from persons who find baits and call telephone numbers printed on them. In August 2009, during the autumn baiting campaign in western Pennsylvania, a woman aged 35 years who was taking immunosuppressive medication for inflammatory bowel disease contacted the Pennsylvania Department of Health (PADOH) after handling a ruptured bait, which had leaked liquid rabies vaccine onto a patch of abraded skin on her right hand. The patient subsequently developed vaccinia virus infection and was treated with human vaccinia immune globulin intravenous (VIGIV) and an investigational antiviral agent. This report describes this case, which was the second case of human vaccinia infection related to the ORV program. Public health agencies should educate the public, and particularly pet owners, regarding potential hazards associated with handling wildlife rabies vaccine baits and should provide guidance for persons exposed to this vaccine.
The ORV program uses an orally delivered animal vaccine, Raboral V-RG (Merial, Inc., Athens, Georgia) (1). This vaccine consists of a partially attenuated recombinant vaccinia virus (Copenhagen strain) with a gene encoding for rabies virus glycoprotein (2). Persons with immunosuppressive conditions or exfoliative dermatologic conditions are at greater risk for complications (e.g., progressive vaccinia or eczema vaccinatum) when exposed to the vaccinia virus contained in Raboral V-RG vaccine. Currently, 15 states distribute ORV baits for raccoons, and Texas distributes baits for gray foxes and coyotes. The V-RG vaccine is delivered via a bait made from a fishmeal block (or a dog food block for coyotes) encasing a sealed plastic packet containing approximately 2 mL of vaccine (Figure 1) (1). During August-September 2009, ORV bait distribution was conducted in nonresidential areas by airplane and in rural communities by hand, in parts of western Pennsylvania, Ohio, and West Virginia.
Case Report
In early August 2009, a woman aged 35 years was picking blackberries in a rural area of Pennsylvania where ORV baits recently had been distributed. Her dog picked up a bait in his mouth and punctured the bait with his teeth. After the dog dropped the bait, the woman picked it up, and V-RG vaccine dripped from the bait onto her right hand and wrist, including sites that had been abraded by blackberry thorns. Approximately 30 minutes passed before the woman was able to wash her hands. She telephoned the ORV helpline number printed on the bait, and her call was referred to PADOH. The woman had a history of inflammatory bowel disease, for which she was taking multiple immunosuppressive medications. These medications included a suppressor of T and B cell proliferation (taken daily) and a tumor necrosis factor-alpha (TNF-alpha) blocker (taken once every 6 weeks, with the last dose on July 13); she had no smallpox vaccination scar and reported no history of smallpox vaccination.
On day 1 after her exposure to the V-RG vaccine, her physician obtained a baseline serum specimen for analysis of rabies virus neutralizing antibody (by direct virus neutralization) and orthopoxirus (vaccinia) immunoglobulin IgG and immunoglobulin IgM antibodies (by enzyme-linked immunosorbent assay). In addition, photos were taken of her right hand abrasions, and she was instructed to watch for signs of infection in her hand. On day 4, she reported several red papules on her right hand. She visited a local hospital emergency department, where scrapings of the papules were obtained and submitted to the PADOH Bureau of Laboratories. The woman was discharged from the emergency department and advised to stop taking her immunosuppressant medications.
On day 5, a real-time polymerase chain reaction (PCR) assay of the scrapings tested positive for nonvariola Orthopoxvirus DNA. Subsequent testing performed at CDC confirmed the presence of vaccinia virus DNA and rabies virus G protein DNA in papule material and serologic evidence of rabies virus neutralizing antibodies. Neither Orthopoxvirus IgG nor IgM antibodies were detected.
On day 6, the papules had increased in number and size, and the patient was hospitalized. Physical examination on admission showed multiple papules on erythematous bases on the patient's right hand, wrist, and arm, including three that appeared to be early vesicles. Because of the patient's immune suppression and concerns about progressive vaccinia, she was administered a single dose (6,000 IU/kg) of VIGIV (Cangene Corporation, Winnipeg, Canada) provided by CDC. Contact precautions were utilized while the patient was hospitalized.
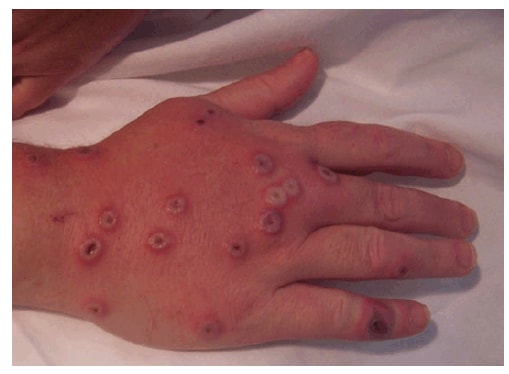
On day 9, she had 26 classic vaccinia virus lesions, including one on a site on her right arm that might not have been in contact with the vaccine initially. The patient was afebrile and not experiencing symptoms related to her inflammatory bowel disease. However, on day 11, she experienced myalgia and headache; she had pronounced redness and edema in her right hand (Figure 2), accompanied by right axillary adenopathy. The patient remained afebrile, but was taking an antipyretic pain medication. At that time, her physician decided that to prevent exacerbation of her inflammatory bowel disease the patient soon would need to restart her immunosuppressive medications. On day 12, in an attempt to reduce viral replication and prevent progressive vaccinia, a second 6,000 IU/kg dose of VIGIV was administered along with the investigational antiviral agent, ST-246 (SIGA Technologies, Corvalis, Oregon). The ST-246 was administered orally for 14 days after a compassionate use authorization was approved by the Food and Drug Administration.
On day 13, the patient began receiving phased reintroduction of her immunosuppressive medications. She was discharged on day 19. By day 28, all scabs from her lesions had separated and her underlying inflammatory bowel disease condition was stable. The patient remained Orthopoxvirus IgM negative throughout her illness.
Reported by: V Dato, MD, C Moose, N Rea, PhD, G Fraser, J Seiders, C Rittle, MPH, V Urdaneta, MD, S Ostroff, MD, Pennsylvania Dept of Health. C Encarnacion, MD, Pennsylvania. M Reynolds, PhD, I Damon, MD, K Karem, PhD, Y Li, PhD, W Davidson, MPH, K Wilkins, E McDowell, CE Rupprecht, VMD, L Orciari, MS, M Niezgoda, MS, S Smith, MS, Div of Viral and Rickettsial Diseases, National Center for Zoonotic, Vector-Borne, and Enteric Diseases; A Roess, PhD, EIS Officer, CDC.
Editorial Note:
The patient described in this report is only the second person documented with human vaccinia infection associated with the ORV baiting program, and the first infected person taking immunosuppressive medications. The one previous documented human vaccinia infection occurred in September 2000 in a pregnant woman aged 28 years with eczema who was bitten while pulling a ruptured bait from her dog's mouth (2). The patient in the current report was of particular concern for three reasons. First, she was immunosuppressed from medications to treat her inflammatory bowel disorder. Second, she had fresh abrasions and prolonged contact with V-RG vaccine (approximately 30 minutes) before washing her hands. Third, she did not exhibit features of a normal immune response for a person previously naive for Orthopoxvirus infection (i.e., an IgM antibody response was not detected.) Careful monitoring and prompt diagnostic evaluation allowed for timely medical interventions, and the vaccinia virus infection resolved. Although her treatment with 2 doses of VIGIV and ST-246 might have contributed to her recovery, the role of these agents is difficult to assess.
Since 1990, approximately 100 million doses of Raboral V-RG animal vaccine have been distributed in the United States by depositing baits by airplane or by hand in areas with dense populations of target wildlife species (e.g., raccoons in the eastern United States). Human or domestic animal contact with ORV baits has been reported rarely. In 2008, among 16 states where 10,339,969 baits were placed, 291 vaccine-related telephone calls involving human or domestic animal contact (2.8 per 100,000 baits) were reported by public health authorities (3).
The ORV packets containing the V-RG vaccine are durable and typically do not leak unless punctured. Both documented instances of human vaccinia virus infections caused by V-RG occurred after baits were found and punctured by dogs. Humans interacting with domestic animals (typically dogs, but occasionally cats) that find and eat baits are one of the most common sources of potential vaccine exposures (4). The Pennsylvania case described in this report underscores the importance of continuing to alert the public, particularly pet owners, during baiting season to the possibility of pets finding and ingesting baits, or carrying them into the home. Raboral V-RG has been tested for safety in dogs. Owners should not attempt to remove baits from a dog's mouth. Gloves or plastic bags should be used to pick up and examine baits if necessary and additional baits that might be picked up by dogs or other pets should be removed from the immediate area. Interviews with all persons who contact the ORV program helpline printed on the baits can enable early identification of persons at high risk for vaccinia virus infection. All callers should be asked about the nature of their contact with the bait and should be counseled to report any skin lesions. Persons with eye exposure should irrigate the eye with clean water for 15 minutes and should be referred promptly for an ophthalmologic examination. In the event of a documented infection, care should be taken to avoid transmission of the virus to household and other contacts of the patient (5).
Persons with a history of atopic dermatitis or other active exfoliative skin conditions and those with immune deficiencies or immunosuppressive conditions are at greater risk for adverse outcomes (e.g., eczema vaccinatum and progressive vaccinia) after infections with nonhighly attenuated variants of vaccinia virus (6), including Raboral V-RG (7). Health-care practitioners should collect specimens promptly from the earliest signs of rash and small papules and submit them to an approved laboratory for testing for vaccinia virus by real-time PCR. Most state health department laboratories and others participating in the Laboratory Response Network are equipped to test clinical specimens for the presence of nonvariola Orthopoxvirus DNA.
References
- US Department of Agriculture Animal and Plant Health Inspection Service. Wildlife Services: National Rabies Management Program. Oral rabies vaccination. Washington, DC: USDA; 2009. Available at http://www.aphis.usda.gov/ws/rabies/vaccine.html. Accessed October 30, 2009.
- Rupprecht CE, Blass L, Smith K, et al. Human infection due to recombinant vaccinia-rabies glycoprotein virus. N Engl J Med 2001;345:582--6.
- Roess AA, Rea N, Dato V, Rupprecht C; ORV Surveillance Network. National surveillance for human and pet contact with oral rabies vaccine (ORV) baits, 2008. Presented at: XX International Conference on Rabies in the Americas, Quebec City, Canada, October 17--23, 2009.
- McGuill MW, Kreindel SM, DeMaria A, et al. Human contact with bait containing vaccine for control of rabies in wildlife. J Am Vet Med Assoc 1998;213:1413--7.
- Sepkowitz KA. How contagious is vaccinia? N Eng J Med 2003;348:439--46.
- CDC. Vaccinia (smallpox) vaccine: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 2001;50(No. RR-10).
- Hanlon CA, Niezgoda M, Shankar V, Niu HS, Koprowski H, Rupprecht CE. A recombinant vaccinia-rabies virus in the immunocompromised host: oral innocuity, progressive parenteral infection, and therapeutics. Vaccine 1997;15:140--8.
|
What is already known on this topic? Cases of human vaccinia virus infection after contact with oral rabies vaccination (ORV) program baits are rare, but pose serious risks to persons with a history of eczema, immune deficiencies, or immunosuppressive conditions. What is added by this report? This report describes the second documented case in 9 years of human vaccinia virus infection related to a multistate ORV program and reinforces the need to educate the public, particularly pet owners, regarding potential hazards associated with handling wildlife rabies vaccine baits. What are the implications for public health practice? Public health agencies should educate persons to call the telephone number printed on any ORV bait they touch, and health-care practitioners should test such persons for vaccinia promptly if they develop signs of the disease. |
FIGURE 1. Rabies vaccine delivered to raccoons via a bait consisting of a fishmeal block (left) encasing a sealed plastic packet containing the vaccine (right) --- Pennsylvania, 2009

Photo/U.S. Department of Agriculture Wildlife Services
Alternative Text: The figure above is a photograph of a gloved hand holding a raccoon rabies vaccine bait, which consists of a fishmeal block and the sealed plastic packet that is encased inside the block and contains the vaccine.
FIGURE 2. Female patient aged 35 years exhibits vaccinia virus lesions and pronounced redness and edema on her right hand on day 11 after contact with an animal rabies vaccine bait --- Pennsylvania, 2009
Photo/C. Encarnacion, MD
Alternative Text: The figure above is a photograph showing the right hand of a woman aged 35 years with vaccinia virus lesions and pronounced redness and edema on day 11 after contact with a raccoon rabies vaccine bait in Pennsylvania in 2009.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services. |
All MMWR HTML versions of articles are electronic conversions from typeset documents. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 11/5/2009


