 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Progress Toward Poliomyelitis Eradication -- People's Republic of China, 1990-1996In 1988, the Western Pacific Region of the World Health Organization (WHO) adopted a resolution to eradicate poliomyelitis from the region by the end of 1995. The People's Republic of China has made rapid progress toward this goal by implementing the four WHO-recommended strategies for polio eradication: 1) achieving and maintaining high routine vaccination coverage levels among children aged less than 1 year with at least three doses of oral poliovirus vaccine (OPV); 2) administering supplementary doses of OPV to all young children during National Immunization Days (NIDs) * to rapidly interrupt poliovirus transmission; 3) establishing sensitive systems of surveillance for acute flaccid paralysis (AFP) and virologic surveillance for poliovirus; and 4) conducting "supplementary immunization activities" (SIAs) -- localized campaigns targeted at high-risk areas where poliovirus transmission is most likely to persist at low levels. These strategies have resulted in the apparent interruption of wild poliovirus transmission in China. In 1995, no indigenous wild polioviruses were detected despite a strengthened surveillance system. This report describes progress toward achievement of the eradication goal and updates the status of polio-eradication efforts in China during 1990-1996 (1). Routine Vaccination The use of OPV in China began in 1964. WHO's Expanded Program on Immunization was started in China in 1978, and the cold chain was strengthened in 1982 (2). Routine vaccination coverage rates increased from 79% in 1983 to greater than 90% in the 1990s. In 1995, only two of 30 provinces reported coverage below 90%: Jiangsu (84%) and Guangdong (88%) (3). Focal areas of lower vaccination coverage persist at lower administrative levels, and routine vaccination services are more difficult to provide to some persons (e.g., children of migrant workers). Subnational and National Immunization Days Initial supplemental vaccination activities began during 1990-1993 with limited provincewide subnational immunization days (SNIDs). During this period, there were province-specific differences in the age groups targeted for vaccination, number of participating counties, number of rounds, and timing of the SNIDs. The first two rounds of coordinated NIDs were conducted in December 1993 and January 1994; 82 million and 83 million children aged 0-3 years, respectively, were vaccinated (2). Three NIDs were conducted, with subsequent NIDs during December 1994-January 1995 and December 1995-January 1996. Routine Poliomyelitis Surveillance A routine surveillance system for notifiable diseases, including polio, has been operational in China since the 1950s. Polio cases are diagnosed by individual health professionals' clinical judgment without use of a standard case definition; case-specific information and virologic confirmation of reported cases are not available. This system has documented a decline in polio cases, from 9625 cases in 1981, to 5065 cases in 1990 (when SNIDs were initiated), to 62 cases in 1995 (Figure_1). Acute Flaccid Paralysis Surveillance A surveillance system to detect all cases of AFP was initiated in 1990 and includes case-based information, electronic data transfer from the provincial to the central level, a network of 30 provincial laboratories to process stool specimens for isolation of poliovirus, and one national reference laboratory for intratypic differentiation of poliovirus as wild or vaccine-related. An annual nonpolio AFP rate of greater than or equal to 1 per 100,000 persons aged less than 15 years is used as an indicator of effective surveillance. During 1993-1995, the national nonpolio AFP rate increased from 0.4 to 1.5. WHO standard guidelines recommend investigation of reported AFP cases within 48 hours and collection of two stool specimens 24-48 hours apart from each AFP case-patient to determine the presence of poliovirus. During 1992-1995, case investigations performed within 48 hours (target: 80%) increased from 34% to 98% and the percentage of cases from which two stool samples were collected increased from 11% to 88%. In 1995, adequate stool specimens (i.e., collection of two samples within 14 days of paralysis, collection at least 24 hours apart, sufficient quantity, not desiccated, and arrival on ice or frozen ice packs at the provincial laboratory with complete documentation) was reported to be 64% (target: 80%) (3). The improved virologic surveillance system isolated the last indigenous wild polioviruses (all poliovirus type 1) in China in 1994. Supplemental Immunization Activities During November 1995-April 1996, four cases of AFP were reported in persons who crossed the border from Myanmar into Yunnan province in southwestern China. Wild polioviruses type 1 (n=two) and type 3 (n=two) were isolated and confirmed by genomic sequencing to be imported viruses (i.e., representing genotypes not previously detected in China) (3). During March and April 1996, two rounds of SIAs were conducted in all prefectures along the Yunnan-Myanmar border involving house-to-house and fixed-site vaccination of approximately 1 million children aged 0-3 years. Since then, there have been no imported AFP cases from which wild poliovirus was isolated. Active surveillance for AFP (e.g., weekly visits of hospitals and village searches for suspected cases) is continuing along the border prefectures between Myanmar and Yunnan province. Reported by: Z Dai, Z Wang, J Yu, Ministry of Health; K Wang, R Zhang, L Zhang, G Wang, T Xu, Chinese Academy of Preventive Medicine; World Health Organization, Beijing, People's Republic of China. Expanded Program on Immunization Unit, Western Pacific Regional Office, World Health Organization, Manila, Philippines. Div of Viral and Rickettsial Diseases, National Center for Infectious Diseases; Polio Eradication Activity, National Immunization Program, CDC. Editorial NoteEditorial Note: Since 1990, China has made substantial progress toward polio eradication. The use of SIAs, including three NIDs conducted during 1993-1996, has resulted in the apparent elimination of wild poliovirus. Since 1994, no cases of indigenous wild poliovirus infection have been detected in China despite substantial improvements in surveillance. In addition, the number of reported polio cases decreased 99% during 1990-1995. The remaining cases since 1994 were reported as polio based on clinical criteria (none were laboratory confirmed) and may represent misclassification of other causes of polio-like illnesses (e.g., Guillian-Barre syndrome or transverse myelitis) and vaccine-associated polio. China remains at risk for importation of wild poliovirus from areas where polio continues to be endemic. Therefore, the Ministry of Health in China plans to continue supplementary vaccination activities in all provinces for at least 3 more years (targeting greater than 70 million children during December 1996-January 1997) to achieve and maintain high immunity levels in provinces at risk for wild poliovirus importation, to compensate for focal areas of low routine vaccination coverage and inadequate AFP surveillance in some areas, and to ensure that any remaining reservoir of wild poliovirus is eliminated. Areas at "high-risk" for poliovirus transmission include those 1) sharing a border with adjacent countries with endemic polio, 2) in which wild poliovirus has been isolated during the last 3 years, 3) characterized by an AFP rate of less than 1 per 100,000 persons aged less than 15 years, 4) in which the stool collection rate is less than 60%, and 5) characterized by low routine vaccination coverage (less than 90%) (3). SNIDs will especially target unvaccinated, minority, and migrant workers' children. The eradication of polio from China can be certified only after no cases of infection with wild polioviruses have been detected for at least 3 consecutive years in the presence of an effective AFP surveillance system. China can improve the effectiveness of its AFP surveillance system by 1) upgrading the polio laboratory network, 2) implementing a virologic case definition for confirmed polio cases; and 3) improving the completeness and timeliness of investigation and laboratory examination of all AFP cases. Continued international ** support is necessary to accelerate progress toward polio eradication and to meet the requirements for eventual certification of the absence of wild poliovirus in China. References
* Mass campaigns over a short period (days to weeks) in which two doses of OPV are administered to all children in the target age group, regardless of prior vaccination history, with an interval of 4-6 weeks between doses. ** The polio-eradication initiative is supported by a coalition of organizations that includes WHO, the United Nations Children's Fund (UNICEF), the World Bank, the Japanese International Cooperative Agency, the Agency for Cooperation in Health, other bilateral and multilateral organizations, and Rotary International. Figure_1 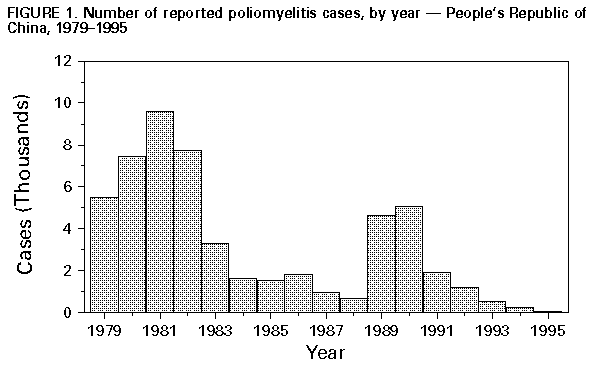 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 09/19/98 |
|||||||||
This page last reviewed 5/2/01
|