 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Congenital Syphilis --- United States, 2000In 1998, CDC initiated intensive efforts to eliminate syphilis from the United States. The following year, the National Syphilis Elimination Plan was launched with the goal of reducing primary and secondary (P&S) syphilis in adults to <0.4 cases per 100,000 population. A decrease in syphilis among women of reproductive age usually is followed by reductions in congenital syphilis (CS) rates. CS occurs when the spirochete Treponema pallidum is transmitted from a pregnant woman with syphilis to her fetus. Untreated syphilis during pregnancy may lead to stillbirth, neonatal death, and infant disorders such as deafness, neurologic impairment, and bone deformities. One of the national health objectives for 2000 was to reduce the rate of CS to <40 cases per 100,000 live-born infants (1). To evaluate progress toward this goal, the CS rate for 2000 was compared with the rate for 1997, the year before syphilis elimination efforts began. This report summarizes 1997--2000 CS surveillance data, which indicate that CS rates have decreased substantially among most racial/ethnic minority populations and that the elimination of CS in the United States is feasible because of the limited number of cases and highly focal distribution. To increase the percentage of women at risk who receive screening for syphilis during pregnancy, collaborative efforts are needed among health-care providers, health insurers, policymakers, and the public. CS surveillance data were reported to CDC from the 50 states and the District of Columbia. A case of CS was defined in a live-born infant who 1) manifested typical signs of CS or in whom T. pallidum was identified from external lesions, placenta, umbilical cord, or autopsy specimens, or whose mother had a syphilitic lesion at delivery; 2) was born to a woman with untreated or inadequately treated syphilis before or during pregnancy; or 3) was born to a woman with syphilis whose serologic response to penicillin therapy was not documented or was documented to be inadequate (i.e., less than a fourfold decline in titer) and had either a radiologic or cerebrospinal fluid (CSF) test consistent with CS or did not undergo a radiologic or CSF examination for signs of syphilis*. Also included are stillbirths among women with untreated syphilis. Reported CS cases include congenitally exposed infants who lack clinical signs of syphilis. Rates of CS per 100,000 live-born infants were determined from U.S. natality data†. In 2000, 529 CS cases were reported for a CS rate of 13.4 per 100,000 live-born infants compared with rates of 14.5 in 1999 and 27.8 in 1997, a 7.6% and 51.8% decrease from 1999 and 1997, respectively. In 2000, CS cases were reported from 155 (5.0%) of the 3115 U.S. counties. The rate was highest in the South (18.8) compared with the Midwest (9.1), the Northeast (10.1), and the West (11.8).§ All states reported rates <40 per 100,000 live-born infants, except Arkansas and South Carolina. In 2000, racial/ethnic minority populations had the highest CS rates (Figure 1): 49.3 among blacks, 22.6 among Hispanics, 13.2 among American Indians/Alaska Natives, and 5.9 among Asians/Pacific Islanders, compared with 1.5 among non-Hispanic whites. Compared with 1997, these rates represent a decline of 59.7% among blacks, 32.5% among Hispanics, 29.8% among Asians/Pacific Islanders, and 58.3% among non-Hispanic whites. Among American Indians/Alaska Natives, the rate increased by 20%; this represented a change from four cases reported in 1997 to five cases in 2000. In 2000, 83.2% of mothers of infants with CS were aged <35 years, compared with 84.3% in 1997. In 2000, the maternal age group with the highest rate (16.0 per 100,000 live-born infants) of infants with CS was adolescent mothers who delivered at age <19 years. This was a decrease of 45.5% from 1997 when the rate was 29.4. Among the 529 cases reported in 2000, 434 (82.0%) occurred because the mother had no documented treatment or had received inadequate treatment of syphilis before or during pregnancy. In 123 (28.3%) of these cases, the mother received no prenatal care; in an additional 35 (8.1%), no information on prenatal care was reported. In 36 (6.8%) cases, the mother was treated adequately but did not have an adequate serologic response to therapy, and the infant was evaluated inadequately for CS. In 30 (5.7%) cases, the mother did not have an adequate serologic response to therapy, and the infant's evaluation revealed laboratory or clinical signs of CS; 29 (5.5%) cases occurred for other reasons (Figure 2). Reported by: State and local health depts. Div of Sexually Transmitted Disease Prevention, National Center for HIV, STD, and TB Prevention; and an EIS Officer, CDC. Editorial Note:Substantial progress has been made in eliminating syphilis in the United States. In 2000, the number of CS cases was the lowest since the revised case definition was implemented in 1988, and all but two states met the national health objective for 2000 (1). Rates in 2000 declined 51.8% since 1997, the year preceding the start of syphilis elimination efforts. Interventions designed to prevent, detect, and treat syphilis in women of reproductive age may have had a substantial role in these declines. Many of these efforts targeted the racial/ethnic minority populations with the highest CS rates and were located mostly in the South. CS elimination is feasible because of the limited numbers of cases and their highly focal distribution; however, the cornerstone of CS elimination is early detection of syphilis and treatment with penicillin, which is inexpensive, widely available, effective, and safe for the mother and fetus (2). Lack of prenatal care, late or limited prenatal care, and maternal use of illicit drugs are associated with CS (3--5). Racial/ethnic minority populations, particularly those in southern states, are disproportionately affected by CS; syphilis rates are higher among these populations than among non-Hispanic whites, and the use of and access to early and comprehensive prenatal care by minority women may be limited. Limited use of and access to prenatal care appear to be the reasons that rates of CS are high among infants born to women aged <19 years; rates of syphilis are rarely high among these women. Lack of health-care provider adherence to CS screening recommendations also may result in CS. In a 1998 national survey, only 85% of obstetrician/gynecologists reported routinely screening pregnant women for syphilis (6). Many providers screen for syphilis only once during pregnancy, usually during the initial clinical visit, despite national recommendations for more frequent testing among women at high risk (e.g., uninsured women, women living in poverty, commercial sex workers, and illicit drug users). Recent trends in U.S. health-care delivery may present substantial barriers to early detection and treatment of syphilis in pregnant women, including the growing number of uninsured women, the limited expansion of prenatal care provided by Medicaid managed care and child health insurance programs, and decreased funding of publicly supported clinics, emergency departments, and other providers that serve poor, uninsured, racial/ethnic minority women and adolescents (7). The findings in this report are subject to at least one limitation. Although the analysis was limited by inconsistent application of the CS case definition and incomplete reporting of asymptomatic CS cases (8), these factors were unlikely to have accounted for the declines because no evidence has suggested that application of the case definition for CS or reporting practices changed during this period. CDC recommends syphilis testing for all women during the early stages of pregnancy. In areas where syphilis prevalence is high or among women at high risk, testing should be done twice in the third trimester, including once at delivery. All women who deliver a stillborn infant after 20 weeks' gestation should be tested. In populations in which use of prenatal care is not optimal, CDC recommends rapid plasma reagin (RPR) card-test screening and treatment (if the RPR-card test is reactive) at the time pregnancy is determined (9). Syphilis screening also should be offered in emergency departments, jails, prisons, and other settings that provide episodic care to pregnant women at high risk for syphilis (10). Access to and use of comprehensive prenatal care for women and adolescents who are uninsured or covered by public insurance programs (e.g., Medicaid, migrant health clinics, and the Indian Health Service) should be promoted by communities, health-care providers, and government organizations, and public awareness should be increased about the persistent risk for CS. Care for women with syphilis who use prenatal health services could be improved by increasing providers' adherence to screening and treatment guidelines with reminders and feedback about their prenatal syphilis screening and treatment practices. Ongoing efforts to form and maintain coalitions to develop, implement, and evaluate syphilis elimination activities and interventions also may assist in reducing the prevalence of syphilis among women of reproductive age and, in turn, eliminating CS. References
*Congenital Syphilis Case Investigation and Report Form 73.126. † From the National Center for Health Statistics, Vital Statistics: natality tapes 1989--1998. § Northeast=Connecticut, Maine, Massachusetts, New Hampshire, New Jersey, New York, Pennsylvania, Rhode Island, and Vermont; Midwest=Illinois, Indiana, Iowa, Kansas, Michigan, Minnesota, Missouri, Nebraska, North Dakota, Ohio, South Dakota, and Wisconsin; South=Alabama, Arkansas, Delaware, District of Columbia, Florida, Georgia, Kentucky, Louisiana, Maryland, Mississippi, North Carolina, Oklahoma, South Carolina, Tennessee, Texas, Virginia, and West Virginia; and West=Alaska, Arizona, California, Colorado, Hawaii, Idaho, Montana, Nevada, New Mexico, Oregon, Utah, Washington, and Wyoming. Figure 1 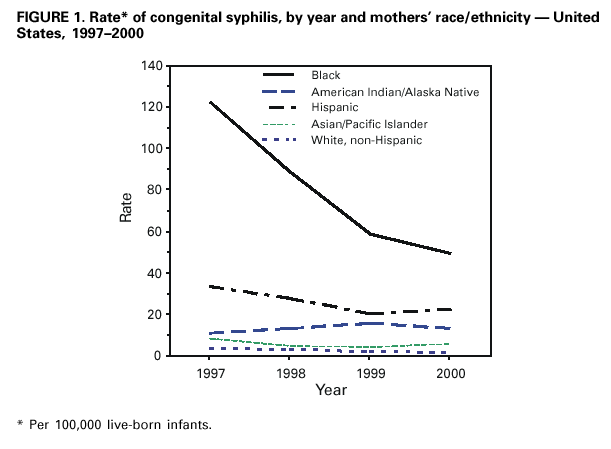 Return to top. Figure 2 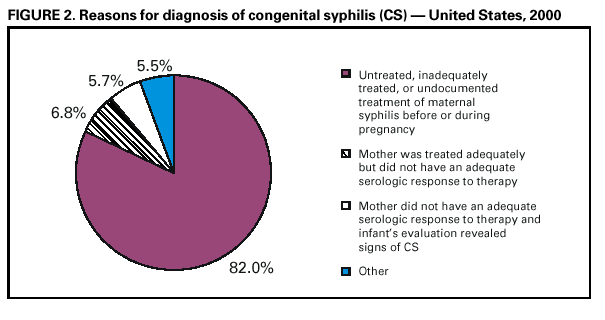 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 7/13/2001 |
|||||||||
This page last reviewed 7/13/2001
|