 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Update: Diphtheria Epidemic -- New Independent States of the Former Soviet Union, January 1995-March 1996Epidemic diphtheria reemerged in the New Independent States (NIS) of the former Soviet Union, beginning in the Russian Federation in 1990 and affecting all 15 NIS by the end of 1994 (1,2). Approximately 90% of all diphtheria cases reported worldwide during 1990-1995 were reported from the NIS (World Health Organization {WHO}, unpublished data, 1996). During 1993-1994, WHO, partner organizations, and national ministries of health developed a strategy to control the epidemic with a priority goal of achieving coverage of greater than 90% among persons aged greater than or equal to 3 years with a single dose of diphtheria toxoid through mass vaccination campaigns and achieving coverage for routine childhood vaccination (i.e., four doses of diphtheria and tetanus toxoids and pertussis vaccine by age 2 years) of greater than 95%. This report summarizes data provided to WHO about the incidence of diphtheria (Table_1) and efforts to implement control measures in the NIS during 1995 and January-March 1996. Overall, from 1994 to 1995, reported diphtheria cases in the NIS increased 5.2%, from 47,628 to 50,412 cases (Figure_1) with approximately 1500 deaths in 1995. Since the epidemic began in 1990, approximately 125,000 cases and 4000 deaths have been reported in the NIS. The number of cases in 1995 reflects an 11% decrease in reported cases in the Russian Federation but a near doubling of cases from other NIS. However, following expanded control efforts, by the end of 1995 most countries began reporting decreases from the same periods in 1994. During January-March 1996, a total of 6179 diphtheria cases were reported in the NIS, a 59% decrease from the 14,931 cases reported during the same period in 1995. Russian Federation. During 1990-1995, approximately 97,000 diphtheria cases and 2500 deaths were reported in the Russian Federation. Reported cases decreased from 39,582 (26.9 cases per 100,000 population) in 1994 to 35,652 (24.3) in 1995. Reported deaths decreased from 1104 (case-fatality rate: 2.8%) in 1994 to 740 (2.1%) in 1995. Vaccination coverage among persons aged greater than or equal to 18 years with at least one dose of diphtheria toxoid during the preceding 10 years had increased to 70%-80% by the end of 1995 from an estimated 20% in 1990. In 1995, reported coverage with a primary series of diphtheria toxoid among children aged 12-23 months was 92.7%. Western NIS and Baltic countries. In Ukraine, reported cases nearly doubled, from 2990 (5.8 cases per 100,000) in 1994 to 5280 (10.3) in 1995. Mass vaccination campaigns targeting adults were conducted in seven of the 27 regions in April 1995; the remaining regions were targeted for mass vaccination in late 1995 and in 1996. Overall, vaccine coverage in adults was an estimated 60%. In Belarus, Latvia, and Lithuania, increases in diphtheria cases were reported in 1995 (Table_1); however, mass adult vaccination efforts in 1995 and 1996 achieved coverage of 60%-80%, and efforts to complete vaccination of all adults are continuing. In Moldova, reported cases increased from 376 (8.5 cases per 100,000 population) in 1994 to 418 (9.4) in 1995. A mass vaccination campaign was conducted in the summer and fall of 1995, increasing coverage among adults to greater than 80%; a sustained decline in cases began in October 1995. In Estonia, cases increased from seven to 19, almost exclusively among ethnic Russians in border areas. Approximately 30% of adults were vaccinated during the campaign in 1995. Central Asia. All of the Central Asian republics reported increases in reported diphtheria cases in 1995. In Tajikistan, reported cases increased from 1912 (31.8 cases per 100,000 population) in 1994 to 4455 (73.0) in 1995, with cases reported from all regions of the country. A mass vaccination campaign was conducted during October 1995 and reached greater than 90% of adults (aged less than or equal to 50 years). The other Central Asian republics (Kazakstan, Kyrgyzstan, Uzbekistan, and Turkmenistan) reported a total of 2523 cases in 1995 (Table_1), representing a 135% increase over the total reported cases in 1994. Mass vaccination campaigns targeting both adults and children were initiated in Kazakstan, Kyrgyzstan, and Uzbekistan in 1995 and are to be completed during 1996. In Turkmenistan, vaccination campaigns for children were conducted in 1995; adults are being targeted in 1996. Caucasus. Georgia reported a 43% increase in diphtheria cases during 1995 (419) over 1994 (294), and Azerbaijan reported a 5% increase; Armenia continued to report few cases (Table_1). Following revaccination efforts among schoolchildren, resulting in coverage of 96%, Azerbaijan conducted mass vaccination of persons aged 0-55 years during November 1995, achieving coverage of 95%. Reported cases in Azerbaijan during January-March 1996 are 85% below the same period in 1995. In Georgia and Armenia, adult vaccination campaigns began in 1995; however, coverage is less than 60% in some regions. Reported by: Regional Office for Europe, World Health Organization, Copenhagen, Denmark. International Federation of Red Cross and Red Crescent Societies, Geneva, Switzerland. Child Vaccine Preventable Disease Br, Epidemiology and Surveillance Div, National Immunization Program, CDC. Editorial NoteEditorial Note: The findings in this report highlight the first evidence of progress toward controlling the diphtheria epidemic in the NIS. WHO continues to consider the epidemic an international public health emergency. At the time of the disintegration of the former Soviet Union in 1991, all NIS relied on supplies of vaccine and antitoxin from Russia, and most lacked the financial resources to procure them from the international market. Governmental and nongovernmental organizations, such as the United States Agency for International Development, European Community Humanitarian Office, International Federation of Red Cross and Red Crescent Societies, WHO, United Nations Children's Fund (UNICEF), the government of Japan, and others, have initiated an effort, monitored by the Interagency Immunization Coordination Committee, to mobilize the needed materials (i.e., vaccine, syringes, needles, antitoxin, and antibiotics) for all of the NIS except Russia, which continues to be self-sufficient for all these materials. This epidemic has been characterized by a high proportion of cases among adults; children were the predominant age group affected in previous epidemics (3). Much of the variation in incidence rates in the NIS during 1994 and 1995 was attributed to differences in the timing of the onset of the epidemic between countries; however, the success in implementing control measures is beginning to affect incidence rates. In countries that have achieved high coverage among adults, diphtheria incidence has decreased substantially, regardless of the level of incidence before the vaccination campaigns. The impact of mass vaccination of the adult population on slowing the course of the epidemic underscores the need for rapidly completing the vaccination campaigns in the other countries. All the NIS have attempted to increase childhood vaccination coverage, including decreasing perceived contraindications to childhood vaccination and increasing the routine use of full-strength vaccine preparations in the primary series; routine childhood coverage is now high in most countries. Most countries also have reinstituted a school-entry booster dose and many of the national mass campaigns have included preschool-aged and school-aged children and adolescents in the target population. Other control measures are important in efforts to further reduce transmission of diphtheria. Most of the NIS have made efforts to improve early diagnosis and treatment of cases. Moldova, Lithuania, and Azerbaijan have adopted the WHO recommendation to use empiric antibiotic treatment for close contacts of persons with diphtheria. Providing additional doses of diphtheria toxoid to at least certain portions of the population (e.g., adults aged 30-50 years in whom risk for severe disease and death is highest) will be needed to fully protect all persons. Because some epi-demiologic features of this epidemic differ from those of the prevaccine era and the epidemiologic situation may vary among the NIS, ongoing surveillance and additional field studies are needed to track the course of the epidemic in each country. The reemergence of epidemic diphtheria throughout the NIS after greater than 30 years of successful control and the reports of greater than 20 imported cases from the NIS into Europe (1,2) and Mongolia (WHO, unpublished data, 1996) emphasize the need for achieving and maintaining high levels of diphtheria immunity among both adults and children in the United States and other countries. The Advisory Committee on Immunization Practices recommends that all children receive a routine series of five doses of diphtheria toxoid-containing vaccine with doses at ages 2, 4, 6, and 12-20 months and 4-6 years; boosters of diphtheria and tetanus toxoids should then be administered beginning at age 11-12 years (provided at least 5 years have passed since the last dose of diphtheria toxoid-containing vaccine) and every 10 years thereafter (4-6). Travelers to areas with diphtheria activity should review their vaccination status and receive age-appropriate vaccinations as needed. References
TABLE 1. Number and rate of diphtheria cases, 1994-1995, percentage change in number of cases from 1994 to 1995 and first
quarter of 1995 to first quarter of 1996, and level of coverage with diphtheria toxoid, 1995, by country -- New Independent
States of the former Soviet Union
===========================================================================================================================================
% Change in number of cases % Coverage, 1995
1994 1995 ----------------------------- -----------------------
-------------- ------------- 1994 to 1st quarter 1995 to Primary series 1 dose,
Country Population * Cases Rate + Cases Rate 1995 1st quarter 1996 at age 1 year adult
---------------------------------------------------------------------------------------------------------------------
Armenia 3.6 36 1.0 29 0.8 -19 -14 NA & 60 @
Azerbaijan 7.5 841 11.3 883 11.7 5 -85 >90 95
Belarus 10.1 230 2.3 322 3.2 40 -46 96 65
Estonia 1.6 7 0.5 19 1.2 171 -29 >90 30 **
Georgia 5.4 294 5.4 419 7.7 43 -28 NA 65 @
Kazakstan 17.0 489 2.9 1105 6.5 126 -31 93 50
Kyrgyzstan 4.7 299 6.4 693 14.6 132 -15 93 70
Latvia 2.6 250 9.7 369 14.4 48 -71 NA 30 ++
Lithuania 3.6 38 1.0 43 1.2 13 -69 97 70
Moldova 4.4 376 8.5 418 9.4 11 -85 NA >80
Russia 146.7 39,582 26.9 35,652 24.3 -10 -59 93 75
Tajikistan 6.1 1912 31.8 4455 73.0 133 -42 94 >95
Turkmenistan 4.1 60 1.5 87 2.1 45 90 92 && 30
Ukraine 51.3 2990 5.8 5280 10.3 77 -37 NA 60
Uzbekistan 22.8 224 1.0 638 2.8 185 -40 NA 50
Total 298.5 47,628 16.1 50,412 16.9 5 -59 -- --
---------------------------------------------------------------------------------------------------------------------
* In millions.
+ Per 100,000 population per year.
& Not available.
@ Adult coverage for Armenia and Georgia is based on less complete data than for other countries.
** Approximately 75% of adults were vaccinated during 1985-1987.
++ The Latvian Ministry of Health considers coverage to be three doses for adults; an estimated 60%-70% have received at least one
dose.
&& Coverage figure is for four doses of diphtheria and tetanus toxoids and pertussis vaccine (primary series plus first revaccination) at
age 2 years.
===========================================================================================================================================
Return to top. Figure_1 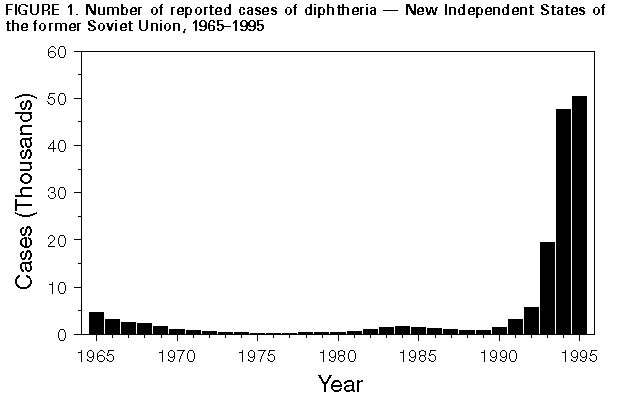 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 09/19/98 |
|||||||||
This page last reviewed 5/2/01
|