Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Bubonic and Pneumonic Plague --- Uganda, 2006
Plague is a life-threatening fleaborne disease caused by the bacterium Yersinia pestis. The most common clinical form is bubonic plague, which is characterized by high fever and regional lymphadenitis. Without treatment, infection can spread from lymph nodes to the lungs, resulting in pneumonic plague and the potential for person-to-person transmission through respiratory droplets (1,2). In November 2006, the Uganda Ministry of Health received reports of an increase in bubonic plague cases and a possible outbreak of pneumonic plague among residents in the Arua and Nebbi districts. In response, the Uganda Ministry of Health and CDC conducted a joint investigation in the two districts during November 28--December 30, 2006. Overall, 127 clinical plague cases were identified, along with evidence of a focal pneumonic outbreak in Nebbi District. Median age of the patients was 14 years (range: 2 weeks--65 years); 65 (51%) were female. Twenty-eight (22%) of the 127 patients died. Among the 102 patients with documented symptoms, 90 (88%) had bubonic plague, and 12 (12%) had pneumonic plague. The results of this investigation underscore the need to 1) continue efforts to educate residents of rural Uganda regarding the source, signs, and symptoms of plague and the life-saving importance of seeking treatment; 2) strengthen plague surveillance and diagnostic capabilities; and 3) improve emergency response and vector-control capacity, especially in remote regions of the country.
In rural Uganda, where laboratory capacity is limited, clinicians generally rely on clinical criteria for the diagnosis of plague. These criteria, as established by the Uganda Ministry of Health, are as follows: sudden onset of fever, chills, malaise, headache, or prostration accompanied by either painful regional lymphadenitis (bubonic plague) or cough with hemoptysis (pneumonic plague). For this investigation, a plague case was defined as clinically diagnosed plague with onset during July 1--December 30, 2006, in a resident of Arua or Nebbi districts. Beginning November 28, cases were ascertained through retrospective review of patient logs in eight clinics and two hospitals that historically have accounted for 85% of reported plague cases in the region (Uganda Virus Research Institute, unpublished data, 1999--2005). Information was collected on patient age, sex, village of residence, clinical presentation, and outcome. In addition, clinicians at these facilities and all other health clinics in Arua and Nebbi districts were asked to report immediately by cellular telephone or messenger any new cases identified. When possible, diagnostic samples (i.e., serum and bubo aspirates) were collected from patients with acute or recent illness diagnosed as plague. Laboratory-confirmed plague was defined as isolation of Y. pestis or a four-fold change in antibody titer to F1 antigen between paired acute and convalescent serum samples with a least one sample having a titer ≥1:16.
A total of 127 plague cases with onset dates during July 19--December 30, 2006 (Figure 1), were indentified in Arua and Nebbi districts in northwestern Uganda (Figure 2). Among the 102 patients with documented symptoms, 90 (88%) had bubonic plague, and 12 (12%) had pneumonic plague. Two or more plague cases were reported from nine different villages, including four villages that reported 10 or more cases (Nave, 18; Kestro, 18; Andosi, 17; and Yiapi, 10) The median patient age was 14 years (range: 2 weeks--65 years); 65 patients (51%) were female (Table).
Of 11 pneumonic plague deaths, six (55%) occurred in one village, in which four members of a single family died. Interviews with village residents indicated that the index case in that family was in a boy aged 10 years who developed fever and a cervical bubo, followed by hemoptysis; he died on November 3. Within 7 days, the child's mother, grandmother, and aunt, all of whom had cared for the child, also developed hemoptysis and died. Initially, ill village residents did not seek health care because they believed the illness was of supernatural origin stemming from a local feud. However, when two additional village members not involved in the feud died of similar symptoms on November 8, ill villagers began seeking care at the health clinic, where they were treated with appropriate antibiotics and recovered.
Bubo aspirates were obtained from 11 (12%) of 90 patients with bubonic manifestations. None of the cases met the criteria for laboratory confirmation. Y. pestis was not recovered from any of the 11 aspirates; however, eight of the 11 patients had been treated with antibiotics before sample collection. Paired acute and convalescent blood samples were obtained from seven (6%) of the 127 patients and convalescent blood samples alone from another 31 (24%). No paired serum samples had a four-fold change in antibody titer. However, convalescent sera from five (13%) of the 38 total patients with convalescent samples had single titers that ranged from 1:16 to 1:256 (median: 1:64), which is suggestive of recent or previous Y. pestis infection. Samples were not available from any of the 12 patients who received a diagnosis of pneumonic plague; 11 of these patients died before the investigation began.
To evaluate access to care, patient behaviors, and potential plague exposures, structured interviews were conducted during December 1--16 with a convenience sample of 39 plague patients in six Arua and Nebbi villages. Dates of illness onset for interviewed patients were from October 13 to December 14. Twenty-seven patients (69%) reported that they walked to the local health clinic for treatment of plague symptoms, and 12 (31%) rode a bicycle. Fifteen patients (38%) said travel to the nearest health clinic took >2 hours, and 24 patients (62%) said travel took ≤2 hours. Seventeen (44%) of the interviewed patients reported taking medications, including acetaminophen, chloroquine, or traditional herbs, obtained in the village before visiting the clinic. Twenty-three patients (59%) reported seeing dead rats in their homes during the 2 weeks preceding their illness; some villagers reported recently finding and burying dead rats near their homes.
While visiting villages, investigators in the two districts recovered eight dead rats (Rattus rattus), of which four tested positive for Y. pestis by direct fluorescent antibody staining and two tested positive by culture isolation. Live R. rattus species trapped in two affected villages were found to have an average of two fleas per rat; recovery of more than one flea per rat has been associated with increased risk for plague transmission (3). On December 8, 2006, vector-control teams comprised of local villagers began applying dichlorvos, a residual insecticide, to households in affected villages in an effort to interrupt plague transmission. By December 14, the teams had treated 935 houses in 10 Arua District villages that had reported two or more plague cases since September 1. The death of the vector-control team leader from causes unrelated to plague prevented expansion of the spraying operation to affected villages in Nebbi District.
Reported by: A Ogen-Odoi, E Katangole Mbidde, Uganda Virus Research Institute; J Lutwama, MD, J Wamala, MD, A Mucunguzi, MD, M Mugagga, A Kagirita, L Lukwago, M Musenero Musanza, MD, A Talisuna, MD, J Turyagaruki, MD, Uganda Ministry of Health; J Kirungi, Masindi District Health Office; O Namusisi, MD, African Field Epidemiology Network; R Downing, PhD, J Tappero, MD, CDC Uganda. R Enscore, MS, M Schriefer, PhD, S Bearden, PhD, C Sexton, E Zielinski-Gutierrez, DrPH, K Griffith, MD, P Mead, MD, Div of Vector-Borne Infectious Diseases, National Center for Zoonotic, Vector-Borne, and Enteric Diseases; M Duffy, DVM, EIS Officer, CDC.
Editorial Note:
African countries accounted for nearly 90% of the 28,530 plague cases reported to the World Health Organization during the most recent 10-year reporting period (1994--2003) (4). In Uganda, 200--400 clinically diagnosed plague cases are reported annually, with an estimated case-fatality rate of 30% (4). Although human cases typically occur sporadically or in small clusters, the potential for pneumonic outbreaks with spread to other areas is a great concern (5,6). Approximately 6 weeks after this investigation, an outbreak of pneumonic plague with seven deaths was reported from Masindi District, a neighboring but ecologically distinct region where plague is not endemic. The index patient was an adolescent girl aged 15 years who had become ill while visiting relatives in a plague-affected area of Nebbi District.
The investigation described in this report highlights some of the challenges associated with indentifying and controlling plague in rural Africa. Because of limited laboratory capacity, clinicians usually rely on clinical criteria alone when diagnosing illnesses. Consequently, patients with other causes of acute lymphadenitis (e.g., staphylococcal) can be misdiagnosed as having plague, and patients with plague can be misdiagnosed as having other illnesses. When laboratory services are available, impassible roads and other logistic barriers can prevent timely specimen collection. Patients with less serious conditions might survive long enough to be tested; however, patients with more severe Y. pestis infection are likely to die before specimens can be collected. Such differential testing might have contributed to the low rate of seropositivity observed in this investigation among those patients whose specimens were tested. Greater regional laboratory capacity and point-of-care diagnostic assays would help clinicians and health officials identify plague cases more quickly, distinguish these cases from other causes of similar illness, and facilitate more effective control of plague in Africa.
Also highlighted by this investigation is the effect of local beliefs on care-seeking behavior. Anthropologic studies have indicated that tribes living in Arua District hold diverse beliefs about disease causation and the role of the supernatural in human illness (7). Investigators observed that villagers initially ascribed an outbreak of pneumonic plague to feuding rather than to a disease that can be treated with antibiotics. Because prompt antimicrobial treatment can be life-saving for patients with plague, further studies are needed to 1) define local beliefs as they relate to plague, 2) determine how they influence care-seeking, and 3) identify effective messages that will result in plague patients seeking care at their local health center more quickly.
To enhance recognition, treatment, and control of plague in Uganda, CDC has entered into a cooperative agreement with the Uganda Ministry of Health and the Uganda Viral Research Institute. Studies are under way to 1) evaluate rapid, point-of-care diagnostic assays for plague, 2) describe belief systems that influence health-care--seeking behavior, 3) define the relative importance of various rodent and fleas species in plague transmission, and 4) assess opportunities for integrating flea control into existing vector-control programs (e.g., indoor residual spraying for malaria prevention). The overarching goal of this multidisciplinary effort is to create effective prevention programs and develop and exercise local contingency plans for plague outbreak response.
Acknowledgment
This report is based, in part, on contributions by R Mbabaz, Kiryandongo Hospital, Masindi District, Uganda.
References
- Perry RD, Fetherston JD. Yersinia pestis---etiologic agent of plague. Clin Microbiol Rev 1997;10:35--66.
- CDC. Prevention of plague: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 1996;45(No. RR-14).
- Politzer R. Plague. World Health Organization monograph series. No. 22. Geneva, Switzerland: World Health Organization; 1954.
- World Health Organization. Human plague in 2002 and 2003. Wkly Epidemiol Rec 2004;79:301--6.
- Outbreak news. Plague, Democratic Republic of the Congo. Wkly Epidemiol Rec 2006;81:241--2.
- Ratsitorahina M, Chanteau S, Rahalison L, Ratsifasoamanana L, Boisier P. Epidemiological and diagnostic aspects of the outbreak of pneumonic plague in Madagascar. Lancet 2000;355:111--3.
- Barnes-Dean V. Lugbara illness beliefs and social change. Africa: Journal of International African Institute 1986;56:334--51.
FIGURE 1. Number of plague cases (N = 127),* by week of onset and outcome --- Arua and Nebbi districts, Uganda, July 1--December 30, 2006
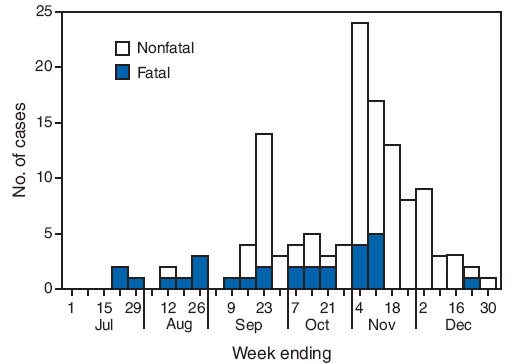
* Defined as sudden onset of fever, chills, malaise, headache, or prostration accompanied by either painful regional lymphadenitis (bubonic plague) or cough with hemoptysis (pneumonic plague).
Alternative Text: The figure above shows the 127 plague cases by week of onset and outcome during July 1-December 30, 2006 in Arua and Nebbi districts, Uganda. The outbreak, which claimed 28 lives, peaked during the first week of November.
Alternative Text: The figure above is a map showing the location of Arua and Nebbi districts in northwestern Uganda.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services. |
All MMWR HTML versions of articles are electronic conversions from typeset documents. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 7/23/2009


