 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Progress Toward Poliomyelitis Eradication -- Nepal, 1996-1999In 1988, the World Health Assembly resolved to eradicate poliomyelitis globally by 2000 (1). In 1996, following the lead established by other countries of the South-East Asia Region (SEAR)*, Nepal accelerated polio eradication strategies by initiating National Immunization Days (NIDs)**. This report summarizes Nepal's progress toward polio eradication, focusing on the implementation of supplemental vaccination activities, the role of designated surveillance officers in the establishment of surveillance for polio eradication, and Nepal's plans for intensified supplemental vaccination to meet the 2000 eradication target (2). Routine and Supplemental Vaccination Programs Nepal's national routine vaccination coverage with three doses of oral poliovirus vaccine (OPV3) was reported to be 83% in 1996, 81% in 1997, and 83% in 1998 (3). However, estimates from an independent cluster survey in 1998 indicated that national OPV3 coverage was 70% (4). Of Nepal's 75 districts, 60 were included in the survey; of these, the 30 districts in the densely populated Terai plains along Nepal's southern border with India had lower OPV3 coverage (60%) than the 30 surveyed districts in the northern hill/mountain belt (79%) (4). Since 1996, NIDs have been conducted in Nepal on one day each in December and January during the low season for poliovirus transmission. NIDs during 1996-1997, 1997-1998, and 1998-1999 targeted children aged less than 5 years, and reached 97%, 96%, and 95% of the target population (3.9 million), respectively. Nepal's NIDs have been synchronized with NIDs in other countries of south and east Asia, including Bangladesh, Bhutan, China, India, Myanmar, Pakistan, and Thailand (5-8). Acute Flaccid Paralysis (AFP) Surveillance AFP surveillance in Nepal was initiated in 1995 with passive reporting of AFP cases through the Early Warning Reporting System, a sentinel system for surveillance of six target diseases***. An expanded nationwide AFP surveillance system was established in July 1998 with the training and deployment of six designated Nepali regional surveillance officers (RSOs). These officers conduct active surveillance for AFP cases in government and private health-care facilities and provide training, technical assistance, and logistic support for polio eradication activities in their regions. Weekly and monthly reporting sites have been recruited since July 1998, and the reporting network continues to expand through inclusion of more peripheral health facilities. AFP surveillance is evaluated by two key indicators: the sensitivity of reporting (target: one nonpolio AFP case per 100,000 population aged less than 15 years) and the completeness of stool specimen collection (target: two stool samples collected within 14 days of paralysis onset). The annualized nonpolio AFP rate increased from 0.2 in 1996 to 1.6 among children aged less than 15 years in 1999 (Table 1). The isolation rate of nonpolio enteroviruses from stool specimens, a measure of specimen condition and laboratory performance, was 33% in 1998 and 28% as of September 15, 1999. Confirmed Polio Cases Nepal uses the World Health Organization (WHO) clinical system for classification**** of polio cases. During 1998, of 69 reported AFP cases, 31 (45%) were confirmed as polio and 38 (55%) as nonpolio AFP (Figure 1). None of the 31 polio cases had collection of adequate stool specimens, and the classification of polio was made on clinical grounds (22 with residual weakness, four lost to follow-up, and five case-patients died before follow-up at 60 days). During 1999, of 164 reported AFP cases, 18 (11%) were classified as polio, 109 (66%) as nonpolio AFP, and 37 (23%) are pending classification (Table 1). The proportion of adequate stool specimens collected from AFP cases improved from 35% in 1998 to 79% in 1999, allowing a larger proportion of AFP cases to be classified as nonpolio AFP based on more accurate virologic information. Isolation of Poliovirus Intratypic differentiation identified wild poliovirus type 1 from one case in 1996 and one case in 1997 (Table 1). These numbers probably underestimate actual wild poliovirus circulation in Nepal because few AFP cases were reported or investigated before July 1998. Reported by: Expanded Program on Immunization, Child Health Div, Ministry of Health, His Majesty's Government of Nepal; Expanded Program on Immunization, World Health Organization; United Nations Children's Fund National Office, Kathmandu. World Health Organization Regional Office of South East Asia, New Delhi, India. Global Program for Vaccines and Immunization, World Health Organization, Geneva, Switzerland. Respiratory and Enteric Viruses Br, Div of Viral and Rickettsial Diseases, National Center for Infectious Diseases; Vaccine Preventable Disease Eradication Div, National Immunization Program; State Br, Div of Applied Public Health Training, Epidemiology Program Office; and an EIS Officer, CDC. Editorial Note:Nepal is a geographic buffer between India, the world's largest reservoir for poliovirus, and China, which has been polio-free since 1995. During 1998, 85% of the world's polioviruses were isolated from polio cases in India (WHO, unpublished data, 1999); Uttar Pradesh and Bihar, two large Indian states on Nepal's southern border, accounted for 54% of India's polioviruses isolated. Uttar Pradesh also was the site of three polio outbreaks during 1997-1999 (7). Residents of Nepal and India may cross borders without passport or visa, and persons from border communities with low vaccination coverage frequently migrate in both directions. In Nepal, the most recent case of paralytic polio confirmed by wild poliovirus isolation in December 1997 occurred in an unvaccinated child residing in a border district. Another case that was clinically consistent with paralytic polio occurred in January 1999 in an Indian child who presented for care in southern Nepal, but from whom adequate stool specimens had not been collected. Because national surveillance for AFP has exceeded the international certification levels only since June 1999, confirmation of the absence of polioviruses is still pending. OPV3 coverage of infants aged 12 months ranged from 39% to 80% in Nepal Terai districts spanning the Indian border (WHO, unpublished data, 1999). In addition to improved routine vaccination and NIDs, intensified supplemental and house-to-house vaccination targeting children aged less than 5 years is needed in areas at high risk for poliovirus transmission. The polio eradication initiative is entering its most difficult and labor-intensive final phase. In a 1-year period, Nepal's RSOs developed a strong national AFP surveillance system (7). A factor contributing to rapid improvement of surveillance for polio eradication has been the participation of eight officers in the CDC Stop Transmission of Polio (STOP) initiative. STOP mobilizes additional trained personnel for 3-month polio eradication assignments in high-priority countries. STOP officers in Nepal worked with RSOs to strengthen AFP surveillance, plan NIDs and sub-NIDs, and mobilize other sectors in support of polio eradication. Fewer than 440 days remain to reach the target for global polio eradication by the end of 2000. Substantial and rapid improvement in NIDs and AFP surveillance has brought Nepal closer to the goal of eradication*****. Priorities for polio eradication in Nepal in 1999 and 2000 include 1) execution of high-quality NIDs and supplemental vaccination campaigns targeting high risk areas and populations (five monthly rounds will be synchronized with India during November 1999-March 2000); 2) maintenance of sensitive AFP surveillance, especially in the densely populated districts bordering India; and 3) improving routine OPV3 coverage. References
* SEAR comprises Bangladesh, Bhutan, Democratic Republic of Korea, India, Indonesia, Maldives, Myanmar, Nepal, Sri Lanka, and Thailand. ** Mass vaccination campaigns over a short period (days to weeks) in which two doses of oral poliovirus vaccine are administered to all children in the target group (usually aged less than 5 years), regardless of previous vaccination history, with an interval of 4-6 weeks between doses. *** Surveillance is conducted for neonatal tetanus, measles, acute flaccid paralysis, kala azar, malaria, and Japanese encephalitis. **** A confirmed case of polio has either wild poliovirus isolation, residual paralysis at 60 days after onset of paralysis, is lost to follow-up, or has died. ***** The polio eradication initiative in Nepal is supported by His Majesty's Government of Nepal, WHO, Rotary International, United Nations Children's Fund, U.S. Agency for International Development, the governments of Norway and Japan, and CDC. Table 1 Note: To print large tables and graphs users may have to change their printer settings to landscape and use a small font size. TABLE 1. Performance indicators for acute flaccid paralysis (AFP) surveillance -- Nepal, 1996-1999
* Annualized as of September 15, 1999. Return to top. Figure 1 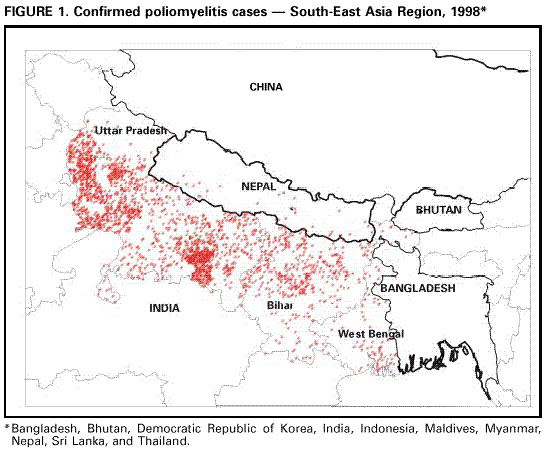 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 10/21/1999 |
||||||||||||||||||||||||||||||||||||||||||||
This page last reviewed 5/2/01
|