 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Outbreak of Pneumococcal Pneumonia Among Unvaccinated Residents of a Nursing Home --- New Jersey, April 2001On April 24, 2001, seven cases of pneumococcal pneumonia with bacteremia among residents of a nursing home were reported to the Hamilton Township Department of Health, New Jersey; all seven diagnoses were confirmed with blood cultures positive for Streptococcus pneumoniae. Illness onset among the residents occurred during April 3--24; four residents died. The New Jersey Department of Health and Senior Services (NJDHSS) was notified on April 24 and initiated an investigation to identify additional cases and implement control efforts. This report summarizes results of the investigation, which underscore the importance of providing pneumococcal polysaccharide vaccine (PPV) to elderly residents of long-term care facilities (LTCFs). The nursing home is a 114-bed facility that employs approximately 200 staff, including nurses, restorative aides, and other administrative and support personnel. None of the employees was known to have pneumonia or laboratory-confirmed pneumococcal disease during this period. On further investigation, two additional residents were identified to have been hospitalized during April 3--24 with pneumonia (Figure 1). Seven of the nine patients had blood cultures positive for S. pneumoniae, which were sent to the New Jersey Public Health and Environmental Laboratory and CDC for serotyping and susceptibility testing; pulsed-field gel electrophoresis was performed. All isolates were serotype 14, belonged to the England 14-9 clonal group, and were penicillin-sensitive and resistant only to erythromycin. Sputum specimens from the two remaining residents were Gram stain positive for diplococci and findings from chest radiographs were consistent with pneumonia. Seven of the residents lived in the same wing of the nursing home. A case-control study was conducted to determine risk factors for pneumococcal pneumonia among residents of the nursing home. Cases included the nine residents hospitalized with pneumonia. Two controls per case-patient were selected randomly from among nursing home residents without pneumonia symptoms who resided in the wing where most of the case-patients resided during March 1--April 26. Nursing home medical records of case-patients and controls were reviewed, and a standardized form was used to abstract data. Case-patients had a median age of 86 years (range: 78--100 years); seven (78%) were women. Controls had a median age of 85 years (58--95 years), and 17 (94%) were women. Illness was strongly associated with lack of documentation of receipt of PPV (none of nine case-patients versus nine of 18 controls; odds ratio=0; 95% confidence interval=0--0.7). Other exposures assessed but not associated with disease included recent antibiotic therapy, history of pneumonia, hospitalizations during the preceding year, medical conditions that are risk factors for pneumococcal disease*, and physical functioning (e.g., mobility and ability to eat and swallow). At the time of the initial case of pneumococcal illness on April 3, 2001, 53 (49%) of the 108 residents had received PPV. When the outbreak was recognized, PPV was offered to all 55 nonvaccinated residents; 37 (67%) received vaccine. The remaining 18 were either ineligible for PPV or refused the vaccine. The nursing home also restricted transfers or admissions of patients with no history of having received PPV. Following the investigation of the nursing home, the NJDHSS Division of Long Term Care Systems surveyed 361 LTCFs during May 21--July 31 about their vaccination policies. Of these, 28 (8%) did not meet the state regulation that requires offering PPV to every resident of a LTCF. During May 24--June 7, the NJDHSS Division of Inspections, Compliance and Complaints investigated hospital compliance with the state regulation that requires offering PPV to every hospitalized patient aged >65 years. Hospitals were selected if they had admitted residents of the nursing home before their long-term care placement or had admitted residents of this nursing home during the preceding year. The selected hospitals' infection control practitioners were interviewed to identify hospital policy on offering the vaccine to PPV-eligible patients. NJDHSS staff reviewed medical records of seven case-residents and randomly selected medical records of patients aged >65 years to determine whether PPV was offered and administered. Four hospitals were identified, and medical records of 52 patients were reviewed; at the time of the review, 49 of these 52 patients were discharged, and three remained hospitalized. Each hospital had a form to facilitate physician identification and documentation of PPV-eligible patients; 35 (67%) of 52 medical records contained completed screening or assessment forms. Of the 52 patients, 13 (25%) had received PPV before hospital admission and 34 (65%) had no history of having received PPV and no contraindications to the vaccine; none of these patients had documentation of receipt of PPV while hospitalized. Reported by: E Bresnitz, MD, State Epidemiologist, C Grant, S Ostrawski, C Morris, J Calabria, B Reetz, New Jersey Dept of Health and Senior Svcs; S Clugston, Hamilton Township Dept of Health, Hamilton, New Jersey. Respiratory Diseases Br, Div of Bacterial and Mycotic Diseases, National Center for Infectious Diseases; Epidemiology Program Office; and an EIS Officer, CDC. Editorial Note:The findings in this report suggest that lack of pneumococcal vaccine may contribute to invasive pneumococcal disease in adults in LTCFs. Adults in LTCFs are especially vulnerable to pneumococcal disease and death because of their advanced age, the frequent presence of chronic illnesses, and residence in a setting that might increase the risk for bacterial transmission. PPV is considered safe and is cost-effective and potentially cost-saving among persons aged >65 years for prevention of bacteremia (1). Pneumococcal vaccinations are covered by Medicare, and virtually all state Medicaid plans cover vaccinations for high-risk groups (e.g., residents of nursing facilities). However, PPV coverage among elderly adults in LTCFs remains low, and outbreaks of pneumococcal pneumonia still occur in LTCFs with low vaccine coverage (2). In 1999, PPV coverage among a sample of nursing home residents in the United States was 38% (3). The low rate of PPV vaccination among institutionalized elderly has been attributed, in part, to a lack of physician emphasis on PPV administration (2). In addition, incomplete documentation of vaccination history of nursing home residents and misconceptions about adverse reactions after unintended revaccination with PPV may discourage health-care providers from vaccinating those with unknown vaccination history; however, the incidence of serious adverse events following revaccination is low (4). A study of Medicare inpatients indicated that opportunities to provide pneumococcal vaccines were missed for up to 80% of eligible elderly persons hospitalized with pneumonia (5). One of the national health objectives for 2010 is to achieve 90% pneumococcal vaccination coverage among nursing home residents and adults aged >65 years (objective 14-29) (6). Several methods have been developed for improving vaccine delivery. Standing orders programs, which authorize certain licensed health-care providers to administer vaccinations according to institutional and physician-approved protocols, improve vaccination rates in adults (7). Regulations that mandate hospitals, adult day-care facilities, and LTCFs, including nursing homes and assisted living facilities, to offer and document pneumococcal vaccinations may improve vaccination coverage for LTCF residents. However, this investigation highlights the limitations of regulations for ensuring vaccine coverage. Better documentation would facilitate tracking of the vaccination status of residents and provide medical history information to other health-care facilities when a resident is hospitalized or transferred or if an outbreak of pneumococcal illness occurred. For this report, only 361 of 853 LTCFs in New Jersey had been evaluated to date for compliance with the state's vaccination requirements. These facilities may not be representative of all LTCFs in New Jersey. This outbreak underscores the importance of providing pneumococcal vaccines to LTCF residents. The outbreak occurred in a setting of low vaccination coverage despite state regulations designed to improve vaccine delivery. A multifaceted approach that both facilitates delivery through standing orders programs and increases awareness of the importance of preventing pneumococcal disease may be needed to ensure optimal vaccine delivery to LTCF residents. References†
*Chronic cardiovascular disease, chronic obstructive pulmonary disease, chronic liver disease, diabetes mellitus, and renal dysfunction. † All MMWR references are available on the Internet at <http://www.cdc.gov/mmwr>. Use the search function to find specific articles. Figure 1 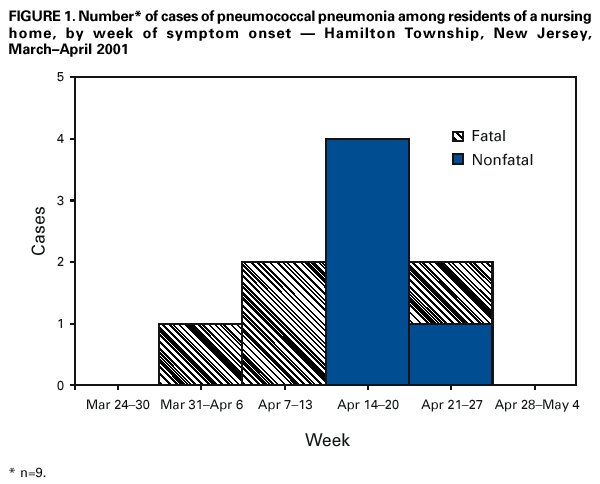 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 8/24/2001 |
|||||||||
This page last reviewed 8/24/2001
|