 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Murine Typhus --- Hawaii, 2002Murine typhus, a zoonotic disease caused by Rickettsia typhi, is uncommon in the United States. Hawaii typically reports five or six cases annually; however, 47 cases were reported in 2002. This report summarizes clinical data for three of these cases and describes murine typhus activity in Hawaii during 2002 and control efforts of the Hawaii Department of Health (HDH). The high number of reported cases in 2002 underscores the need for community education to prevent murine typhus and an assessment of environmental factors that might contribute to local disease transmission. Case ReportsCase 1. In June 2002, a previously healthy man aged 49 years from Maui was hospitalized after 10 days of fever, headache, fatigue, chills, nausea, vomiting, abdominal pain, and bloody stools. On examination, he had a petechial rash on his trunk and extremities, fever of 103.0° F (39.4° C), and mental status changes. The patient had a hematocrit of 25% (normal: 42%--52%), white blood cell count (WBC) of 22,000/mm3 (normal: 4,500--11,000/mm3), and platelet count of 38,000/mm3 (normal: 150,000--400,000/mm3). Serum creatinine was 12.4 mg/dL (normal: 0.7--1.5 mg/dL). Shortly after admission, he had a seizure and was intubated. Cerebrospinal fluid showed a WBC of 103/mm3 (normal: 0--5/mm3), red blood cell count of 29/mm3 (normal: 0/mm3), and protein of 140 mg/dL (normal: 15--50 mg/dL). The patient also had elevated serum lactic dehydrogenase, aspartate aminotransferase (AST), and alanine aminotransferase (ALT). Brain magnetic resonance imaging showed bitemporal and thalamic cerebritis. He was treated with intravenous (IV) ceftriaxone and IV doxycycline. The patient also underwent hemodialysis for renal failure. Serum obtained on day 2 of hospitalization was tested by indirect immunofluorescence assay (IFA) for antibodies reactive with typhus group rickettsiae; IgM and IgG were present at titers of 8,192 and 512, respectively. Polymerase chain reaction (PCR) testing of serum revealed DNA of R. typhi. After a gradual recovery, the patient had persistent frontal and temporal lobe dysfunction. The patient had no recent history of travel, camping, or hiking. A site investigation of his beach house in July revealed rat infestations in the yard and attic. Case 2. In July 2002, a previously healthy man aged 23 years from Molokai sought medical care at a local emergency department (ED) after 9 days of fever (104° F [40° C]), chills, frontal headache, and myalgia. On examination, his temperature was 102.9° F (39.4° C), and he had meningismus, photophobia, conjunctivitis, and hepatosplenomegaly. The patient had a hematocrit of 40%, platelet count of 113,000/mm3, and elevated serum AST and ALT. He was treated with IV doxycycline and showed immediate marked clinical improvement. Serum collected 23 days after illness onset was tested by IFA and contained IgM and IgG antibodies to typhus group rickettsiae at titers of 256. The patient reported no recent travel outside Hawaii. He cared for many animals at home, including dogs and domestic farm animals. He reported no direct contact with rodents but reported exposure to fleas. Case 3. In September 2002, a previously healthy boy aged 15 years from Molokai was taken to a local ED after 5 days of fever, headache, malaise, myalgia, neck pain, and abdominal pain. On examination, he had a fever of 103.0° F (39.4° C), diffuse tenderness of the lower extremities, and hepatomegaly. He had a platelet count of 82,000/mm3, elevated AST and ALT, a partial thromboplastin time of 44 seconds (normal: 25--38 seconds), and a fibrinogen level of 460 mg/dL (normal: 200--400 mg/dL). He was treated with IV ceftriaxone, but his symptoms persisted. A chest radiograph revealed mild pulmonary edema with small bilateral pleural effusions and hepatosplenomegaly. IV clindamycin and IV penicillin were added to the treatment regimen for a presumptive diagnosis of leptospirosis. Serum samples were tested by IFA for antibodies to typhus group rickettsiae and showed approximately a fourfold change in titer, from nonreactive on day 1 of hospitalization to IgM and IgG titers of 256 and 512, respectively, on day 4. The patient was discharged after 10 days and was later treated with doxycycline. The patient lived in the same town as patient 2; he reported no recent travel. He reported a noticeable increase in the number of rodents around his home and admitted to handling them, but did not recall any flea exposure. Murine Typhus SurveillanceDuring 2002, a total of 47 cases of murine typhus were identified in Hawaii, the largest number recorded annually since 1947. Of these cases, 25 (53%) were confirmed*, and 22 (47%) were probable†; 34 (72%) cases occurred during July--October (Figure). The median age of the patients was 38 years (range: 1--68 years); 28 (60%) were male. Five islands reported cases of murine typhus: Maui (35 cases), Molokai (six), Oahu (three), Kauai (two), and Hawaii (one). The most common symptoms reported among the 47 patients were fever (98%), malaise (89%), headache (87%), myalgia (81%), loss of appetite (81%), chills (81%), arthralgia (72%), nausea (66%), vomiting (54%), backache (53%), abdominal pain (51%), stiff neck (47%), and skin rash (45%). Moderate-to-severe disease was observed, including acute renal failure (two cases), gastrointestinal bleeding (two), meningitis (two), encephalitis (one), pneumonitis (one), and congestive heart failure with pleural effusion (one). Control MeasuresHDH applied rodenticide (i.e., zinc phosphate oat bait) to grasslands and pasture areas believed to be major sources of peridomestic rodents. Dramatic decreases in the numbers of trapped rodents on Maui and other foci followed. HDH conducted rodent trapping, environmental assessments, and rodent-proofing education at the homes of patients with suspected and confirmed cases. Public awareness campaigns/programs and education about murine typhus were conducted through physician notices, press conferences, and television and newspaper interviews. Clinical manifestations, mode of transmission, and peridomestic rodent- and flea-control measures were emphasized. Reported by: S Hoskinson, MD, M Lipetz, MD, Maui Memorial Hospital, Wailuku; C Sherer, MD, S Fraser, MD, E Taniguchi, Tripler Army Hospital, Honolulu; A Tice, MD, D Behling, B Tanabe, MD, D Kwock, MD, Univ of Hawaii School of Medicine, Honolulu; P Effler, MD, L Pang, MD, E Brown, L Granville, K Mills, D Sasaki, DVM, M Ching Lee, H Matsubayashi, W Warashina, A Ueno, C Takekuma, J Haruno, S Oshiro, Hawaii Dept of Health. P Kitsutani, MD, Div of Applied Public Health Training, Epidemiology Program Office; J McQuiston, DVM, C Paddock, MD, J Sumner, J Krebs, Div of Viral and Rickettsial Diseases, National Center for Infectious Diseases, CDC. Editorial Note:R. typhi, the etiologic agent of murine typhus (Box), is maintained in nature by a rodent-flea cycle, with rats (most commonly Rattus spp.) as the primary reservoirs. Humans acquire infection after exposure of abraded or flea-bitten skin to infectious flea feces (1). Although murine typhus usually is a self-limiting febrile illness, life-threatening disease and death can occur (2). Before World War II, murine typhus was widespread, and approximately 42,000 cases were reported in the United States during 1931--1946 (1,3). Improved rodent- and ectoparasite-control practices have decreased transmission substantially in the United States, and <50 cases are reported annually, most often in California, Texas, and Hawaii (4). Murine typhus is considered endemic on the Hawaiian islands of Kauai, Maui, and Oahu (5). This report also suggests a possible endemic focus on Molokai. The animal reservoir in Hawaii is unknown, but the disease is suspected to involve a rodent-flea cycle. Rodents on Hawaii that have demonstrated antibodies reactive with typhus group rickettsiae include the Polynesian rat (Ra. exulans), the black rat (Ra. rattus), the Norway rat (Ra. norvegicus), and the house mouse (Mus musculus) (6; HDH, unpublished data, 2002--2003). The Indian mongoose (Herpestes auropunctatus) also is a possible reservoir (7). Infection with R. typhi is diagnosed typically by using IFA with paired acute- and convalescent-phase serum; however, antibody often is not detected during early stages of the illness. Because delay in appropriate antimicrobial therapy can result in severe disease, physicians should treat suspected cases of murine typhus on the basis of clinical and epidemiologic findings. Doxycycline is the treatment of choice. The findings in this report are subject to at least two limitations. First, because murine typhus can have a wide range of nonspecific clinical signs and might resemble other diseases (e.g., dengue or leptospirosis), surveillance for this disease can be difficult. Second, although HDH instituted active surveillance for murine typhus during 2002 through physician alerts and mandatory laboratory reporting of R. typhi test requests, additional cases probably occurred but were missed. The most effective method of controlling outbreaks of murine typhus is to reduce the rodent reservoir responsible for maintenance of the pathogen and the ectoparasites responsible for transmission to humans. If rodent depopulation programs are instituted, simultaneous application of appropriate insecticides should be considered to prevent infected arthropods from transmitting disease to humans (8). Assessment of rodent numbers and serologic testing of rodents in areas where human murine typhus cases have occurred will help guide prevention efforts. References
* Clinically compatible illness with a fourfold change (or equivalent) in serum antibody titer reactive with typhus group rickettsiae between paired serum specimens, demonstration of typhus group rickettsiae by immunohistochemical methods, demonstration of R. typhi by nucleic acid detection, or isolation of R. typhi by cell culture. † Clinically compatible illness with serologic evidence of antibody reactive with typhus group rickettsiae in a single serum sample at a titer considered indicative of current or previous infection (cutoff titers are determined by individual laboratories). Figure 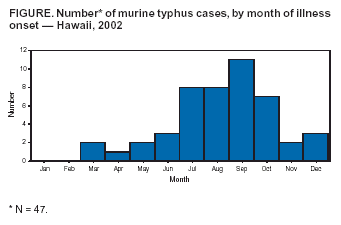 Return to top. Box  Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 12/18/2003 |
|||||||||
This page last reviewed 12/18/2003
|