 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Tickborne Relapsing Fever Outbreak After a Family Gathering --- New Mexico, August 2002On August 2, 2002, the New Mexico Department of Health (NMDOH) and the Indian Health Service (IHS) were notified of a tickborne relapsing fever (TBRF) outbreak after a 1-day family gathering held in late July at a remote, previously uninhabited cabin located in a mountainous region of northern New Mexico (elevation: approximately 8,000 feet). Approximately 40 persons attended the event; at least half slept overnight in the cabin. This report summarizes the investigation of this outbreak, which indicates that prompt diagnosis and collaboration among clinicians and public health authorities can reduce morbidity associated with TBRF. Persons living in areas where TBRF is endemic should avoid sleeping in rodent-infested buildings, rodent-proof susceptible buildings, and consider fumigation of buildings that harbor rodents. The index patient arrived with other family members 3 days before the event to clean the cabin. Four days after the event ended, the patient sought medical care at a local hospital for a 2-day history of fever, chills, myalgia, and a raised pruritic rash on the forearms. A laboratory technician identified spirochetes on a peripheral blood smear obtained from the patient, which led to a diagnosis of TBRF and prompted an epidemiologic investigation. During July 27--August 7, a total of 39 attendees sought medical care or were visited by a public health nurse. A retrospective cohort study of these attendees was conducted to describe the outbreak, determine risk factors for infection, and assist the cabin owners with prevention measures. A case of TBRF was defined as laboratory-confirmed borreliosis (growth of Borrelia hermsii in blood culture or visualization of spirochetes on Giemsa- or Wright-stained peripheral blood smear) in a person who attended the gathering and had a fever. A total of 14 (36%) attendees had reported fever and at least one of the following: chills, diaphoresis, headache, myalgia, arthralgia, rash, or a tick bite. Peripheral blood smear examinations were performed on samples taken from all 14 symptomatic attendees; spirochetes were observed on samples from nine attendees. Blood samples from 13 of the 14 symptomatic attendees were sent to CDC for culture. Two samples that had not demonstrated spirochetes on peripheral smear examination grew B. hermsii. A total of 11 patients had laboratory findings consistent with the case definition, yielding an attack rate of 28% among attendees. The median age of the 11 patients was 51 years (range: 4--80 years); eight (73%) were female. The median incubation period was 5 days (range: 3--7 days). Six (55%) patients had a documented fever of >100.4º F (>38.0º C), eight (73%) had headache, seven (64%) had body aches (arthralgias and/or myalgias), and four (36%) reported some kind of rash. All 14 symptomatic attendees received antibiotic therapy, and eight asymptomatic attendees received antibiotic prophylaxis. A total of 18 attendees received doxycycline alone, two had treatment with doxycycline and penicillin, and two received erythromycin. Nine patients were admitted to the hospital for treatment; six had a documented Jarisch-Herxheimer reaction (i.e., fever, chills, myalgias, increased heart and respiratory rate, and hypotension). Risk analysis revealed that persons who arrived early to clean the cabin were more likely to be patients than persons who did not arrive early (three of three versus eight of 36; risk ratio = 4.5; 95% confidence interval = 2.4--8.3). Immediately after diagnosis of TBRF in the index patient, local clinicians and community public health nurses identified and treated symptomatic attendees. IHS environmental health-care workers and NMDOH staff visited the event site to inspect the cabin and its surroundings. The inspection revealed an abundance of rodent nesting material and droppings within the walls of the cabin (Figure 1). Gaps were observed in the exterior chinking and the foundation of the cabin that allowed rodents to have easy entry. CDC led a second site visit to collect environmental samples and trap rodents; however, in the interim the cabin had been fumigated twice with a commercial over-the-counter fogger containing pyrethrin and permethrin. One live soft tick (Ornithodoros hermsi), four chipmunks, one wood rat, and two deer mice were recovered. The tick was allowed to feed on a laboratory mouse, that subsequently had B. hermsii spirochetemia, confirming that the tick was infected and supporting the mode of transmission. Blood samples from the trapped animals were negative for spirochetes. The cabin owner was provided written material to assist with rodent-proofing the premises. Reported by: PJ Ettestad, DVM, RE Voorhees, MD, CM Sewell, DrPH, New Mexico Dept of Health. M Bonnell, MD, J Iralu, MD, J Cheek, MD, KJ Secord, MPH, D Mosier, MPH, Indian Health Svc. RE Enscore, MS, ME Schriefer, PhD, S Marshall, MPH, Div of Vector-Borne Infectious Diseases, National Center for Infectious Diseases; RJ Groves, MD, CB Smelser, MD, EIS officers, CDC. Editorial Note:TBRF is caused by infection with any of several species of the genus Borrelia (Box). In the United States, the majority of cases occur in western states and are caused by B. hermsii or B. turicatae, which are transmitted to humans by argasid (soft) ticks of the genus Ornithodoros. O. hermsi is typically found associated with active rodent nests at altitudes of >1,500 feet in warm, humid microenvironments. Although the primary tick hosts are chipmunks and other small rodents, ticks might bite persons, typically at night during sleep. Caves and rural mountain cabins accessible by rodents are often sites of human exposure to these ticks and have been associated with previous outbreaks (1--3). Ornithodoros ticks feed for <1 hour; their bites are painless and typically unnoticed by humans. After a median incubation period of 7 days (range: 2--18 days), TBRF is characterized clinically by recurring fevers with a median duration of 3 days (range: 2--7 days) and alternating afebrile periods with a median duration of 7 days (range: 4--14 days). Fevers might be accompanied by headache, rigors, diaphoresis, arthralgia, myalgia, dizziness, nausea, or vomiting. Without treatment, up to 10 relapsing episodes might occur (4,5). Relapsing episodes are caused by antigenic variation that enables the organism to evade neutralizing antibodies (1,6). No relapsing episodes occurred in this outbreak, likely a result of rapid identification and treatment of the index patient and symptomatic attendees. Diagnosis of TBRF is confirmed by blood culture or visualization of spirochetes on examination of a peripheral blood smear. Paired acute and convalescent antibody titers can be used to confirm diagnosis when culture or blood smear is not available or nondiagnostic. Although not a nationally notifiable disease, TBRF reporting to state and local health departments is required in Arizona, California, Colorado, Idaho, Montana, Nevada, New Mexico, Texas, Utah, Washington, and Wyoming. During 1990--2000, a total of 247 cases were reported from these states, with the majority from California (82) and Washington (60) (Figure 2). The majority (90%) of cases were reported from 11 counties in six states and occurred in the late summer and early fall. In 2000, a neonatal death in Washington was the first reported neonatal TBRF death in the United States since 1969 (CDC, unpublished data, 2000). Health-care providers should consider TBRF in the differential diagnosis of febrile patients with potential exposure to soft ticks. Penicillins or tetracyclines are the antibiotic treatment of choice, although cephalosporins, erythromycin, or chloramphenicol also can be used (4,5). The Jarisch-Herxheimer reaction is common after the initiation of therapy (onset: 1--4 hours), with one case series reporting an incidence of 54% (1). Without treatment, case-fatality rates can approach 10%. Measures to prevent TBRF include avoiding sleeping in rodent-infested buildings, using insect repellents containing DEET, and rodent-proofing buildings in areas where disease is endemic (7). Fumigation with preparations containing pyrethrin and permethrin might reduce the tick burden in rodent-infested buildings; however, results from this investigation indicate that fumigation might not kill all infected ticks. Remediation also should include efforts to identify and remove rodent nesting material. Acknowledgments The report is based on data contributed by J Butler, Public Health Nurses, and Environmental Health Officers, Indian Health Svc. References
Figure 1  Return to top. Figure 2 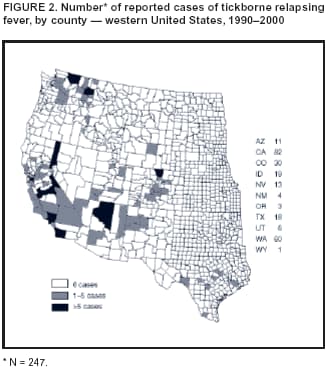 Return to top. Box  Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 8/28/2003 |
|||||||||
This page last reviewed 8/28/2003
|