 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Suspension of Rotavirus Vaccine After Reports of Intussusception ---United States, 1999Please note: An erratum has been published for this article. To view the erratum, please click here. On July 16, 1999, CDC recommended that health-care providers suspend use of the licensed rhesus-human rotavirus reassortant-tetravalent vaccine (RRV-TV) (RotaShield®, Wyeth Laboratories, Inc., Marietta, Pennsylvania) in response to 15 cases of intussusception (i.e., a bowel obstruction in which one segment of bowel becomes enfolded within another segment) among infants who received RRV-TV (1). The Vaccine Adverse Event Reporting System (VAERS) monitored for adverse events following licensure of RRV-TV on August 31, 1999. After the recommendation to suspend use of the vaccine, no additional cases were reported (2). This report describes the surveillance activities used to identify this vaccine adverse event, the emergency response, and follow-up investigations. Suspension of RRV-TV after the initial cases of intussusception parallels the removal of the Broad Street pump handle in response to John Snow's epidemiologic studies; both were decisive, life-saving public health actions. VAERS, operated by CDC and the Food and Drug Administration (FDA), is a national passive surveillance system that monitors the safety of vaccines (3). Health-care providers, consumers, and vaccine manufacturers are encouraged to report adverse events involving all U.S.-licensed vaccines. During 1998--1999, CDC and FDA monitored VAERS for reports of intussusception and other severe gastrointestinal events among RRV-TV recipients. As a requirement for FDA licensure, the vaccine manufacturer funded a postlicensure phase IV trial of RRV-TV at Northern California Kaiser Permanente (NCKP) to monitor possible adverse events (4). Intussusception had been observed at low rates in prelicensure clinical trials, but a causal association with the vaccine was not proven; this information and a request for reporting to VAERS were included in the vaccine product label and recommendations of the Advisory Committee on Immunization Practices (ACIP). During December 18, 1998--June 2, 1999, a total of 10 cases of intussusception were reported to VAERS (Figure 1). Four cases of intussusception involving other vaccines had been reported to VAERS during the preceding 8 years (2,5). The cases appeared to be nonrandom in distribution; among the majority of infants, intussusception occurred after receiving the first dose of RRV-TV and within 1 week after receiving any dose. The temporal clustering after receipt of RRV-TV suggested a causal relationship. These early findings prompted CDC to request data from NCKP and initiate two validation studies, a 19-state case-control study and case-series analysis (6), and a cohort study in 10 managed care organizations (4). CDC alerted ACIP on June 17, 1999. By July 6, 1999, the number of cases reported to VAERS had increased to 15, a higher number than expected, accounting for likely underreporting, available baseline estimates of intussusception, and the estimated number of doses of RRV-TV distributed (1; Table). Preliminary data from both NCKP and from Minnesota, a state participating in one of the CDC studies, suggested a similar elevated risk for intussusception within the first week after RRV-TV vaccination (4). On July 16, 1999, in an MMWR report, CDC recommended temporarily suspending use of RRV-TV, pending the results of CDC studies that subsequently confirmed the initial observation that the greatest risk for intussusception was within 3--7 days after the first dose of RRV-TV (4,6). By December 31, 1999, a total of 112 cases of intussusception with illness onset before August 15, 1999, had been reported to VAERS (Figure 2) 1 month after the suspension of the rotavirus vaccination program (7). Ninety-five cases among infants were confirmed by review of medical records, and three were confirmed by primary-care providers (2). No cases of RRV-TV--associated intussusception occurred in infants and children vaccinated after July 16, 1999. When the VAERS findings were confirmed by more definitive studies (4,6), the manufacturer voluntarily recalled the vaccine, and the ACIP recommendations were withdrawn in October 1999 (2). Reported by: Food and Drug Administration. J Iskander, MD, P Haber, MPH, TV Murphy, RT Chen, MD, Epidemiology and Surveillance Div, National Immunization Program; M Sabin, PhD, EIS Officer, CDC. Editorial Note:The suspected association between RRV-TV and intussusception based on a review of VAERS data led CDC, in conjunction with state and local health departments, to implement a case-control study and case-series analysis (6) and a retrospective cohort study (4). These studies subsequently confirmed that intussusception was associated with RRV-TV in vaccine recipients. On the basis of evaluation of all available information, on February 21, 2002, ACIP reaffirmed its decision to withdraw the recommendation for use of RRV-TV in the United States (8). The case-control study was conducted in 19 U.S. states among 429 infants and 1,763 matched controls. Infants who received RRV-TV were 37 times more likely to have intussusception 3--7 days after the first dose than infants who did not receive RRV-TV (95% confidence interval [CI] = 12.6--110.1) (6). The population-based, retrospective cohort study among 463,277 children in managed care organizations demonstrated that of 56,253 infants vaccinated with RRV-TV, those who received the vaccine were 30 times more likely to have intussusception 3--7 days after the first dose than infants who did not receive the vaccine (95% CI = 8.8--104.9). The excess risk was estimated between one case in 5,000 vaccinees and one case in 11,000 vaccinees (4,6). In addition to intussusception (5), subsequent analysis of all VAERS reports documented a broader spectrum of gastrointestinal illnesses (e.g., bloody stool, vomiting, diarrhea, gastroenteritis, and abdominal pain) after receipt of RRV-TV. Passive surveillance systems such as VAERS are subject to multiple limitations, including underreporting, reporting of purely temporal associations or unconfirmed diagnoses, lack of denominator data, and unbiased comparison groups (2). In addition, determining causal associations between vaccines and adverse events from VAERS reports is not possible. Nevertheless, because VAERS is a national surveillance system with a simple reporting mechanism, it yields timely information and has high sensitivity for new vaccine safety concerns. Despite estimated underreporting of intussusception after RRV-TV of approximately 50% (7), VAERS successfully provided an alert. Recent reevaluations of the RRV-TV experience have assessed the strength of association, the attributable risk, and the possibility that RRV-TV triggered intussusception in those at risk (8). Follow-up of the managed care cohort showed equal risk for intussusception among vaccinated and unvaccinated infants >21 days postvaccination, arguing against the trigger hypothesis (9). Rotavirus infection is associated with increased distal ileum wall thickness and lymphadenopathy, suggesting a possible mechanism by which rotavirus infection or RRV-TV could cause intussusception (10). Rotavirus is the most common cause of severe gastroenteritis in the United States, resulting in approximately 500,000 physician visits, 50,000 hospitalizations, and 20--40 deaths annually; worldwide, rotavirus accounts for an estimated 600,000 deaths annually among children aged <5 years (3). In the United States, where high routine vaccination coverage has nearly eliminated childhood vaccine-preventable diseases, concerns regarding vaccine safety are increasing. Rotavirus vaccines under development will be measured for safety by lessons learned from the RRV-TV/intussusception experience (2,9). The causal association between RRV-TV and intussusception was established in the postlicensure period because a unique adverse event occurred shortly after vaccination, at a frequency (approximately one case per 10,000 doses) detectable by surveillance tools. This frequency contrasts with paralysis after oral poliovirus vaccine (no longer used in the United States) of one case in 750,000--1.2 million doses. Evaluation of other vaccine safety concerns, which include disorders involving delayed/insidious onset (e.g., autism) or other diseases (e.g., yellow fever vaccine--associated viscerotropic and neurotropic disease) in smaller populations, is more challenging. CDC and FDA will continue to develop new surveillance tools for timely detection of rare or unanticipated vaccine adverse events and to ensure confidence in vaccination. The investigation of RRV-TV and intussusception had 145 years of historical precedent in John Snow's epidemiologic methodology. The rapid elimination of risk based on systematic investigation, surveillance, and ongoing scientific confirmation averted any other cases of intussusception associated with RRV-TV. Acknowledgments The findings in this report are based in part on contributions by state and local health departments. L Zanardi Blevins, MD, Vermont Dept of Health. W Zhou, MD, M McCauley, MTSC, P Gargiullo, PhD, G Mootrey, DO, S Chu, PhD, National Immunization Program, CDC. References
Figure 1 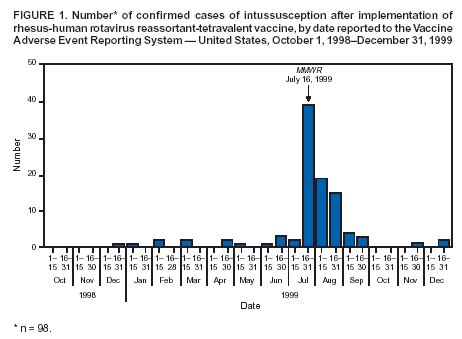 Return to top. Figure 2 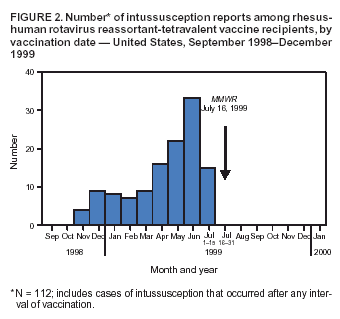 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 9/2/2004 |
|||||||||
This page last reviewed 9/2/2004
|