 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Acute Hepatitis C Virus Infections Attributed to Unsafe Injection Practices at an Endoscopy Clinic --- Nevada, 2007On January 2, 2008, the Nevada State Health Division (NSHD) contacted CDC concerning surveillance reports received by the Southern Nevada Health District (SNHD) regarding two persons recently diagnosed with acute hepatitis C. A third person with acute hepatitis C was reported the following day. This raised concerns about an outbreak because SNHD typically confirms four or fewer cases of acute hepatitis C per year. Initial inquiries found that all three persons with acute hepatitis C underwent procedures at the same endoscopy clinic (clinic A) within 35--90 days of illness onset. A joint investigation by SNHD, NSHD, and CDC was initiated on January 9, 2008. The epidemiologic and laboratory investigation revealed that hepatitis C virus (HCV) transmission likely resulted from reuse of syringes on individual patients and use of single-use medication vials on multiple patients at the clinic. Health officials advised clinic A to stop unsafe injection practices immediately, and approximately 40,000 patients of the clinic were notified about their potential risk for exposure to HCV and other bloodborne pathogens. This report focuses on the six cases of acute hepatitis C identified during the initial investigation, which is ongoing; additional cases of acute hepatitis C associated with exposures at clinic A might be identified. Comprehensive measures involving viral hepatitis surveillance, health-care provider education, public awareness, professional oversight, licensing, and improvements in medical devices can help detect and prevent transmission of HCV and other bloodborne pathogens in health-care settings. The objectives of the investigation were to conduct case-finding and review health histories of infected persons, to determine the source of transmission and implement control measures, to identify other patients at risk for exposure, and to assist in development of recommendations to prevent HCV transmission in health-care settings. Persons with acute hepatitis C were interviewed, and blood samples were obtained after these persons gave oral consent. Blood samples were sent to CDC for testing for HCV genotype at the NS5b region and phylogenetic relatedness at the hypervariable 1 region (HVR1) to help determine whether a common source of transmission existed (1). Specimens also were tested for other bloodborne infections (hepatitis B virus [HBV]) and human immunodeficiency virus [HIV]). Case-finding activities included SNHD's review of acute hepatitis C surveillance records, cross-matching of local HCV laboratory records with clinic A procedure logs, review of medical records for patients who underwent procedures at clinic A on the same day as HCV-infected persons, and serologic HCV, HBV, and HIV testing of staff. An extensive review of the clinic practices and procedures also was conducted, including observation of several endoscopic procedures and endoscopic reprocessing, observation of anesthesia practices, and interviews with staff members regarding their infection-control practices. For this investigation, a person was defined as having health-care--associated acute hepatitis C if he or she 1) had symptoms of acute hepatitis within 6 months of having a procedure performed at clinic A during July--December 2007; 2) had laboratory-confirmed HCV infection (antibodies to HCV [anti-HCV]) by enzyme immunoassay (EIA) and recombinant immunoblot assay (RIBA) or EIA with an appropriate signal-to-cutoff ratio for a given assay, or presence of HCV RNA by polymerase chain reaction (PCR) in the absence of acute hepatitis A virus (HAV); and 3) did not have other risks for HCV infection. In addition to the three persons identified initially, three other persons were determined to have health-care--associated acute hepatitis C, for a total of six cases diagnosed during July--December 2007. One of the three cases was identified by review of surveillance records, another by cross-matching local laboratory records with procedure records at clinic A, and the third by physician report after the start of the investigation. The six persons ranged in age from 37 to 72 years; four were female. All had signs and symptoms of acute hepatitis, including jaundice, abdominal discomfort, and laboratory evidence of liver inflammation with alanine aminotransferase (ALT) levels of 552--1,165 units/L.* Four of the six persons required hospitalization as a result of their HCV infection. The six persons with acute hepatitis C had onset of symptoms in late October 2007 and November 2007, 35--90 days after undergoing procedures at clinic A (Figure 1) and within the typical incubation period of 15--160 days. None had significant risk factors for HCV infection and none had other common exposures. One of the procedures was performed in July 2007; the other five were performed on the same day in September 2007. Five persons (four with procedures on the same day) for whom blood specimens were available at the time of this report had HCV genotype 1a. The four who had procedures on the same day had viral sequences with 99%--100% genetic similarity at HVR1, pointing to a common source of infection. The viral sequence from the HCV-infected person who had the procedure in July 2007 was not genetically related to the other cluster, suggesting a separate transmission incident. During the 2 days in which persons with health-care--associated hepatitis C had procedures at clinic A, 120 additional persons had procedures at the clinic. HCV test results for those persons are pending. Thirty-eight staff members at the clinic involved in direct patient care were available for testing during the investigation, and none had evidence of previous or current HCV infection. None of the persons with health-care--associated acute hepatitis C and none of the staff tested positive for HBV or HIV infections. Inappropriate reuse of syringes on individual persons and use of medication vials intended for single-person use on multiple persons was identified through direct observation of infection-control practices at clinic A (Figure 2). Specifically, a clean needle and syringe were used to draw medication from a single-use vial of propofol, a short-acting intravenous anesthetic agent. The medication was injected directly through an intravenous catheter into the patient's arm. If a patient required more sedation, the needle was removed from the syringe and replaced with a new needle; the new needle with the old syringe was used to draw more medication. Backflow from the patient's intravenous catheter or from needle removal might have contaminated the syringe with HCV and subsequently contaminated the vial. Medication remaining in the vial was used to sedate the next patient. As soon as improper injection practices were observed, health officials advised clinic A to stop these practices and educated staff about the risks. Clinic A is a free-standing private endoscopy clinic in southern Nevada that primarily performed upper endoscopies and colonoscopies (approximately 50--60 procedures a day, 5 days a week). For at least the 4 years that clinic A occupied its existing location, the unsafe injection practices had been commonly used among some staff members who administered anesthesia, according to those who were interviewed. On February 27, 2008, SNHD began notifying approximately 40,000 persons who underwent procedures requiring anesthesia at the clinic from March 1, 2004, through January 11, 2008, via mail and through the media, to undergo screening for HCV, HBV, and HIV infections. Results of this screening are pending. Reported by: B Labus, MPH, L Sands, DO, P Rowley, Southern Nevada Health District, Las Vegas; IA Azzam, MD, Nevada State Dept of Health and Human Svcs. SD Holmberg, MD, Div of Viral Hepatitis, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention; JF Perz, DrPH, PR Patel, MD, Div of Healthcare Quality Promotion, National Center for Preparedness, Detection, and Control of Infectious Diseases; GE Fischer, MD, M Schaefer, MD, EIS officers, CDC. Editorial Note:Although case-control studies have not indicated an increased risk for acquiring HCV from medical, surgical, or dental procedures in the United States (2), outbreaks of HCV in health-care settings have long been recognized (3). These outbreaks have been identified primarily through clusters of temporally related cases detected by routine viral hepatitis surveillance, a method that likely underestimates the magnitude of transmission. Surveillance for viral hepatitis typically is passive, with little or no capacity to investigate cases suggestive of transmission during health care and determine their cause (4). Among persons with acute HCV infections, 60%--70% are asymptomatic (2). Additionally, currently available laboratory tests cannot distinguish acute from chronic HCV infection, which makes identifying newly acquired cases difficult. The investigation described in this report identified six cases of acute hepatitis C in persons who underwent procedures at clinic A 35--90 days before the onset of their illness. None of the persons had significant risk factors for HCV infection within the typical incubation period (15--160 days before onset of symptoms), and five of the cases had procedures on the same day (September 21, 2007). The genetic relatedness of the viruses from case patients who had procedures on September 21, 2007, supports the epidemiologic findings and points to a common source of infection. The lack of genetic relatedness to the patient seen in July 2007 suggests a separate transmission incident. The two distinct clusters suggest patient-to-patient transmission rather than staff-to-patient transmission. Most outbreaks of health-care--associated HCV have involved patient-to-patient transmission attributed to unsafe injection practices (3,5). The reuse of syringes and needles or mishandling of medication vials usually have been implicated (6--8). In some situations, syringes or needles used on HCV-infected persons were directly reused on other persons. In other instances, syringes or needles used on HCV-infected persons were reused to draw medication from a vial from which medicine was then drawn and administered to multiple persons, as was found in this investigation. When gross errors or high-risk infection-control breaches that could lead to bloodborne pathogen transmission are recognized, including unsafe injection practices, potentially exposed persons should be notified and tested, even if transmission has not been confirmed (9). Those persons who are found to be infected can then obtain proper medical care. In addition to approximately 40,000 notifications that occurred as a result of this outbreak, in unrelated incidents, unsafe injection practices at three other outpatient clinics in two states have resulted in approximately 28,000 patient notifications during the preceding year (CDC, unpublished data, 2008). These situations could have been avoided if standard infection-control precautions, which include basic safe injection practices, had been followed (Box) (10). This outbreak highlights the importance of surveillance and investigation in detecting viral hepatitis transmission in health-care settings. Prevention of transmission in these settings requires understanding and adherence to recommended infection-control practices. Medical and nursing school curricula and other health-care professional training, licensing, and continuing education requirements should include infection-control content, including the safe handling and administration of parenteral medications, as areas of competency. Although hospitals employ infection-control professionals and regularly evaluate infection-control practices, such oversight might be limited in outpatient settings that are not associated with hospitals. As use of these settings grows, appropriate methods will be needed to provide similar oversight for outpatient clinics. Better surveillance, education, and oversight are needed to detect and prevent bloodborne pathogen transmission in ambulatory and other health-care settings. References
* The normal ALT range varies according to age, sex, and other factors. An upper limit of 28--55 units/L is generally considered normal.
Figure 1 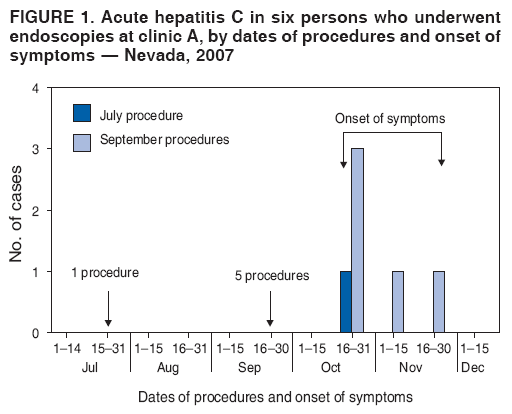 Return to top. Figure 2 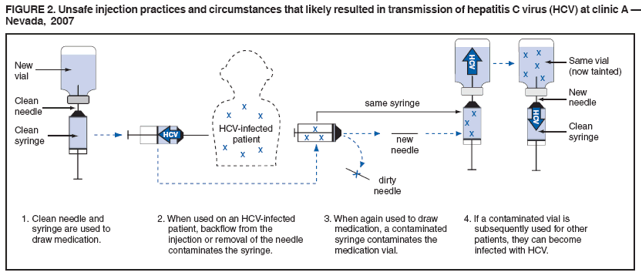 Return to top. Box 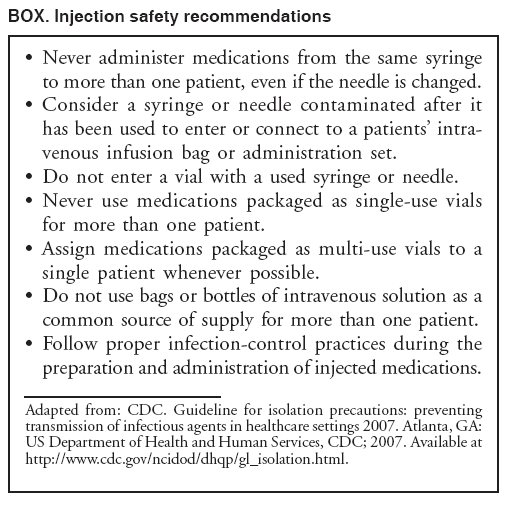 Return to top.
All MMWR HTML versions of articles are electronic conversions from typeset documents. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 5/15/2008 |
|||||||||
|