 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Q Fever Outbreak -- Germany, 1996In May 1996, the Health Department of Marburg-Biedenkopf in Marburg, Hessen, Germany, was notified of a cluster of persons with high and persistent fever who resided in a rural town (Rollshausen {1996 population: 300}) and in five surrounding towns approximately 0.5-2.0 miles from Rollshausen, in the district of Lohra. Serologic testing of some patients by local health authorities suggested acute Q fever. In Germany, Q fever is a reportable disease and 27-100 cases are reported annually; during 1995, no cases had been reported from Lohra. In July 1996, the Robert Koch Institute (RKI) was invited to assist in an investigation of this cluster. This report summarizes the investigation of this outbreak, which indicated a high attack rate of Q fever in persons residing near the zoonotic origin of infection. Before the outbreak, two flocks of sheep were kept near Rollshausen. One flock included 1000-2000 sheep that had been maintained on farm property northwest of Rollshausen from October 1995 through May 1996; lambing occurred both indoors and outdoors in December 1995 and January 1996. The second flock included 20 sheep and, since 1995, had been kept northeast of Rollshausen. To characterize the extent of and risk factors for this outbreak, RKI and local health authorities conducted a retrospective cohort study of all Rollshausen residents aged greater than or equal to 15 years. On July 10 and 11, 1996, a self-administered questionnaire was distributed to all households, and Coxiella burnetii antibody testing was offered to all residents. The questionnaire asked about symptoms since January 1, 1996, demographics, occupation, livestock exposure, drinking raw milk, tick bites, and outdoor activities. In addition, family doctors and hospitals serving the area were contacted to identify possible cases. A clinical case was defined as fever greater than or equal to 102.2 F ( greater than or equal to 39 C) lasting greater than 2 days and three or more symptoms (i.e., chills, sweats, severe headache, cough, aching muscles/joints, back pain, fatigue, or feeling ill) with onset after January 1, 1996. A laboratory-confirmed case was defined as a positive result for IgM C. burnetii antibodies. C. burnetii antibody testing was conducted by an enzyme-linked immunosorbent assay. Human serum was tested for IgG and IgM antibodies; in animal samples, IgG and IgM were not distinguished. Of the 239 eligible residents, 200 (84%) submitted a blood sample (120 {50%}) and/or completed the questionnaire (193 {81%}). A total of 49 (25%) of the 200 residents had either clinical (35 {18% of those completing questionnaire}) or laboratory-confirmed (35 {29% of those with antibody testing}) cases. Onsets of illness occurred from January through June; the first persons with laboratory-confirmed Q fever had onset in February (Figure_1). The 49 case-patients resided in all parts of Rollshausen. Attack rates (AR) were similar for males (24%) and females (25%) and did not vary by age. The most common symptoms were fatigue (80%), fever (78%), feeling ill (76%), and chills (71%). Of the 35 persons with clinical cases, four (11%) were hospitalized, and all had radiologically confirmed pneumonia. Risk for Q fever was twofold greater among residents who reported proximity to sheep (i.e., having been near a sheep stable or pasture) than those without this exposure (AR: 36% versus 19%; risk ratio {RR}=1.9; 95% confidence interval {CI}=1.2-3.1) and in residents who reported walking near the large sheep farm (AR: 33% versus 18%; RR=1.8; 95% CI=1.1-2.9). Although walking as a leisure activity was not an independent risk factor, among the 121 persons who reported walking as a leisure activity, the risk was nearly fourfold greater among those who had walked near the large sheep farm than those who had not (AR: 35% versus 9%; RR=3.8; 95% CI=1.5-9.2). Cases also were identified in 12 persons residing in towns other than Rollshausen (clinical {11} and/or laboratory {11}). Onsets of illness occurred from January through May. Eight persons resided in immediately neighboring towns, and four resided in a town approximately 19 miles south of Rollshausen -- the latter had spent weekends in a cottage adjacent to the large sheep farm in Rollshausen; all four residents of the town south of Rollshausen had onset of fever during March, and two required hospitalization. Of 20 sheep tested from the large flock, 15 were positive for C. burnetii antibodies, and the nine tested from the small flock were negative. Meteorologic data (obtained from the German Weather Service/Climate and Environmental Evaluation) indicated that from December 1995 through April 1996, the wind blew from the northwest (from the direction of the large sheep farm toward Rollshausen) an average of 17 days per month. In addition, there were 5.2 inches of rain compared with 7.4-15.0 inches during each of the 3 previous years. In January 1996, there were only 0.2 inches of rain, compared with 3.2-3.8 inches during each of the 3 previous years. Reported by: O Lyytikainen, MD, L Petersen, MD, B Schwartlander, MD, Robert Koch Institute, Berlin; P Matzdorff, MD, C Kuhnhen, MD, Health Dept of Marburg-Biedenkopf, Marburg; C Burger, Institute for Hygiene and Infectious Diseases of Animals of the Justus-Liebig Univ, Gie en; W Krug, State Veterinary Center, Marburg, Germany. T Ziese, MD, Swedish Institute for Infectious Disease Control, Stockholm, Sweden. European Program for Intervention Epidemiology Training, Brussels, Belgium. Viral and Rickettsial Zoonoses Br, Div of Viral and Rickettsial Diseases, National Center for Infectious Diseases, CDC. Editorial NoteEditorial Note: Q fever is a zoonotic disease caused by the rickettsial organism C. burnetii. Its most common reservoirs are domesticated ruminants, primarily cattle, sheep, and goats. Humans typically acquire Q fever by inhaling infectious aerosols and contaminated dusts generated by animals or animal products. Although many infections are asymptomatic, the protean manifestations of acute infections include self-limited influenza-like illness, hepatitis, pneumonia, myocarditis, pericarditis, and meningoencephalitis. Mortality associated with acute infections generally is low (less than 1%) but may be as high as 2.4% (1,2). Endocarditis and other chronic complications occur in a small proportion of patients and often are fatal. Although most cases of Q fever occur sporadically, three features of the organism and its route of transmission account for occasional outbreaks of clustered disease: 1) coxiellae are highly resistant to desiccation and to a variety of physical and chemical agents, and viable organisms may persist in contaminated soils for several months (3); 2) C. burnetii is among the most infectious of all bacteria, and inhalation of a single organism can produce infection in a susceptible host (4); and 3) airborne particles containing bacteria can initiate infections in susceptible hosts at distances of greater than or equal to 0.5 miles from the origin of the particles. The findings in this report indicate that the large sheep farm was the most likely source of this outbreak and the principal mode of transmission of C. burnetii was airborne. The lambing period in December and January immediately preceded the outbreak, and the first persons documented to have IgM antibody had onsets of illness in February, consistent with the average 20-day incubation period for Q fever (4). Outbreaks of Q fever commonly occur after lambing because C. burnetii is reactivated in ewes during pregnancy. Because of multiplication of C. burnetii in the placental villi, high numbers of coxiellae (i.e., as many as one billion organisms per gram of placenta) may be present in placentae, amniotic fluid, and fetal membranes (3). The attack rate among Rollshausen residents was high (25%) and case-patients resided in all parts of the town, suggesting a ubiquitous exposure consistent with airborne transmission. This finding probably reflects the outdoor lambing, the exceptionally dry weather, and the wind pattern (blowing from the direction of the large sheep farm toward the town). Infected birth products can contaminate the ground and dry periods may enhance the formation and propagation of infectious dusts and aerosols (1,3). Other associated factors include the high percentage of infected ewes (75%), the increased risk among persons who had been in contact with sheep and walking in the areas near the large sheep farm, and the occurrence of Q fever among the four persons who had spent weekends next to the large sheep farm. Tetracycline compounds are the treatment of choice for persons with Q fever. Doxycycline 100 mg twice a day for 15-21 days is recommended for patients with acute disease. The optimal regimen for chronic disease has not been established but generally involves prolonged treatment with a tetracycline in combination with rifampin or trimethoprim-sulfamethoxazole, administered for a minimum of 2-3 years (4). Effective control and prevention of Q fever in humans requires the identification of infections in domesticated animal populations. When C. burnetii infection is suspected or detected in a sheep flock, prevention efforts should focus on reducing environmental contamination from infected placental membranes and aborted materials and subsequent airborne spread and inhalation of C. burnetii. Lambing should not take place outdoors, and separate indoor facilities should be appointed for parturition. After parturition, appropriate disposal of placentae, fetal membranes, and aborted material is critical. Birth products should be destroyed by incineration, and the lambing area should be treated with an effective disinfectant (e.g., 1% Lysol{Registered} * or 5% hydrogen peroxide). Persons at risk for Q fever include abattoir workers, dairy farmers, workers involved in meat or dairy processing, and veterinarians. When livestock operations are close to human habitation, communication between veterinarians, local public health officials, and health-care providers facilitates recognition of disease in exposed persons. Human Q fever vaccine is commercially available in Australia and Eastern Europe, but not in Germany or in the United States. References
* Use of trade names and commercial sources is for identification only and does not imply endorsement by the Public Health Service or the U.S. Department of Health and Human Services. Figure_1 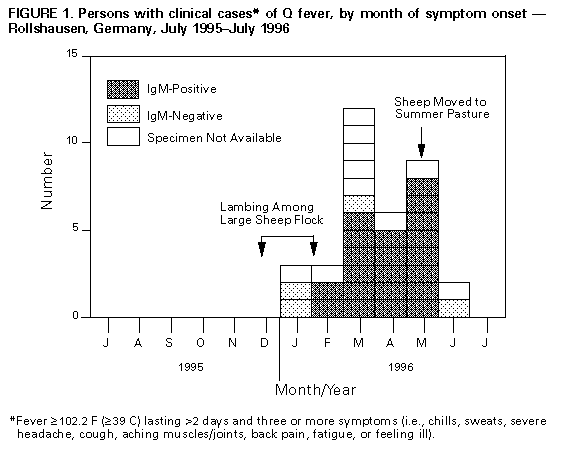 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 09/19/98 |
|||||||||
This page last reviewed 5/2/01
|